Nephrotic Syndrome
Nephrotic Syndrome is a clinical syndrome defined by massive proteinuria (>3.5 g/day in adults or >40 mg/m²/hour in children), hypoalbuminemia (<3.5 g/dL), generalized edema, and hyperlipidemia. It results from disruption of the glomerular filtration barrier, particularly the podocyte architecture, leading to unrestricted protein loss in the urine. It is one of the most common presentations of glomerular disease and requires systematic evaluation to identify its underlying cause.
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
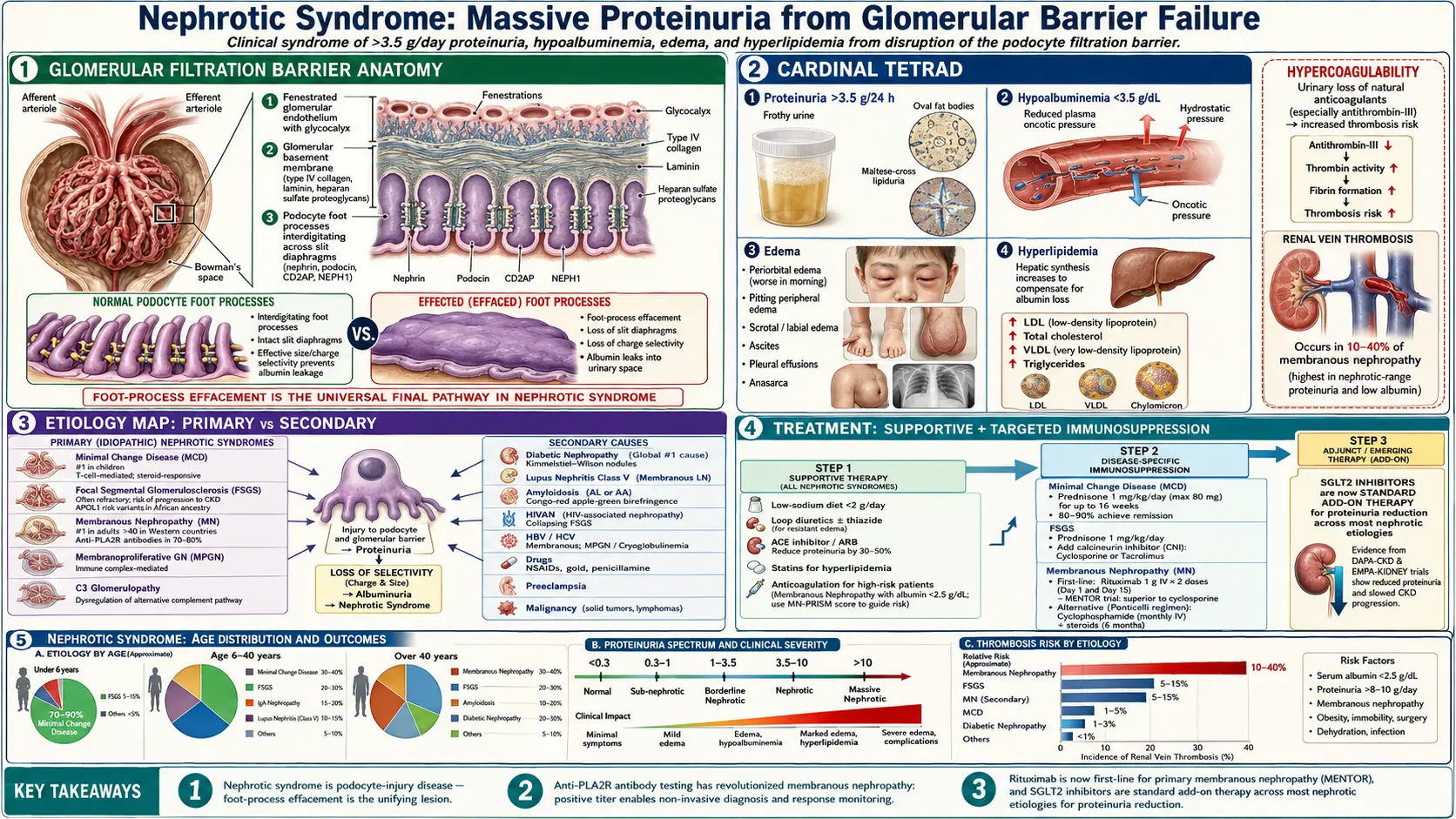
Nephrotic syndrome represents a final common pathway of diverse glomerular diseases that compromise the integrity of the glomerular filtration barrier. The glomerular filtration barrier consists of three components: the fenestrated glomerular endothelium, the glomerular basement membrane (GBM), and the visceral epithelial cells (podocytes) with their interdigitating foot processes connected by the slit diaphragm. Injury to any of these components — but particularly the podocyte — leads to proteinuria. The four cardinal features are:
- Proteinuria: >3.5 g/24h (adults); spot urine protein-to-creatinine ratio >3.5 g/g
- Hypoalbuminemia: Serum albumin <3.5 g/dL (often <2.5 g/dL in severe cases)
- Edema: Peripheral, periorbital, ascites, pleural effusions
- Hyperlipidemia: Elevated LDL cholesterol, total cholesterol, VLDL, and triglycerides
Lipiduria and a hypercoagulable state are also characteristic findings. Nephrotic syndrome may be primary (idiopathic glomerular disease) or secondary to systemic disorders.
2. Epidemiology
In children, nephrotic syndrome has an annual incidence of approximately 2–7 per 100,000 children, with minimal change disease (MCD) accounting for 70–90% of cases under age 6. In adults, the annual incidence is approximately 3 per 100,000, with membranous nephropathy (MN) being the most common cause in patients over 40 in Western countries (30–40% of adult nephrotic biopsies), and focal segmental glomerulosclerosis (FSGS) being more prevalent in younger adults and particularly in individuals of African ancestry.
