Metabolic Syndrome
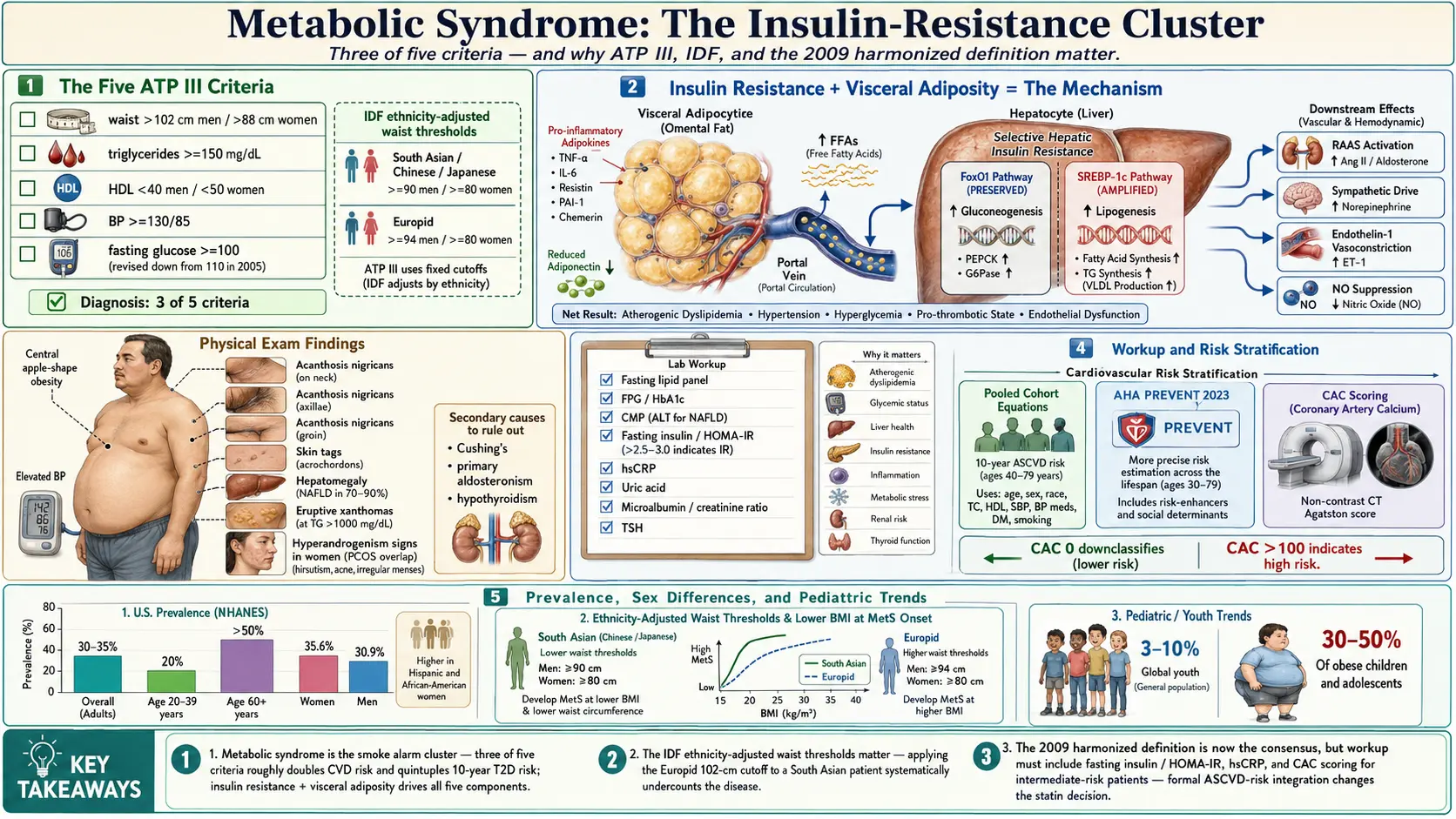
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
- Featured Videos
1. Overview
Metabolic syndrome (MetS) is a cluster of interrelated metabolic abnormalities that together substantially increase the risk of cardiovascular disease (CVD), type 2 diabetes mellitus (T2DM), and all-cause mortality. Rather than a discrete disease entity, it represents a convergent pathophysiologic state characterized by central adiposity, dyslipidemia, hypertension, and impaired glucose metabolism. The unifying pathophysiologic thread linking these components is insulin resistance, though the precise mechanistic hierarchy remains under active investigation.
The syndrome has been defined by multiple major organizations including the World Health Organization (WHO), the National Cholesterol Education Program Adult Treatment Panel III (NCEP ATP III), the International Diabetes Federation (IDF), and the American Heart Association/National Heart, Lung, and Blood Institute (AHA/NHLBI). Harmonization efforts in 2009 produced a joint statement that unified the diagnostic criteria, acknowledging population-specific waist circumference thresholds as a key refinement.
Recognition of metabolic syndrome carries immediate clinical utility: it prompts lifestyle intervention, guides pharmacologic decision-making across multiple organ systems simultaneously, and identifies patients warranting intensified surveillance for atherosclerotic cardiovascular disease (ASCVD) and T2DM.
