Atrial Fibrillation
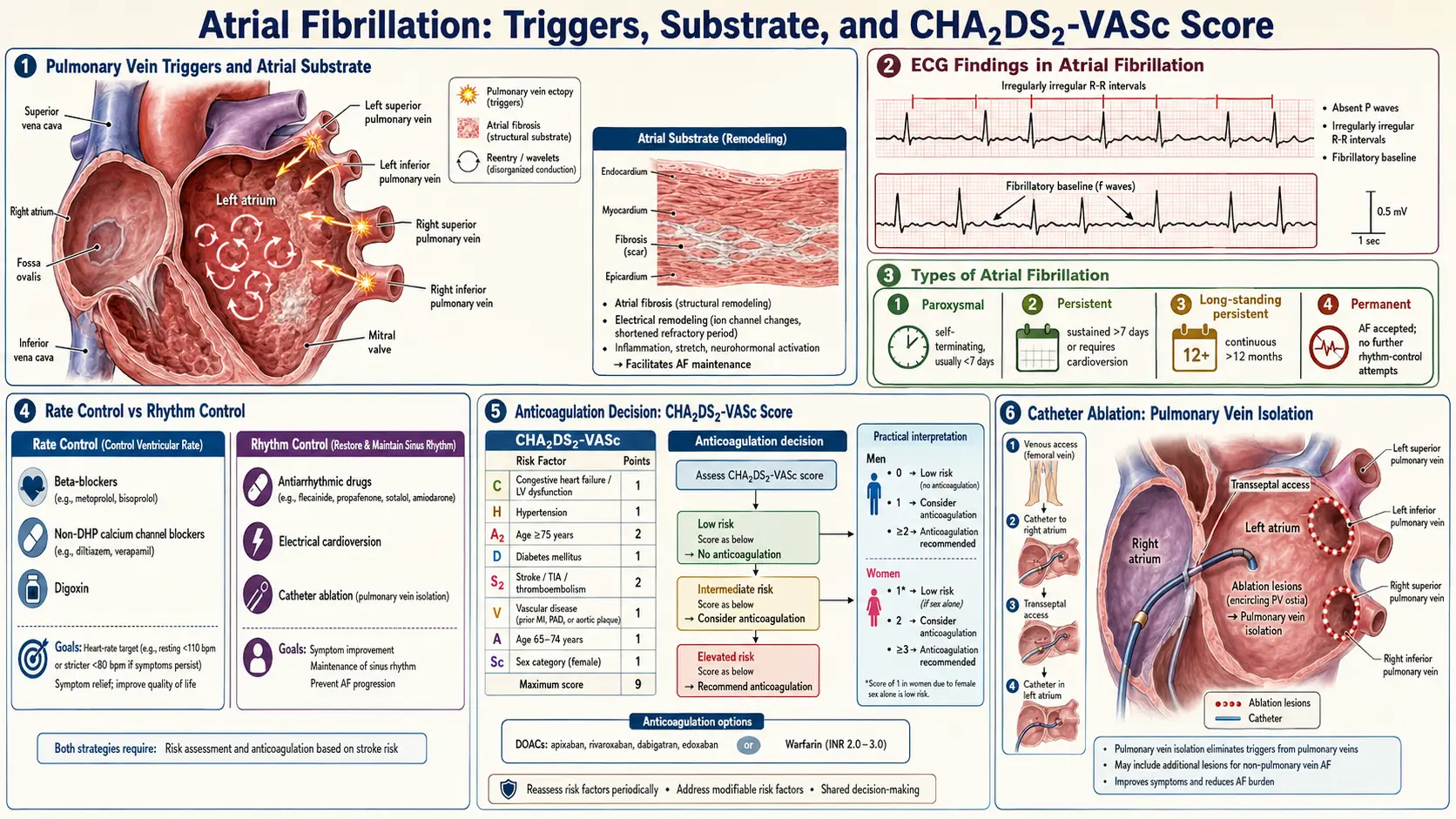
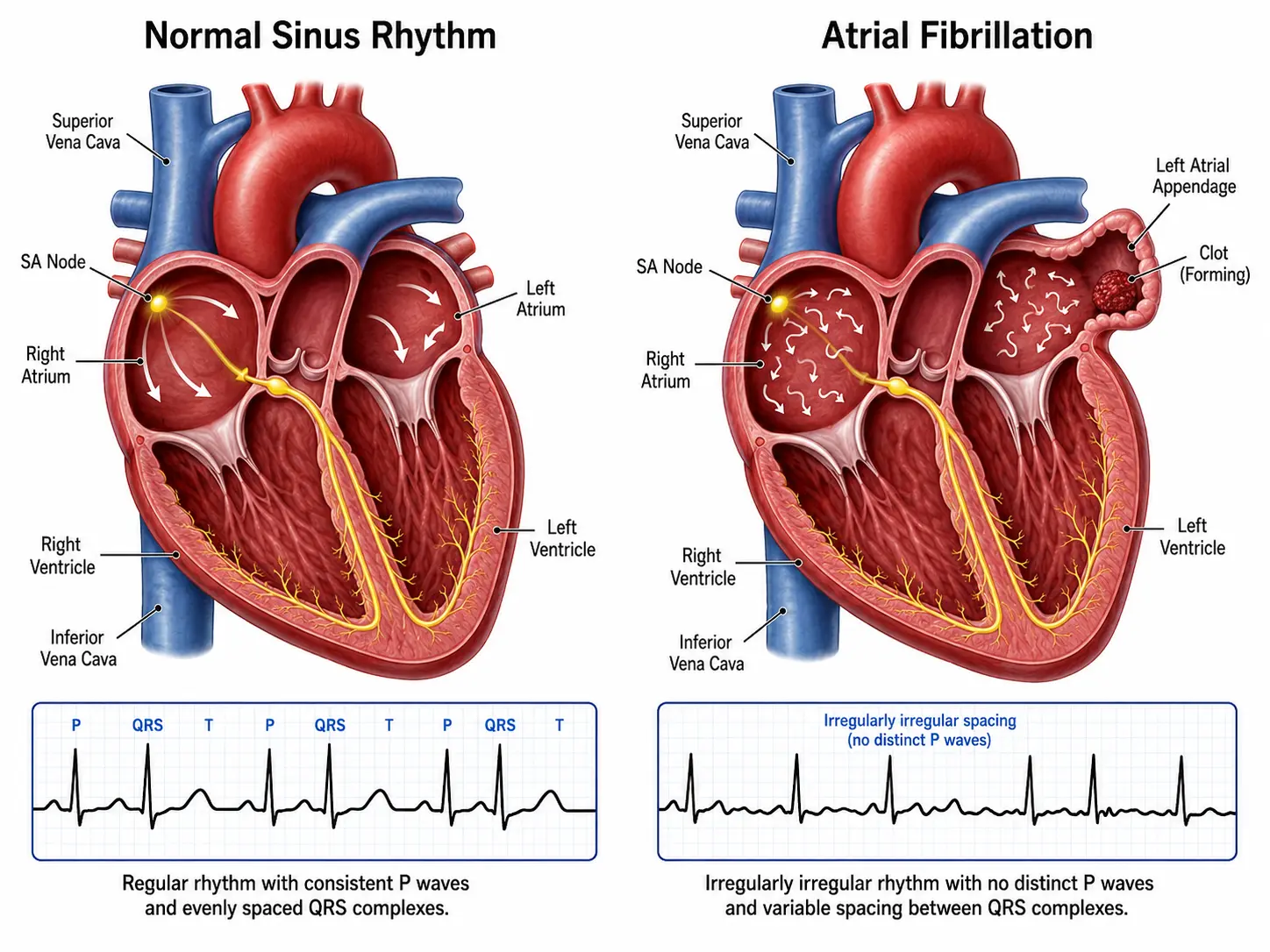

Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Atrial fibrillation (AF or AFib) is the most common sustained cardiac arrhythmia, characterized by rapid, disorganized electrical activity in the atria leading to an irregular and often rapid ventricular response. The normal coordinated atrial contraction is replaced by chaotic electrical impulses firing at rates of 350–600 beats per minute within the atrial myocardium, rendering the atria functionally ineffective as pumping chambers.
AF is classified by its temporal pattern: paroxysmal (self-terminating within 7 days), persistent (lasting more than 7 days or requiring cardioversion), long-standing persistent (continuous AF for more than 12 months), and permanent (when cardioversion has been abandoned by mutual patient-physician decision). A fifth category, subclinical AF, refers to episodes detected by implanted devices without clinical symptoms.
AF carries a five-fold increased risk of ischemic stroke and is independently associated with heart failure, cognitive decline, and increased all-cause mortality, making it a major public health burden.