Diabetic nephropathy is the most common secondary cause of nephrotic-range proteinuria globally, accounting for approximately one-third of all ESKD in developed nations. Secondary causes collectively account for 30–40% of adult nephrotic syndrome. The condition has a slight male predominance in adults; in children, the male-to-female ratio is approximately 2:1.
Geographic variation exists: MCD is more prevalent in Asian children; FSGS rates have increased in the United States, particularly in Black Americans due to APOL1 genetic risk variants.
3. Pathophysiology
Glomerular Filtration Barrier Disruption
The glomerular filtration barrier normally restricts proteins >60–70 kDa based on size and charge selectivity. The podocyte slit diaphragm, composed of nephrin, podocin, CD2AP, and NEPH1 proteins, provides the primary structural and signaling scaffold limiting protein filtration. Injury to podocyte foot processes (effacement) is the universal final pathway in nephrotic syndrome, regardless of etiology.
Proteinuria and Hypoalbuminemia
Massive albuminuria (predominantly albumin due to its high concentration and negative charge) exceeds hepatic synthetic capacity, resulting in hypoalbuminemia. Hepatic albumin synthesis increases to 14–24 g/day (normal 12–14 g/day) but is insufficient to compensate for urinary losses of 3.5–40+ g/day. Hypoalbuminemia reduces plasma oncotic pressure, promoting Starling force-driven fluid shift from the intravascular to interstitial compartment, causing edema.
Edema Formation — Two Theories
- Underfill hypothesis: Hypoalbuminemia → reduced plasma oncotic pressure → capillary fluid leak into interstitium → effective arterial blood volume depletion → RAAS and ADH activation → sodium and water retention. Applies predominantly to MCD.
- Overfill hypothesis: Primary renal sodium retention (increased ENaC expression in collecting duct, reduced responsiveness to atrial natriuretic peptide) → expanded blood volume → secondary redistribution into interstitium. Applies to most adult primary and secondary GN.
Hyperlipidemia
Reduced oncotic pressure stimulates hepatic lipoprotein synthesis (increased apoB-100, VLDL, LDL production). Simultaneously, reduced LPL activity (urinary loss of lipoprotein lipase activators) and decreased LCAT activity impair lipoprotein clearance. The result is mixed hyperlipidemia with elevated total cholesterol, LDL, VLDL, and triglycerides.