2. Epidemiology
Metabolic syndrome has reached pandemic proportions globally, closely tracking the worldwide rise in obesity and sedentary lifestyles. Prevalence varies substantially by the diagnostic criteria applied, the population studied, age, sex, and ethnicity.
- United States: NHANES data consistently demonstrate an overall adult prevalence of 30–35%. Prevalence increases steeply with age: approximately 20% in adults aged 20–39 years, rising to over 50% in those aged 60 years and older.
- Global estimates: The IDF estimates that 20–25% of the world's adult population meets criteria. Rapid urbanization in South Asia, Southeast Asia, and sub-Saharan Africa is driving accelerating prevalence in previously low-risk populations.
- Sex differences: In the United States, prevalence is slightly higher in women (35.6%) than men (30.9%), particularly among Hispanic and African-American women, though this pattern varies internationally.
- Ethnic disparities: South Asians, Hispanics, and East Asians develop metabolic syndrome at lower body mass index (BMI) thresholds than individuals of European ancestry, reflecting differential visceral fat accumulation and insulin sensitivity. This underpins population-specific waist circumference cutoffs in the IDF definition.
- Pediatric trends: Metabolic syndrome in children and adolescents, once rare, now affects an estimated 3–10% of youth globally and up to 30–50% of obese children.
- Secular trends: Despite modest reductions in dyslipidemia attributable to statin use, rates of abdominal obesity and impaired fasting glucose continue to rise, sustaining overall prevalence at high levels.
3. Pathophysiology
The pathophysiology of metabolic syndrome is multifactorial and bidirectionally reinforcing. Insulin resistance and central (visceral) adiposity are widely regarded as the two core mechanistic drivers, though their temporal and causal relationship is debated.
Insulin Resistance
Insulin resistance refers to a subnormal biological response to a given insulin concentration in peripheral tissues — principally skeletal muscle, hepatocytes, and adipocytes. At the molecular level, defects occur at multiple nodes of insulin signaling:
- Reduced tyrosine phosphorylation of insulin receptor substrate-1 (IRS-1) and IRS-2, often mediated by serine phosphorylation from inflammatory kinases (IKK-β, JNK-1).
- Impaired activation of phosphatidylinositol 3-kinase (PI3K) and its downstream effector Akt/PKB, diminishing GLUT4 translocation to the plasma membrane in skeletal muscle and adipose tissue.
- Intact or even enhanced MAPK/ERK signaling, which mediates mitogenic and pro-inflammatory effects of insulin independently of metabolic effects — a selective insulin resistance contributing to endothelial dysfunction and vascular smooth muscle proliferation.
In the liver, insulin resistance paradoxically preserves gluconeogenesis (through the FoxO1 pathway) while lipogenesis continues via SREBP-1c, producing the characteristic combination of hyperglycemia and hypertriglyceridemia.
Visceral Adiposity and Adipokine Dysregulation
Visceral (intra-abdominal) adipose tissue is metabolically distinct from subcutaneous adipose tissue. It is characterized by:
- Higher lipolytic activity and greater sensitivity to catecholamine-driven free fatty acid (FFA) release into the portal circulation.
- Increased macrophage infiltration and secretion of pro-inflammatory adipokines: tumor necrosis factor-alpha (TNF-α), interleukin-6 (IL-6), plasminogen activator inhibitor-1 (PAI-1), resistin, and chemerin.
- Reduced secretion of adiponectin, an insulin-sensitizing, anti-inflammatory, and anti-atherosclerotic adipokine. Hypoadiponectinemia is one of the strongest biochemical correlates of metabolic syndrome.
- Ectopic lipid deposition in liver (non-alcoholic fatty liver disease), skeletal muscle, pancreatic beta cells, and the myocardium — directly impairing organ-specific insulin signaling through lipotoxicity mediated by diacylglycerol and ceramide accumulation.
Dyslipidemia
The dyslipidemia of metabolic syndrome — hypertriglyceridemia, low HDL-C, and elevated small dense LDL particles — is mechanistically linked to insulin resistance in the liver and adipose tissue:
- Elevated hepatic VLDL synthesis driven by excess portal FFA flux and de novo lipogenesis.
- Impaired lipoprotein lipase (LPL) activity, reducing VLDL clearance and triglyceride hydrolysis.
- Accelerated CETP-mediated exchange of cholesterol ester from HDL to VLDL, depleting HDL-C and generating small, dense, oxidation-prone LDL particles.
Hypertension
Multiple mechanisms link insulin resistance and visceral obesity to hypertension:
- Compensatory hyperinsulinemia activates the sympathetic nervous system and stimulates renal sodium reabsorption through the Na-K ATPase pump and ENaC channels.
- Hyperinsulinemia impairs nitric oxide (NO) synthesis in vascular endothelium, reducing vasodilation.
- Activation of the renin-angiotensin-aldosterone system (RAAS) by adipose-derived angiotensinogen.
- Increased endothelin-1 production and reduced prostacyclin synthesis shift the vasoactive balance toward vasoconstriction.