2. Epidemiology
AF affects an estimated 37–59 million individuals worldwide as of 2023. In the United States, prevalence is approximately 6–7 million, projected to reach 12 million by 2030. Lifetime risk in individuals over age 40 is approximately 25% in developed nations.
Prevalence increases markedly with age: AF affects less than 1% of adults under 60 but rises to 10–17% in those over 80. Men have a 1.5-fold higher age-adjusted incidence than women, though women comprise nearly half of all AF patients due to longer life expectancy. AF is more prevalent in populations of European descent; individuals of African ancestry have historically had lower rates despite higher hypertension burden, though recent data suggest this gap is narrowing.
The annual incidence of AF-related stroke is 3–5%, accounting for approximately 15–20% of all ischemic strokes globally. AF-related strokes tend to be more severe and disabling than strokes from other causes. The economic burden exceeds $26 billion annually in the United States alone.
3. Pathophysiology
The pathophysiology of AF involves two interrelated processes: initiation triggers and perpetuating substrate.
Triggers
Rapid ectopic firing from pulmonary vein (PV) ostia represents the dominant trigger in paroxysmal AF. The myocardial sleeves extending into the PVs possess automaticity and re-entrant properties. Autonomic modulation — particularly simultaneous sympathetic and parasympathetic activation — plays a critical role in initiating these triggers. Non-PV triggers include the superior vena cava, ligament of Marshall, coronary sinus musculature, and posterior left atrial wall.
Substrate
Structural remodeling of the atrial myocardium creates an anatomical and electrophysiological substrate that perpetuates AF. Key components include:
- Atrial fibrosis: Activated fibroblasts transform into myofibroblasts under angiotensin II, TGF-β1, and inflammatory cytokine stimulation, depositing excess collagen. Fibrosis disrupts electrical coupling between cardiomyocytes, creating zones of conduction block and reentry circuits.
- Electrical remodeling: Shortened atrial effective refractory periods (AERPs) via downregulation of L-type calcium channels (CaV1.2) and upregulation of inward rectifier potassium current (IK1) and acetylcholine-activated potassium current (IKACh) reduce wavelength and favor reentry.
- Calcium handling abnormalities: Hyperphosphorylation of ryanodine receptor 2 (RyR2) by CaMKII and PKA leads to spontaneous calcium release from the sarcoplasmic reticulum, generating delayed afterdepolarizations (DADs) and triggered activity.
- Autonomic nervous system remodeling: Heterogeneous sympathetic and parasympathetic innervation of the atria, particularly the ganglionated plexi at PV–left atrial junctions, contributes to both initiation and maintenance.
The concept of "AF begets AF" (Wijffels et al., 1995) reflects the progressive nature of atrial remodeling: prolonged AF shortens AERP and promotes further structural changes, making the arrhythmia increasingly difficult to terminate over time.
4. Etiology and Risk Factors
Cardiac Causes
- Hypertension (most prevalent modifiable risk factor, present in up to 70% of AF patients)
- Coronary artery disease and myocardial infarction
- Heart failure (both HFrEF and HFpEF)
- Valvular heart disease (particularly mitral stenosis and mitral regurgitation)
- Hypertrophic cardiomyopathy
- Congenital heart disease (atrial septal defect)
- Sick sinus syndrome and other conduction disorders
- Pericarditis and myocarditis
- Cardiac surgery (postoperative AF occurs in 20–50% of cardiac surgery patients)
Non-Cardiac Systemic Causes
- Hyperthyroidism
- Obstructive sleep apnea (OR 2.19 for AF development)
- Obesity (BMI >30 associated with 50% increased AF risk)
- Diabetes mellitus
- Chronic kidney disease
- Pulmonary embolism and pneumonia
- Excessive alcohol consumption ("holiday heart syndrome")
- Electrolyte disturbances (hypokalemia, hypomagnesemia)
Genetic Factors
Family history confers a 40% increased risk. Genome-wide association studies (GWAS) have identified over 100 loci associated with AF, including variants near PITX2, ZFHX3, SCN5A, SCN10A, and HCN4. Rare monogenic causes include mutations in KCNQ1, KCNE1, SCN5A, and NPPA.
Lone AF
The term "lone AF" (AF without identifiable structural heart disease or risk factors) is increasingly discouraged as advanced imaging and genetic testing reveal subclinical substrates in most such patients.