Hypercoagulability
A prothrombotic state results from: urinary loss of anticoagulant proteins (antithrombin III, protein C, protein S, plasminogen); elevated procoagulant factors (fibrinogen, factors V, VIII, von Willebrand factor); platelet hyperactivation; and hyperviscosity from hyperlipidemia. This is most clinically significant in membranous nephropathy (renal vein thrombosis in 10–40%).
4. Etiology and Risk Factors
Primary (Idiopathic) Glomerular Disease
- Minimal Change Disease (MCD): Most common in children; associated with T-cell dysfunction, lymphokine release; secondary causes include NSAIDs, lithium, Hodgkin lymphoma
- Focal Segmental Glomerulosclerosis (FSGS): Primary (circulating permeability factors — suPAR, CLCF1); secondary (hyperfiltration from obesity, solitary kidney, reflux nephropathy; HIV-associated nephropathy [HIVAN]; heroin nephropathy; anabolic steroids); genetic (NPHS1, NPHS2/podocin, WT1, INF2, TRPC6 mutations)
- Membranous Nephropathy (MN): Primary (anti-PLA2R antibodies 70–80%, anti-THSD7A 3–5%); secondary (solid organ malignancy, HBV, drugs including NSAIDs, penicillamine, gold, SLE)
- MPGN/C3 Glomerulopathy: Immune complex or complement dysregulation-mediated
Secondary Causes
- Diabetic nephropathy: Most common secondary cause globally; Kimmelstiel-Wilson nodules on biopsy
- Lupus nephritis: Class V (membranous LN) presents as pure nephrotic syndrome
- Amyloidosis: AL (light chain) amyloid or AA (reactive) amyloid; Congo red staining with apple-green birefringence; serum/urine electrophoresis with free light chain assay
- Monoclonal Immunoglobulin Deposition Disease (MIDD): Light chain deposition disease (LCDD)
- Preeclampsia: Nephrotic-range proteinuria with hypertension in pregnancy
- Infections: HIV (HIVAN — collapsing FSGS), HBV (membranous), HCV (MPGN/cryoglobulinemia), malaria (quartan malaria nephropathy — membranoproliferative pattern), syphilis, schistosomiasis
- Allergic reactions: Bee stings, poison ivy (MCD pattern)
- Medications: NSAIDs, gold, penicillamine, captopril, mercury, heroin, pamidronate (collapsing FSGS)
Risk Factors
- APOL1 G1/G2 risk alleles (African ancestry): 7-fold increased FSGS risk
- HLA-DQA1 and HLA-DQB1 variants associated with MN susceptibility
- Obesity, hypertension, diabetes mellitus, pregnancy
- Malignancy (particularly solid tumors with MN, hematological malignancies with MCD)
- Autoimmune diseases, chronic infections
5. Clinical Presentation
Cardinal Features
- Edema: Initially periorbital (characteristically worse in the morning), then progressing to peripheral pitting edema of legs and ankles, scrotal/vulvar edema, ascites, and pleural effusions; anasarca in severe cases
- Foamy urine: Protein foam that persists in the toilet bowl due to surface tension effect of albumin
- Weight gain: Fluid retention may cause rapid weight gain of 10–20 kg
- Fatigue, malaise: Due to hypoalbuminemia, anemia, and metabolic derangements
Examination Findings
- Pitting edema (bilateral, dependent), periorbital puffiness
- Ascites (shifting dullness, fluid wave), pleural dullness to percussion
- Scrotal or labial edema
- Leukonychia (white nails) due to hypoalbuminemia; Muehrcke's lines
- Xanthomas, xanthelasma in severe hyperlipidemia
- Signs of underlying cause: diabetic retinopathy, butterfly rash (SLE), organomegaly (amyloid, malignancy)
Age-Related Patterns
- Children <6 years: MCD predominantly (steroid-responsive nephrotic syndrome)
- Adolescents/young adults: MCD, FSGS, IgA nephropathy
- Adults 20–40 years: FSGS, MCD, lupus nephritis, IgA nephropathy
- Adults >40 years: Membranous nephropathy, FSGS, amyloidosis, diabetic nephropathy
6. Diagnosis
Urinalysis
- Dipstick: 3+ to 4+ proteinuria (≥300 mg/dL)
- Microscopy: oval fat bodies (free lipid droplets from tubular reabsorption of filtered lipids), fatty casts, Maltese cross pattern under polarized light microscopy; few or no RBCs (distinguishes from nephritic syndrome)