Chronic Low-Grade Inflammation and Oxidative Stress
Metabolic syndrome is accompanied by a state of chronic, low-grade systemic inflammation evidenced by elevated high-sensitivity C-reactive protein (hsCRP), IL-6, TNF-α, and fibrinogen. Adipose tissue macrophage polarization toward the M1 pro-inflammatory phenotype, gut microbiome dysbiosis with increased lipopolysaccharide (LPS) translocation (metabolic endotoxemia), and mitochondrial dysfunction all contribute to oxidative stress and perpetuate insulin resistance.
Gut Microbiome
Emerging evidence implicates dysbiosis of the intestinal microbiome in metabolic syndrome pathogenesis. Reduced microbial diversity, depletion of short-chain fatty acid (SCFA)-producing bacteria (e.g., Faecalibacterium prausnitzii, Akkermansia muciniphila), and increased LPS-producing gram-negative species contribute to metabolic endotoxemia, altered bile acid metabolism, and impaired intestinal barrier integrity — all reinforcing systemic insulin resistance.
4. Etiology and Risk Factors
Metabolic syndrome arises from the interaction of genetic susceptibility and environmental exposures. No single etiology is sufficient; rather, the syndrome represents a gene-environment interaction played out across decades.
Non-Modifiable Risk Factors
- Genetics: Heritability estimates for individual components range from 30–70%. Genome-wide association studies (GWAS) have identified loci in FTO, MC4R, TCF7L2, PPARG, KCNJ11, and lipid-related genes. No single gene determines metabolic syndrome; polygenic risk scores are under development.
- Age: Prevalence rises sharply with age due to progressive sarcopenia, reduced physical activity, altered adipose distribution, and declining sex hormone levels.
- Sex and hormonal status: Postmenopausal women experience rapid redistribution of fat from subcutaneous to visceral depots as estrogen declines, dramatically increasing metabolic risk.
- Ethnicity: South Asians and East Asians have greater visceral adiposity at any given BMI. Hispanic individuals show higher rates of insulin resistance. African-Americans have higher hypertension prevalence but often lower triglycerides, illustrating ethnic heterogeneity in component expression.
- Intrauterine programming: Low birth weight and exposure to maternal diabetes or undernutrition program a "thrifty phenotype" that predisposes to insulin resistance and obesity in later life.
Modifiable Risk Factors
- Physical inactivity: Sedentary behavior is independently associated with insulin resistance, visceral adiposity, dyslipidemia, and hypertension beyond its effect on body weight.
- Dietary patterns: High consumption of refined carbohydrates, sugar-sweetened beverages, saturated and trans fats, and ultra-processed foods promotes visceral adiposity and insulin resistance. The Western dietary pattern is a potent driver of metabolic syndrome globally.
- Obesity: Particularly visceral/central obesity. BMI alone underestimates risk; waist circumference and waist-to-hip ratio are stronger predictors.
- Sleep disorders: Obstructive sleep apnea (OSA) causes intermittent hypoxia, sympathetic activation, and RAAS stimulation — independently contributing to all components of metabolic syndrome. Short sleep duration (less than 6 hours/night) is associated with a 40–60% increased risk.
- Psychosocial stress: Chronic stress elevates cortisol, promoting visceral fat deposition, gluconeogenesis, sodium retention, and appetite dysregulation via the hypothalamic-pituitary-adrenal (HPA) axis.
- Medications: Atypical antipsychotics (olanzapine, clozapine), glucocorticoids, some HIV antiretrovirals, and certain beta-blockers and thiazide diuretics at high doses contribute to metabolic dysregulation.
- Gut microbiome dysbiosis: Modifiable through diet, probiotics, and avoidance of unnecessary antibiotics.
- Endocrine disruptors: Bisphenol A (BPA), phthalates, persistent organic pollutants (POPs), and per- and polyfluoroalkyl substances (PFAS) impair adipokine signaling, thyroid function, and insulin receptor sensitivity.
5. Clinical Presentation
Metabolic syndrome is typically asymptomatic in early stages; it is identified through routine screening rather than symptom-driven evaluation. Clinical findings are those of its component conditions and associated comorbidities.
Physical Examination Findings
- Central obesity: Increased waist circumference measured at the iliac crest level, often accompanied by an apple-shaped body habitus. The buffalo hump and moon facies of Cushing syndrome should be excluded when obesity is severe or rapid in onset.
- Acanthosis nigricans: Velvety, hyperpigmented plaques in the neck, axillae, and groin — a cutaneous marker of insulin resistance and hyperinsulinemia. Prevalence is higher in Hispanic and African-American patients.
- Skin tags (acrochordons): Commonly associated with insulin resistance and obesity.
- Hypertension: Elevated blood pressure on office measurement; white coat effect is common and ambulatory blood pressure monitoring may be warranted.
- Xanthomas and xanthelasmas: Eruptive xanthomas on the buttocks and extensor surfaces suggest severe hypertriglyceridemia (greater than 1000 mg/dL).
- Hepatomegaly: Palpable or percussion-evident liver enlargement may indicate non-alcoholic fatty liver disease (NAFLD), present in 70–90% of patients with metabolic syndrome.
- Signs of hyperandrogenism in women: Hirsutism, acne, and oligomenorrhea suggest co-existing polycystic ovary syndrome (PCOS), which shares pathophysiologic mechanisms with metabolic syndrome.