5. Clinical Presentation
Symptoms
AF presentation ranges from asymptomatic (25–30% of patients) to severely symptomatic. Common symptoms include:
- Palpitations — irregular, rapid heartbeat sensation
- Dyspnea and exercise intolerance
- Fatigue and reduced functional capacity
- Presyncope or syncope (particularly with rapid ventricular rates)
- Chest discomfort or angina (especially with underlying CAD)
- Polyuria (due to ANP release from atrial distension)
EHRA Symptom Classification
The European Heart Rhythm Association (EHRA) classification grades AF-related symptoms:
- EHRA I: No symptoms
- EHRA IIa: Mild symptoms; normal daily activity not affected
- EHRA IIb: Moderate symptoms; normal daily activity not affected but patient troubled by symptoms
- EHRA III: Severe symptoms; normal daily activity affected
- EHRA IV: Disabling symptoms; normal daily activity discontinued
Physical Examination
Classic findings include an irregularly irregular pulse, pulse deficit (apical rate exceeding radial pulse rate), variable S1 intensity, and absence of distinct a-waves in the jugular venous pulse. Signs of heart failure (elevated JVP, S3, pulmonary crackles, peripheral edema) may be present when AF precipitates hemodynamic compromise.
6. Diagnosis
Electrocardiogram
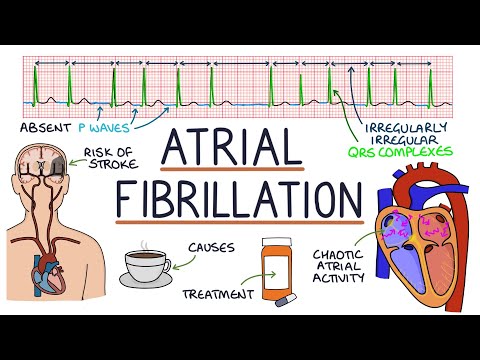
The diagnostic hallmark is the standard 12-lead ECG showing:
- Absent P waves replaced by irregular fibrillatory (f) waves at 350–600 bpm
- Irregularly irregular RR intervals (unless complete AV block is present)
- Narrow QRS complexes (unless aberrant conduction or pre-excitation is present)
Additional Diagnostic Tests
- Holter monitoring (24–48 hours): For paroxysmal AF documentation; sensitivity ~50% for 24-hour monitoring
- Extended cardiac monitoring (14–30 days): Patch monitors or event recorders increase detection yield to 70–80%
- Implantable loop recorders (ILR): Subcutaneous devices providing up to 3-year monitoring; recommended after cryptogenic stroke
- Echocardiography (TTE and TEE): Assesses structural heart disease, left atrial size and function, left ventricular ejection fraction, and — via TEE — rules out left atrial appendage (LAA) thrombus before cardioversion
- Laboratory workup: TSH (thyroid function), CBC, CMP, coagulation studies, BNP/NT-proBNP
- Chest radiograph: Evaluates pulmonary edema, cardiomegaly
- Sleep study: Consider in all AF patients to screen for obstructive sleep apnea
Stroke Risk Stratification: CHA₂DS₂-VASc Score
The CHA₂DS₂-VASc score quantifies annual stroke risk and guides anticoagulation decisions:
- C — Congestive heart failure (1 point)
- H — Hypertension (1 point)
- A₂ — Age ≥75 years (2 points)
- D — Diabetes mellitus (1 point)
- S₂ — Stroke/TIA/thromboembolism history (2 points)
- V — Vascular disease (prior MI, peripheral artery disease, or aortic plaque) (1 point)
- A — Age 65–74 years (1 point)
- Sc — Sex category female (1 point)
Anticoagulation is recommended for men with score ≥2 and women with score ≥3 (2023 ACC/AHA guidelines). Score of 0 in men or 1 in women (with female sex as sole point) indicates low risk where anticoagulation can be omitted.
Bleeding Risk Assessment: HAS-BLED Score
The HAS-BLED score identifies modifiable bleeding risk factors but should not be used to withhold anticoagulation; instead it guides optimization of reversible risk factors (uncontrolled hypertension, labile INR, concurrent NSAID/antiplatelet use, alcohol excess).
Differential Diagnosis
- Atrial flutter (regular sawtooth flutter waves at ~300 bpm with variable AV block)
- Multifocal atrial tachycardia (three or more distinct P-wave morphologies)
- Frequent premature atrial contractions
- AV nodal reentrant tachycardia
- Wolff-Parkinson-White syndrome with AF (wide complex irregular tachycardia — medical emergency)
7. Treatment
AF management follows the 4S-AF scheme (2020 ESC guidelines): Stroke prevention, Symptom management, Substrate and risk factor modification, and Surveillance. The ABC pathway (Anticoagulation/Avoid stroke; Better symptom management; Cardiovascular and comorbidity risk reduction) provides a structured clinical framework.