- Spot urine protein-to-creatinine ratio (PCR): >3.5 g/g equivalent to >3.5 g/24h proteinuria
- 24-hour urine protein collection: Gold standard quantification; assess completeness using urine creatinine (expected 20–25 mg/kg/day)
- Urine albumin-to-creatinine ratio (ACR): >300 mg/g indicates nephrotic-range albuminuria
- Urine electrophoresis: selective (albumin-predominant) vs. non-selective proteinuria; selectivity index (ratio of IgG:transferrin clearance) <0.1 suggests selective proteinuria (MCD)
Blood Tests
- Serum albumin: <3.5 g/dL; severity correlates with edema and thrombotic risk
- Lipid panel: Elevated total cholesterol (>300 mg/dL), LDL, triglycerides; HDL often normal or reduced
- Serum creatinine and eGFR: Often normal in MCD; may be reduced in FSGS, MN, secondary causes
- CBC: Microcytic anemia (iron deficiency from transferrin loss), normocytic anemia
- Coagulation studies: Elevated fibrinogen, factor VIII; reduced antithrombin III (<70% in membranous nephropathy at high thrombotic risk)
- Immunological workup: ANA/anti-dsDNA/complement (SLE); anti-PLA2R/anti-THSD7A (membranous); SPEP/UPEP/free light chains (amyloid, MIDD, myeloma); hepatitis B/C serology; HIV; cryoglobulins
- Glucose, HbA1c: Exclude diabetic nephropathy
- Thyroid function: Hypothyroidism can cause nephrotic-range proteinuria; thyroid-binding globulin lost in urine
Kidney Biopsy
Indicated in adults with nephrotic syndrome to establish diagnosis, guide therapy, and determine prognosis. Generally not performed in children aged 1–8 years with typical steroid-responsive nephrotic syndrome (presumptive MCD). Biopsy guides distinction between MCD (corticosteroid-responsive), FSGS (variable steroid response, often requires additional agents), MN (anti-PLA2R-guided therapy), and secondary causes requiring treatment of the underlying disease.
Pathological findings: MCD shows normal or near-normal light microscopy with diffuse podocyte foot process effacement on EM; FSGS shows segmental scarring; MN shows subepithelial deposits (spike-and-dome pattern on silver stain); amyloid shows amorphous eosinophilic material (Congo red positive).
Imaging
- Renal ultrasound: normal or enlarged kidneys in early nephrotic syndrome; assess for renal vein thrombosis (Doppler)
- CT abdomen/pelvis (with contrast if renal function permits): evaluate for malignancy as secondary cause in adults >40 years
- Age-appropriate cancer screening: mammography, colonoscopy, PSA, tumor markers as clinically indicated
7. Treatment
General Supportive Measures
- Dietary sodium restriction: <2 g/day (88 mmol/day) to reduce fluid retention; avoid excessive fluid restriction
- Dietary protein: Normal protein intake (0.8–1.0 g/kg/day); high-protein diet does not correct hypoalbuminemia and may worsen proteinuria
- Diuretics: Loop diuretics (furosemide 40–120 mg twice daily, or continuous IV infusion); may require high doses due to reduced tubular secretion from hypoalbuminemia and tubular binding by albumin; combined loop + thiazide diuretic (metolazone) for resistant edema; IV albumin infusion before furosemide in severe hypoalbuminemia (<2 g/dL) may improve diuretic response
- RAAS blockade: ACE inhibitors or ARBs reduce proteinuria by 30–50% via efferent arteriolar dilation and direct podocyte cytoprotection; use cautiously with hypoalbuminemia and AKI risk
- Lipid management: Statins (HMG-CoA reductase inhibitors) for hyperlipidemia; ezetimibe as adjunct; goal LDL <100 mg/dL (<70 mg/dL if cardiovascular risk factors present)
- Infection prophylaxis: Prophylactic penicillin in children with severe hypoalbuminemia (<1.5 g/dL)
Anticoagulation
Indicated for documented thrombosis (DVT, PE, renal vein thrombosis). Prophylactic anticoagulation with warfarin (target INR 2–3) or LMWH is recommended in membranous nephropathy with serum albumin <2.5 g/dL when bleeding risk is acceptable (MN-PRISM score >6%). Prophylactic anticoagulation is controversial in other nephrotic causes; individualize based on thrombotic risk assessment.