Symptoms
When present, symptoms reflect end-organ effects:
- Fatigue and daytime somnolence (often from OSA or hyperglycemia)
- Polyuria and polydipsia (if overt T2DM has developed)
- Headache, epistaxis, visual disturbances (hypertensive urgency)
- Exertional dyspnea or chest pain (established ASCVD)
- Right upper quadrant discomfort (NAFLD, cholelithiasis)
- Erectile dysfunction and decreased libido (vascular and hormonal effects)
6. Diagnosis
Metabolic syndrome is diagnosed by fulfilling criteria from established clinical definitions. The most widely applied definitions in clinical practice are the NCEP ATP III and the harmonized 2009 joint statement.
NCEP ATP III Criteria (2001, Updated 2005)
Metabolic syndrome is diagnosed when 3 or more of the following 5 criteria are met:
- Waist circumference: Greater than 102 cm (40 in) in men; greater than 88 cm (35 in) in women.
- Triglycerides: 150 mg/dL (1.7 mmol/L) or greater, or on drug treatment for elevated triglycerides.
- HDL-cholesterol: Less than 40 mg/dL (1.03 mmol/L) in men; less than 50 mg/dL (1.29 mmol/L) in women, or on drug treatment for reduced HDL-C.
- Blood pressure: Systolic 130 mmHg or greater or diastolic 85 mmHg or greater, or on antihypertensive drug treatment in a patient with a history of hypertension.
- Fasting glucose: 100 mg/dL (5.6 mmol/L) or greater, or on drug treatment for elevated glucose.
IDF Criteria (2005)
The IDF definition requires central obesity as a mandatory criterion plus any 2 of the remaining 4 components. The IDF uses ethnicity-specific waist circumference cutoffs:
- Europids: greater than 94 cm (men), greater than 80 cm (women)
- South Asians, Chinese, Japanese, South and Central Americans: greater than 90 cm (men), greater than 80 cm (women)
- Sub-Saharan Africans, Middle Eastern, and Mediterranean: use Europid cutoffs pending further data
Harmonized 2009 Joint Criteria (AHA/NHLBI/IDF/IAS/IASO)
The harmonized definition — now the most widely accepted — requires any 3 of 5 criteria, using population- and country-specific waist circumference thresholds (deferring the mandatory central obesity requirement from the IDF definition). This approach acknowledges the clinical reality that not all patients with metabolic syndrome are centrally obese by standard cutoffs, particularly East Asians with metabolically adverse phenotypes at lower waist circumferences.
WHO Criteria (1998)
Requires evidence of insulin resistance (impaired glucose tolerance, impaired fasting glucose, T2DM, or other insulin resistance measures) plus 2 of: hypertension (greater than 140/90 mmHg), dyslipidemia (triglycerides 150 mg/dL or greater or HDL-C less than 35 mg/dL in men, less than 39 mg/dL in women), central obesity (waist-to-hip ratio greater than 0.9 in men, greater than 0.85 in women, or BMI greater than 30 kg/m²), or microalbuminuria (urinary albumin excretion rate 20 mcg/min or greater). Less commonly used in clinical practice due to complexity.
Laboratory Evaluation
Initial laboratory workup should include:
- Fasting lipid panel (LDL-C, HDL-C, triglycerides, total cholesterol)
- Fasting plasma glucose and/or HbA1c
- Comprehensive metabolic panel (hepatic transaminases for NAFLD, renal function, electrolytes)
- Fasting insulin and HOMA-IR calculation: HOMA-IR = (fasting glucose [mmol/L] × fasting insulin [mIU/L]) / 22.5; values greater than 2.5–3.0 indicate insulin resistance
- High-sensitivity CRP (hsCRP) — values greater than 3.0 mg/L add cardiovascular risk stratification
- Uric acid (elevated in 50–75% of patients with metabolic syndrome)
- Urinalysis with microalbumin-to-creatinine ratio
- TSH (to exclude hypothyroidism as a contributing cause of dyslipidemia)
Cardiovascular Risk Calculation
Metabolic syndrome diagnosis should be integrated into formal ASCVD risk assessment. The Pooled Cohort Equations (PCE), endorsed by the 2019 ACC/AHA guidelines, estimate 10-year risk of fatal or nonfatal MI or stroke. Metabolic syndrome in the absence of T2DM or established ASCVD typically places patients in the intermediate risk category (7.5–20% 10-year ASCVD risk), where coronary artery calcium (CAC) scoring can refine the decision for statin therapy. CAC of zero in a patient with metabolic syndrome substantially downclassifies risk and may support deferring statin therapy; CAC greater than 100 Agatston units indicates high risk warranting immediate treatment. The Reynolds Risk Score incorporates hsCRP, potentially reclassifying a meaningful proportion of intermediate-risk patients.
Ancillary Assessment
- Liver imaging: Hepatic ultrasound, controlled attenuation parameter (CAP) on FibroScan, or MRI-proton density fat fraction (MRI-PDFF) to quantify hepatic steatosis in suspected NAFLD/NASH.
- Sleep study: Polysomnography or home sleep apnea testing for suspected OSA (Epworth Sleepiness Scale, STOP-BANG questionnaire).