Stroke Prevention: Anticoagulation
Oral anticoagulation (OAC) is the cornerstone of AF stroke prevention and reduces stroke risk by approximately 64% compared to placebo.
- Direct oral anticoagulants (DOACs): Preferred over warfarin in non-valvular AF for superior efficacy and safety profile:
- Apixaban 5 mg twice daily (2.5 mg twice daily if ≥2 of: age ≥80, weight ≤60 kg, SCr ≥1.5 mg/dL) — ARISTOTLE trial
- Rivaroxaban 20 mg once daily with evening meal (15 mg if CrCl 15–49 mL/min) — ROCKET-AF trial
- Dabigatran 150 mg twice daily (110 mg in elderly/high bleed risk) — RE-LY trial
- Edoxaban 60 mg once daily (30 mg if CrCl 15–50 mL/min, weight ≤60 kg, or P-gp inhibitor use) — ENGAGE AF-TIMI 48 trial
- Warfarin: Target INR 2.0–3.0; preferred in mechanical heart valves and moderate-to-severe mitral stenosis (valvular AF)
- Left atrial appendage occlusion (LAAO): Watchman FLX device (FDA-approved 2021) for patients with high stroke risk and absolute contraindication to long-term OAC; non-inferior to warfarin for stroke prevention at 5 years in PROTECT-AF and PREVAIL trials
Rate Control
Target resting heart rate <110 bpm (lenient) is acceptable in most patients; tighter control (<80 bpm at rest) may be considered for symptomatic patients.
- Beta-blockers: Metoprolol succinate, carvedilol, bisoprolol — first-line, particularly with concomitant HF or CAD
- Non-dihydropyridine calcium channel blockers: Diltiazem, verapamil — contraindicated in HFrEF
- Digoxin: 0.125–0.25 mg/day; adjunct in heart failure with AF; trough level <1.0 ng/mL to minimize toxicity
- Amiodarone: Reserved for refractory cases; significant adverse effect profile
- AV node ablation + permanent pacemaker implantation: "Ablate and pace" strategy for refractory rapid ventricular rates when rate control medications fail
Rhythm Control
Rhythm control strategies include cardioversion and antiarrhythmic drugs (AADs). The EAST-AFNET 4 trial (2020) demonstrated that early rhythm control (within 1 year of AF diagnosis) reduces cardiovascular death, stroke, and heart failure hospitalization compared to rate control alone, shifting guidelines toward earlier rhythm control intervention.
- Electrical cardioversion (DC cardioversion): Synchronized monophasic or biphasic shock (200 J); requires ≥3 weeks therapeutic anticoagulation or TEE-guided exclusion of LAA thrombus before cardioversion, then ≥4 weeks post-cardioversion regardless of CHA₂DS₂-VASc score
- Antiarrhythmic drugs:
- Flecainide and propafenone (Class IC): First-line in structurally normal hearts; "pill-in-pocket" strategy for paroxysmal AF; contraindicated post-MI, with significant LV dysfunction
- Sotalol (Class III/II): Effective but requires QTc monitoring; initiation in monitored setting
- Dofetilide (Class III): In-hospital initiation required; renally cleared, dose-adjusted
- Amiodarone (Class III): Most effective AAD (~65% SR maintenance at 1 year) but significant long-term toxicity (pulmonary, hepatic, thyroid, dermatologic, neurologic); typically reserved for patients with structural heart disease or when other AADs fail
- Dronedarone (Class III, multichannel blocker): Safer alternative to amiodarone; contraindicated in permanent AF, severe HF, and recent ACS
Catheter Ablation
Pulmonary vein isolation (PVI) is the cornerstone of catheter ablation for AF. Radiofrequency ablation or cryoablation electrically disconnects PV ostia from the left atrial body. PVI is superior to AAD therapy for maintaining sinus rhythm in paroxysmal AF (FIRE AND ICE trial: cryoablation non-inferior to radiofrequency). For persistent AF, additional lesion sets (posterior wall isolation, linear lesions, complex fractionated atrial electrogram ablation) are variably employed, though their incremental benefit remains debated.
Current class I indications (AHA/ACC 2023): AF catheter ablation is recommended for symptomatic paroxysmal or persistent AF when AAD therapy has failed or is not tolerated. Early ablation (EAST-AFNET 4 subanalysis, EARLY-AF trial) is an emerging first-line strategy in younger patients or those with heart failure (CASTLE-AF trial: 38% relative risk reduction in death or worsening HF).