Disease-Specific Immunosuppressive Therapy
- MCD: Prednisone 1 mg/kg/day (max 80 mg) for 4–16 weeks until remission; 80–90% achieve complete remission within 8 weeks; frequent relapses treated with repeat corticosteroids; steroid-dependent or frequently relapsing disease — cyclosporine, tacrolimus, mycophenolate mofetil, or rituximab (MENTOR-like approach)
- FSGS (primary): Prednisone 1 mg/kg/day for up to 16 weeks; complete remission in 25–40%; calcineurin inhibitors (cyclosporine or tacrolimus) for steroid-resistant FSGS; mycophenolate + corticosteroids as alternative; rituximab with limited evidence; sparsentan under investigation
- Membranous nephropathy: Anti-PLA2R-positive with moderate-to-high risk — rituximab 1 g IV × 2 doses (MENTOR trial, superior to cyclosporine at 24 months); alternatively cyclophosphamide + alternating oral methylprednisolone (Ponticelli regimen) for 6 months; obinutuzumab under investigation
- Secondary causes: Treatment of underlying condition (antidiabetic therapy, lupus immunosuppression, antiviral therapy for HBV/HCV, malignancy treatment, drug discontinuation)
SGLT2 Inhibitors
Dapagliflozin and empagliflozin significantly reduce proteinuria and slow eGFR decline in patients with CKD and nephrotic-range proteinuria, independent of diabetes status. Now recommended as standard of care for CKD with proteinuria eGFR >20 mL/min/1.73 m² (DAPA-CKD, EMPA-KIDNEY trials).
8. Complications
- Thromboembolic disease: Deep vein thrombosis, pulmonary embolism, renal vein thrombosis (particularly membranous nephropathy); arterial thrombosis is less common but recognized
- Infections: Increased susceptibility to encapsulated organisms (Streptococcus pneumoniae, Haemophilus influenzae); peritonitis (spontaneous bacterial peritonitis with ascites); cellulitis in edematous limbs; impaired opsonization due to loss of immunoglobulins and complement
- Acute kidney injury: Due to severe intravascular volume depletion (prerenal); renal vein thrombosis; hypovolemia-induced ATN; NSAIDs and diuretic overuse
- Cardiovascular disease: Accelerated atherosclerosis from hyperlipidemia, hypertension, chronic kidney disease; increased risk of MI, stroke
- Endocrine disturbances: Hypothyroidism from loss of thyroid-binding globulin; vitamin D deficiency from loss of vitamin D-binding protein; anemia from loss of transferrin and EPO
- Drug toxicity: Altered drug pharmacokinetics (reduced protein binding of warfarin, phenytoin, furosemide; diuretic resistance)
- Malnutrition: Protein-calorie malnutrition from sustained protein losses; muscle wasting
- CKD progression: To ESKD depending on underlying cause and treatment response
9. Prognosis
Prognosis in nephrotic syndrome depends critically on the underlying etiology and treatment response. Complete remission of proteinuria (<0.3 g/day) is associated with preservation of kidney function; partial remission (<3.5 g/day with >50% reduction) also confers significant renal protection.