- Vascular studies: Carotid intima-media thickness (CIMT) and ankle-brachial index (ABI) in intermediate-risk patients.
- Exclusion of secondary causes: Consider 24-hour urine cortisol or dexamethasone suppression test for Cushing syndrome; aldosterone-to-renin ratio for primary aldosteronism in hypertensive patients; thyroid function tests.
7. Treatment
Management of metabolic syndrome requires a comprehensive, multifactorial approach targeting all components simultaneously. Lifestyle modification is the cornerstone of therapy; pharmacologic interventions address individual components when lifestyle measures are insufficient.
Lifestyle Modification
Weight reduction: A 5–10% reduction in body weight produces clinically meaningful improvements across all metabolic syndrome components. A 7% weight loss goal, as demonstrated in the Diabetes Prevention Program (DPP), reduces T2DM incidence by 58% over 3 years — superior to metformin (31% reduction). Greater weight loss (10–15%) can achieve remission of components in a substantial proportion of patients.
Dietary interventions:
- Mediterranean diet: Rich in olive oil, nuts, fish, vegetables, legumes, and whole grains; reduced in red meat and refined carbohydrates. The PREDIMED trial demonstrated a 30% relative risk reduction in major cardiovascular events. Associated with improvements in all metabolic syndrome components, including blood pressure, lipids, and insulin sensitivity.
- Dietary Approaches to Stop Hypertension (DASH) diet: Emphasizes fruits, vegetables, low-fat dairy, whole grains; reduces sodium to less than 2300 mg/day. Reduces systolic blood pressure by 8–14 mmHg.
- Low-carbohydrate diets: Produce rapid reductions in triglycerides and blood glucose; effects on HDL-C are favorable; sustained adherence and long-term cardiovascular outcomes require further study.
- Caloric restriction: Total caloric reduction of 500–750 kcal/day below estimated energy needs is a reasonable starting point; precise macronutrient composition matters less than overall caloric balance and diet quality for weight loss.
- Specific dietary targets: Limiting added sugar to less than 25 g/day (women) and less than 36 g/day (men); eliminating sugar-sweetened beverages; reducing saturated fat to less than 7% of total calories; increasing dietary fiber to greater than 25–30 g/day.
Physical activity:
- Minimum 150 minutes per week of moderate-intensity aerobic activity (brisk walking, cycling, swimming) or 75 minutes per week of vigorous-intensity activity — consistent with AHA/ACC and WHO guidelines.
- Resistance training 2–3 times per week improves insulin sensitivity, preserves lean body mass during weight loss, and favorably affects lipid profiles independently of aerobic exercise.
- Reducing sedentary time, even without structured exercise, improves postprandial glucose and insulin levels; breaking prolonged sitting with 2-minute movement intervals every 30 minutes is beneficial.
- High-intensity interval training (HIIT) produces equivalent or superior metabolic benefits in less time compared to moderate continuous exercise, with good safety in appropriately selected patients.
Behavioral and psychosocial interventions: Intensive lifestyle intervention programs (16 or more sessions in the first year) combining dietary counseling, physical activity, and behavioral support (consistent with DPP methodology) achieve the greatest weight loss and metabolic improvement. Cognitive behavioral therapy (CBT) addresses emotional eating, food addiction, and adherence barriers.
Pharmacologic Treatment of Individual Components
Dyslipidemia:
- Statins: First-line therapy for elevated LDL-C. High-intensity statins (atorvastatin 40–80 mg, rosuvastatin 20–40 mg) are indicated in patients with established ASCVD or high estimated risk. Moderate-intensity statins for primary prevention in intermediate-risk patients.
- Fibrates (fenofibrate, gemfibrozil): Primary treatment for severe hypertriglyceridemia (greater than 500 mg/dL to reduce pancreatitis risk). In patients with metabolic syndrome and residual hypertriglyceridemia despite statin therapy, icosapentaenoic acid (EPA; icosapent ethyl 4 g/day) demonstrated a 25% relative risk reduction in major cardiovascular events in the REDUCE-IT trial.
- Omega-3 fatty acids (icosapent ethyl): FDA-approved as an adjunct to diet to reduce cardiovascular risk in adults with TG 150 mg/dL or greater and established CVD or diabetes plus additional risk factors; 4 g/day dose.
- Niacin: Raises HDL-C but has not demonstrated cardiovascular benefit in statin-treated patients (AIM-HIGH, HPS2-THRIVE) and is generally not recommended for routine use.
Hypertension:
- ACE inhibitors or angiotensin receptor blockers (ARBs) are preferred as first-line agents in patients with metabolic syndrome, T2DM, microalbuminuria, or left ventricular hypertrophy, due to their neutral or beneficial effect on insulin sensitivity and their renal protective properties.
- Thiazide diuretics and beta-blockers should be used with caution as monotherapy in metabolic syndrome, as high-dose thiazides worsen insulin resistance and dyslipidemia, and conventional beta-blockers reduce peripheral glucose uptake and mask hypoglycemia. Cardioselective beta-blockers (bisoprolol, metoprolol succinate) have less metabolic impact and are appropriate when indicated (heart failure, post-MI).
- Calcium channel blockers are metabolically neutral and are a reasonable option, particularly in combination therapy.
- Target blood pressure: less than 130/80 mmHg per the 2017 ACC/AHA guidelines for most adults with metabolic syndrome.
Impaired fasting glucose / prevention of T2DM:
- Metformin: Reduces T2DM incidence by 31% in DPP; particularly effective in patients with BMI greater than 35 kg/m², age under 60, and women with prior gestational diabetes. Dose: 850–1000 mg twice daily with meals.
- GLP-1 receptor agonists: Semaglutide, liraglutide, and dulaglutide produce substantial weight loss (10–15% with semaglutide 2.4 mg/week), improve all components of metabolic syndrome, and reduce cardiovascular events in established ASCVD (LEADER, SUSTAIN-6, SELECT trials). Increasingly used in obesity management independent of diabetes status.
- SGLT-2 inhibitors: Empagliflozin, canagliflozin, and dapagliflozin lower glucose, blood pressure, body weight, and uric acid; demonstrate cardiovascular and renal protection in trials. Growing role in metabolic syndrome management particularly when heart failure or chronic kidney disease is present.
- Pioglitazone (TZD): Improves insulin sensitivity through PPAR-γ activation; reduces progression to T2DM and may benefit NASH. Limited by weight gain, fluid retention, and fracture risk.
Obesity pharmacotherapy:
- FDA-approved agents for chronic weight management (BMI greater than or equal to 30 or greater than or equal to 27 with comorbidity): orlistat, phentermine/topiramate ER, naltrexone/bupropion SR, liraglutide 3.0 mg/day, semaglutide 2.4 mg/week (Wegovy), tirzepatide 5–15 mg/week (GIP/GLP-1 dual agonist, most effective).
- Tirzepatide achieves mean weight loss of 15–22% — approaching bariatric surgery outcomes.
Bariatric and metabolic surgery:
- Indicated in patients with BMI greater than or equal to 40 kg/m² or greater than or equal to 35 with at least one obesity-related comorbidity (including metabolic syndrome components), after failure of conservative measures.
- Roux-en-Y gastric bypass (RYGB) and sleeve gastrectomy achieve durable weight loss (25–30% and 20–25% respectively at 5 years) with metabolic syndrome resolution in 70–95% of patients.
- T2DM remission occurs in 40–80% of patients, largely through weight-independent mechanisms including altered gut hormone secretion (GLP-1 surge), bile acid metabolism, and microbiome changes.
Non-alcoholic fatty liver disease (NAFLD/NASH) management:
- Weight loss of 7–10% reduces hepatic steatosis; 10% or more can improve fibrosis.
- Vitamin E (800 IU/day) is recommended for biopsy-proven NASH in non-diabetic patients per AASLD guidelines.
- Pioglitazone for biopsy-proven NASH in patients with T2DM or prediabetes.
- GLP-1 agonists and SGLT-2 inhibitors show promising antifibrotic effects in ongoing trials.
8. Complications
Metabolic syndrome multiplies risk for a wide spectrum of serious complications:
Cardiovascular Disease
- Metabolic syndrome confers a 2-fold increased risk of incident CVD events and a 1.5-fold increased risk of CVD mortality compared to matched controls without the syndrome.
- The risk is partly additive to traditional Framingham risk factors; metabolic syndrome reclassifies a meaningful proportion of intermediate-risk patients to higher risk.
- Subclinical atherosclerosis (elevated CIMT, coronary artery calcium) progresses more rapidly. Left ventricular hypertrophy and diastolic dysfunction are common precursors to heart failure with preserved ejection fraction (HFpEF).
- Atrial fibrillation risk is increased 1.5–2-fold, mediated by left atrial enlargement, hypertension, and RAAS activation.
Type 2 Diabetes Mellitus
- Metabolic syndrome increases T2DM risk 5-fold. The majority of patients who develop T2DM have antecedent metabolic syndrome.
- Once diabetes develops, all microvascular and macrovascular complications become additional concerns.
Non-alcoholic Fatty Liver Disease / NASH / Cirrhosis
- NAFLD affects 70–90% of patients with metabolic syndrome. Of these, 20–30% progress to NASH, and 10–20% of NASH cases progress to cirrhosis over 10–20 years.
- Hepatocellular carcinoma (HCC) can arise in NASH-related cirrhosis and, importantly, even in the absence of cirrhosis.
Chronic Kidney Disease
- Metabolic syndrome is associated with a 2.5-fold increased risk of CKD, mediated by hypertension, hyperglycemia, hyperuricemia, and RAAS activation.
Obstructive Sleep Apnea
- Bidirectional relationship: OSA worsens insulin resistance and all metabolic syndrome components; metabolic syndrome (particularly visceral obesity) promotes OSA through fat deposition in pharyngeal tissues.
Malignancy
- Metabolic syndrome is associated with increased risk of colorectal, endometrial, breast (postmenopausal), pancreatic, renal cell, and esophageal (adenocarcinoma) cancers — mediated by hyperinsulinemia, IGF-1 signaling, adipokine dysregulation, and chronic inflammation.
Polycystic Ovary Syndrome
- PCOS and metabolic syndrome share insulin resistance as a central pathophysiologic driver; co-occurrence substantially amplifies reproductive and cardiometabolic risk.