- MCD in children: Excellent prognosis; 95% achieve complete remission with corticosteroids; 40–50% relapse but rarely progress to CKD; adult MCD has similar remission rates but higher relapse frequency
- FSGS: 50–70% of untreated patients progress to ESKD within 10 years; steroid-resistant FSGS has the worst prognosis; complete remission dramatically improves renal survival
- Membranous nephropathy: "Rule of thirds" — one-third spontaneous complete remission within 5 years; one-third partial remission; one-third progressive decline to ESKD; anti-PLA2R titer predicts likelihood of spontaneous remission (low titer favors remission)
- Diabetic nephropathy: Once nephrotic-range proteinuria is established, median time to ESKD is 6–10 years without aggressive management; RAAS blockade + SGLT2 inhibitors slow progression
- Amyloidosis: Highly variable; AL amyloid prognosis improved dramatically with anti-plasma cell therapy (bortezomib-based regimens, daratumumab)
10. Prevention
- Early treatment of conditions predisposing to secondary nephrotic syndrome: tight glycemic control in diabetes (HbA1c <7%), treatment of HBV/HCV, SLE management
- Avoidance of nephrotoxic medications (NSAIDs, gold, penicillamine) in susceptible patients
- RAAS blockade and blood pressure control for patients with proteinuria to slow progression
- SGLT2 inhibitors in patients with CKD and proteinuria to reduce proteinuria and preserve GFR
- Age-appropriate cancer surveillance to detect secondary MN early
- Prophylactic anticoagulation in high-risk membranous nephropathy
11. Recent Research and Advances
- Rituximab in MCD and steroid-dependent nephrotic syndrome: Multiple randomized trials (NEMO, RITUX-AREN) confirm rituximab superiority over corticosteroids for relapse prevention and reducing cumulative steroid exposure in children and adults
- Obinutuzumab in MN: More potent anti-CD20 depletion than rituximab; phase II/III trials ongoing for primary membranous nephropathy with promising early efficacy data
- Sparsentan for FSGS: Dual ETA receptor/angiotensin receptor antagonist; DUPLEX trial showed significant proteinuria reduction; FDA accelerated approval sought
- Inaxaplin (VX-147): APOL1 inhibitor specifically targeting APOL1 G1/G2-mediated podocyte injury; phase 2/3 trials (ZENITH trial) in APOL1-associated FSGS showed marked proteinuria reduction — potential first precision therapy for APOL1 nephropathy
- SGLT2 inhibitors: Now established as standard-of-care therapy for proteinuric CKD independent of diabetes (DAPA-CKD 2020, EMPA-KIDNEY 2022); mechanisms include hemodynamic (tubuloglomerular feedback), anti-fibrotic, and anti-inflammatory effects
- Circulating permeability factors in FSGS: Soluble urokinase plasminogen activator receptor (suPAR) and cardiotrophin-like cytokine factor 1 (CLCF1) implicated in primary FSGS recurrence after transplantation; therapeutic targeting underway
- Anti-nephrin antibodies in MCD: 2023 discovery of anti-nephrin IgG4 antibodies in a subset of MCD patients represents a breakthrough, suggesting MCD is at least partly an autoimmune nephropathy analogous to anti-PLA2R MN
12. References
- KDIGO 2021 Clinical Practice Guideline for the Management of Glomerular Diseases. Kidney International. 2021;100(4S):S1–S276.
- Fervenza FC, Appel GB, Barbour SJ, et al. Rituximab or cyclosporine in the treatment of membranous nephropathy (MENTOR). New England Journal of Medicine. 2019;381(1):36–46.
- Heerspink HJL, Stefansson BV, Correa-Rotter R, et al. Dapagliflozin in patients with chronic kidney disease (DAPA-CKD). New England Journal of Medicine. 2020;383(15):1436–1446.
- The EMPA-KIDNEY Collaborative Group. Empagliflozin in patients with chronic kidney disease. New England Journal of Medicine. 2023;388(2):117–127.