Cognitive Decline and Dementia
- Metabolic syndrome increases risk of vascular dementia and Alzheimer disease; the term "type 3 diabetes" has been applied to cerebral insulin resistance as a putative mechanism in AD.
Gout
- Hyperuricemia, present in the majority of patients with metabolic syndrome, increases gout risk. Fructose-driven uric acid production and reduced renal urate excretion (hyperinsulinemia-mediated) are the main mechanisms.
9. Prognosis
The prognosis of metabolic syndrome is determined by the number and severity of components present, the degree of target organ damage, comorbid conditions, and the effectiveness of intervention. Key prognostic considerations include:
- Cardiovascular mortality: A meta-analysis of over 950,000 patients (Mottillo et al., JACC 2010) demonstrated a 2-fold increased risk of all-cause mortality, cardiovascular disease, MI, and stroke, and a 3.5-fold increased risk of T2DM.
- Reversibility: Metabolic syndrome is not a fixed state. Sustained lifestyle intervention achieving 5–10% weight loss can resolve metabolic syndrome in 30–50% of patients within 1–3 years. Bariatric surgery achieves resolution in 70–95%.
- Component count: Risk increases with the number of criteria fulfilled. Patients with all 5 components have substantially higher cardiovascular risk than those meeting just 3.
- Temporal dynamics: Metabolic syndrome components can appear, disappear, and reappear over time. Longitudinal studies demonstrate that patients who develop metabolic syndrome in midlife carry a lifelong increased cardiovascular risk burden even if criteria are no longer met later.
- Liver disease progression: Once NASH-related cirrhosis or HCC develops, prognosis is significantly worsened; early NAFLD is reversible with intervention.
10. Prevention
Prevention operates at primary (before metabolic syndrome develops), secondary (preventing complications once the syndrome is present), and tertiary (preventing further morbidity in established disease) levels.
Primary Prevention
- Maintaining a healthy body weight throughout life, particularly preventing visceral adiposity accumulation.
- Regular aerobic and resistance exercise: a minimum of 150 minutes of moderate activity per week throughout adulthood.
- Adherence to a predominantly plant-based, minimally processed diet (Mediterranean, DASH, or Nordic dietary patterns).
- Limiting sugar-sweetened beverages and ultra-processed food consumption.
- Adequate sleep (7–9 hours/night) and management of chronic stress.
- Avoidance of tobacco and limitation of alcohol (no more than 1 drink/day for women, 2 for men).
- Minimizing unnecessary antibiotic use to preserve microbiome diversity.
- Reducing environmental toxin exposures (BPA-free containers, filtered water, reduced plastic use).
Population-Level Strategies
- Sugar-sweetened beverage taxes (associated with 10–20% reductions in consumption in implemented jurisdictions).
- Mandatory front-of-package nutritional labeling.
- Built environment modifications to promote active transportation (walking/cycling infrastructure).
- School-based obesity prevention programs targeting diet and physical activity from early childhood.
- Workplace wellness programs addressing sedentary occupational behavior.
Secondary Prevention
- Intensive structured lifestyle programs (DPP model) for patients with metabolic syndrome to prevent T2DM and CVD.
- Routine surveillance: annual fasting glucose/HbA1c, lipid panel, blood pressure monitoring, and hepatic transaminases.
- Pharmacologic intervention initiated promptly when lifestyle measures fail to achieve targets for individual components.
- Referral to a registered dietitian, certified diabetes educator, and behavioral health specialist as part of multidisciplinary care.
11. Recent Research
Research in metabolic syndrome is advancing rapidly across multiple fronts:
- Tirzepatide (GIP/GLP-1 dual agonist): The SURMOUNT-1 and SURMOUNT-2 trials demonstrated unprecedented 15–22% mean body weight reduction with tirzepatide 10–15 mg/week, with resolution of metabolic syndrome components in the majority of treated patients. The SURPASS cardiovascular outcomes trial is ongoing.
- Gut microbiome therapeutics: Fecal microbiota transplantation (FMT) from lean donors transiently improved insulin sensitivity in metabolic syndrome patients (Vrieze et al., Gastroenterology). Engineered next-generation probiotics targeting butyrate production, bile acid metabolism, and LPS reduction are in early clinical trials.
- Ketone bodies and metabolic reprogramming: Exogenous ketone supplementation and ketogenic dietary interventions demonstrate rapid improvements in triglycerides, glucose, and inflammation in metabolic syndrome; mechanisms include NLRP3 inflammasome inhibition by beta-hydroxybutyrate.
- Chronobiology and time-restricted eating (TRE): Studies demonstrate that aligning eating to earlier daylight hours (e.g., 8-hour eating window from 7 AM to 3 PM) improves insulin sensitivity, blood pressure, and oxidative stress independently of caloric restriction, by harmonizing peripheral circadian clocks with the central circadian pacemaker.
- NASH pharmacotherapy: Resmetirom (THR-β agonist), lanifibranor (pan-PPAR agonist), and semaglutide demonstrate NASH resolution and fibrosis regression in Phase 3 trials. Resmetirom received FDA approval in 2024 — the first approved therapy for NASH with fibrosis.
- Polygenic risk scores: Multi-ancestry GWAS data are enabling polygenic risk scores for metabolic syndrome components that, combined with clinical risk factors, may substantially improve early identification of high-risk individuals who would benefit from intensified prevention.
- Adipose tissue browning: Cold exposure, beta-3 adrenergic receptor agonism (mirabegron), and FGF21 analogs promote beige/brown adipocyte thermogenesis, increasing energy expenditure independent of behavior. Clinical trials are investigating these pathways as metabolic syndrome therapeutics.
- Epigenetics and intergenerational risk: Maternal and paternal obesity and metabolic syndrome program epigenetic changes (DNA methylation, histone modification) in offspring that increase their susceptibility to metabolic dysregulation — highlighting the importance of preconception and prenatal metabolic health.
12. References
- Grundy SM, Cleeman JI, Daniels SR, et al. Diagnosis and management of the metabolic syndrome: an American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation. 2005;112(17):2735-2752.
- Alberti KG, Eckel RH, Grundy SM, et al. Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention. Circulation. 2009;120(16):1640-1645.
- Mottillo S, Filion KB, Genest J, et al. The metabolic syndrome and cardiovascular risk: a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56(14):1113-1132.
- Knowler WC, Barrett-Connor E, Fowler SE, et al. Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin. N Engl J Med. 2002;346(6):393-403.
- Estruch R, Ros E, Salas-Salvadó J, et al. Primary prevention of cardiovascular disease with a Mediterranean diet supplemented with extra-virgin olive oil or nuts. N Engl J Med. 2018;378(25):e34.
- Bhatt DL, Steg PG, Miller M, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380(1):11-22.
- Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205-216.
- Loomba R, Hartman ML, Lawitz EJ, et al. Tirzepatide for metabolic dysfunction-associated steatohepatitis with liver fibrosis. N Engl J Med. 2024;391(4):296-308.
- Zinman B, Wanner C, Lachin JM, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373(22):2117-2128.
- Marso SP, Daniels GH, Brown-Frandsen K, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375(4):311-322.
- Lim EL, Hollingsworth KG, Aribisala BS, et al. Reversal of type 2 diabetes: normalisation of beta cell function in association with decreased pancreas and liver triacylglycerol. Diabetologia. 2011;54(10):2506-2514.
- Harrison SA, Bedossa P, Guy CD, et al. A Phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis. N Engl J Med. 2024;390(6):497-509.
- Ley RE, Turnbaugh PJ, Klein S, Gordon JI. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444(7122):1022-1023.
- Sutton EF, Beyl R, Early KS, et al. Early time-restricted feeding improves insulin sensitivity, blood pressure, and oxidative stress even without weight loss in men with prediabetes. Cell Metab. 2018;27(6):1212-1221.
- Garvey WT, Mechanick JI, Brett EM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology comprehensive clinical practice guidelines for medical care of patients with obesity. Endocr Pract. 2016;22(Suppl 3):1-203.
- Virani SS, Newby LK, Arnold SV, et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease. J Am Coll Cardiol. 2023;82(9):833-955.
- Ryan DH, Yockey SR. Weight loss and improvement in comorbidity: differences at 5%, 10%, 15%, and over. Curr Obes Rep. 2017;6(2):187-194.
- Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA guideline for the prevention, detection, evaluation, and management of high blood pressure in adults. J Am Coll Cardiol. 2018;71(19):e127-e248.
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — metabolic syndrome
- PubMed — metabolic syndrome criteria
- PubMed — abdominal obesity metabolic syndrome
- PubMed — dyslipidemia metabolic syndrome
- PubMed — insulin resistance metabolic syndrome
- PubMed — metabolic syndrome cardiovascular risk
- PubMed — Mediterranean diet metabolic syndrome
- PubMed — exercise metabolic syndrome
- PubMed — NAFLD metabolic syndrome
- PubMed — hypertension metabolic syndrome
- PubMed — metabolic syndrome children
- PubMed — metabolic syndrome pharmacotherapy
Connections
- Insulin Resistance
- Diabetes
- Obesity
- Hypertension
- Cardiovascular Disease
- Non-Alcoholic Fatty Liver Disease
- Magnesium
- Chromium
- Vitamin D3
- Fasting
- Polycystic Ovary Syndrome
- Metabolic Syndrome Cardiovascular Risk
- Uric Acid
- Fasting Insulin and HOMA-IR Testing
- Fasting Insulin
- Obstructive Sleep Apnea
- Visceral Fat
- Ultra-Processed Foods
Featured Videos

Metabolic Syndrome Solution (Cause & Cure of Syndrome X) 2026

Dr. Paul Mason - 'Treating Metabolic Syndrome'

What Is Metabolic Syndrome? How To Check For It.

Your Doctor is Wrong! How to CURE METABOLIC SYNDROME - 2026

Reversing Metabolic Disease

Obesity and Metabolic Syndrome | Clinical Medicine

Move From Metabolic Syndrome to Metabolic Health - 4 Things That Make a Difference

What is Metabolic Syndrome, and Can You Cure It? 2026

Metabolic Syndrome (Obesity; High BP, High Blood Sugar; High Triglycerides & Low Good Cholesterol)