- Trachtman H, Nelson P, Adler S, et al. DUPLEX: a randomized controlled trial with sparsentan versus irbesartan in patients with primary FSGS. Kidney International Reports. 2022;7(9):2008–2020.
- Sethi S, Fervenza FC, Zhang Y, et al. C3 glomerulonephritis: clinicopathological findings, complement abnormalities, glomerular proteomic profile, treatment, and follow-up. Kidney International. 2012;82(4):465–473.
- Kidney Disease: Improving Global Outcomes (KDIGO). KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International. 2024;105(4S):S117–S314.
- Doublier S, Salvidio G, Lupia E, et al. Nephrin expression is reduced in human diabetic nephropathy: evidence for a distinct role for glycated albumin and angiotensin II. Diabetes. 2003;52(4):1023–1030.
- Kronbichler A, Gauckler P, Windpessl M, et al. Rituximab for immune-mediated glomerular disease: therapeutic considerations. Nature Reviews Nephrology. 2021;17(7):435–450.
- Kopp JB, Nelson GW, Sampath K, et al. APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. Journal of the American Society of Nephrology. 2011;22(11):2129–2137.
- Ronco P, Beck L, Debiec H, et al. Podocyte-specific loss of functional microRNAs leads to rapid glomerular and tubular injury. Journal of the American Society of Nephrology. 2011;22(7):1240–1252.
- Tomas NM, Beck LH Jr, Meyer-Schwesinger C, et al. Thrombospondin type-1 domain-containing 7A in idiopathic membranous nephropathy. New England Journal of Medicine. 2014;371(24):2277–2287.
- Sinha A, Bagga A, NHS Working Group. Consensus guidelines for management of idiopathic nephrotic syndrome. Indian Pediatrics. 2021;58(1):49–69.
- Guerry MJJ, Brenchley PNC. Mechanisms of proteinuria in nephrotic syndrome. Clinical Journal of the American Society of Nephrology. 2023.
- Watts AJB, Keller KH, Lerner G, et al. Discovery of autoantibodies targeting nephrin in minimal change disease supports a novel autoimmune etiology. Journal of the American Society of Nephrology. 2022;33(1):238–252.
Research Papers
Explore current literature on nephrotic syndrome via PubMed topic searches. These links open live PubMed searches for the listed keywords — results update as new studies are indexed.
- Clinical trials on nephrotic syndrome — PubMed search
- Clinical trials on minimal change disease — PubMed search
- Clinical trials on focal segmental glomerulosclerosis FSGS — PubMed search
- Clinical trials on membranous nephropathy — PubMed search
- Clinical trials on nephrotic syndrome children — PubMed search
- Clinical trials on rituximab nephrotic syndrome — PubMed search
- Clinical trials on proteinuria treatment — PubMed search
Connections
- Kidney Disease
- Glomerulonephritis
- Acute Kidney Injury
- Diabetes
- Lupus
- Hypertension
- Deep Vein Thrombosis
- Edema
- Creatinine
- eGFR
- Kidney Function Tests
- Anemia
- Lipid Panel
- Lymphoma
- Cardiovascular Disease
- Hepatitis B
- Urinalysis
- Pleural Effusion
Featured Videos

Nephrotic Syndrome: Alterations in Health - Pediatric Nursing | @LevelUpRN

Nephrotic Syndrome Nursing NCLEX Lecture on Pathophysiology, Treatment in Children (Pediatric)

What is Nephrotic Syndrome? Top Symptoms, Treatment, and Causes You SHOULD Know!
Nephrotic Syndrome - Overview (Sign and symptoms, pathophysiology)

Nephrotic vs. Nephritic syndrome | Symptoms, diagnosis, causes, treatment | Visual explanation

Nephrotic Syndrome: Complications and Treatment– Nephrology | Lecturio

Nephrotic Syndrome vs Nephritic Syndrome Explained (Nephritic vs Nephrotic)

Nephrotic Syndrome | Clinical Presentation, Causes and Treatment
Suni Lee's Kidney Disease and 5 Things To Know About Nephrotic Syndrome