Surgical ablation: Cox-Maze IV procedure (bipolar radiofrequency or cryothermia lesion sets) during concomitant cardiac surgery achieves ~90% SR maintenance at 1 year. Thoracoscopic AF surgery (Wolf mini-Maze) is an option for failed catheter ablation.
Risk Factor and Lifestyle Modification
- Weight reduction: Every 10% body weight loss reduces AF burden significantly (LEGACY trial)
- Blood pressure optimization (target <130/80 mmHg)
- Glycemic control in diabetes
- OSA treatment with CPAP
- Alcohol moderation or abstinence (HOLIDAY HEART — dose-dependent relationship)
- Regular moderate aerobic exercise (structured exercise program reduces AF burden — ACTIVE-AF trial)
8. Complications
- Ischemic stroke: Annual risk 3–5% without anticoagulation; strokes are more severe (higher NIHSS, larger infarct volume) and disabling than non-AF strokes
- Systemic thromboembolism: Mesenteric, renal, limb ischemia from LAA thrombus embolization
- Heart failure: Tachycardia-induced cardiomyopathy with prolonged rapid ventricular rates; reversible with rate/rhythm control
- Cognitive impairment and dementia: AF independently associated with 40% increased dementia risk (REGARDS, Rotterdam cohort); mechanism includes subclinical microemboli and reduced cerebral perfusion
- Hemodynamic compromise: Loss of atrial contribution to ventricular filling ("atrial kick") reduces cardiac output by 10–30%, particularly in patients with diastolic dysfunction
- Sudden cardiac death: Increased 2-fold, partly mediated by associated structural heart disease and proarrhythmic effects of antiarrhythmic drugs
- Depression and anxiety: Prevalent in 30–40% of AF patients; bidirectional relationship with symptom burden
- Catheter ablation complications: Pulmonary vein stenosis, pericardial tamponade (1–2%), esophageal injury/atrio-esophageal fistula (rare but fatal), phrenic nerve palsy, stroke (0.5–1%)
9. Prognosis
AF carries a 1.5- to 2-fold increase in all-cause mortality, largely attributable to associated cardiovascular disease and thromboembolic events. With optimized anticoagulation and rhythm/rate control, the excess mortality risk is substantially attenuated.
- Stroke risk: Without OAC, CHA₂DS₂-VASc score 2 carries ~2% annual stroke risk; score 6 carries ~9.8% annual risk
- Sinus rhythm maintenance: At 5 years post-first catheter ablation, approximately 60–70% of paroxysmal AF patients remain in sinus rhythm; 40–50% of persistent AF patients require repeat procedures
- Progression: Paroxysmal AF progresses to persistent/permanent in ~15% per year; AF recurrence post-cardioversion is 50% at 1 year without AADs
- Quality of life: Successful rhythm control significantly improves QoL, exercise capacity, and functional class (AFFIRM, RACE II trials)
- Prognostic factors: Left atrial diameter >50 mm, long AF duration, advanced age, significant LV dysfunction, and multiple comorbidities predict worse outcomes
10. Prevention
Primary Prevention
- Aggressive hypertension management (strongest modifiable risk factor)
- Heart failure prevention and optimal treatment
- Weight management and obesity prevention
- Diabetes prevention and glycemic control
- OSA screening and treatment
- Alcohol moderation (risk increases with heavy use; low-to-moderate consumption may be acceptable)
- Regular moderate-intensity physical activity (excessive endurance training paradoxically increases AF risk — "athlete's heart")
- Mediterranean diet adherence associated with reduced AF incidence (PREDIMED study subanalysis)
Secondary Prevention (Recurrence Prevention)
- Sustained sinus rhythm maintenance via rhythm control strategy
- Upstream therapy: RAAS inhibition (ACE inhibitors, ARBs, ARNIs) reduces atrial remodeling and AF recurrence, particularly in HF patients
- Aldosterone antagonists (spironolactone, eplerenone) reduce atrial fibrosis
- SGLT2 inhibitors (empagliflozin, dapagliflozin) reduce AF risk and burden in HF patients (EMPEROR-Reduced, DAPA-HF subanalyses)
- Statin therapy reduces post-operative AF (mechanism: anti-inflammatory, antioxidant)
- Omega-3 fatty acids: mixed evidence; high-dose icosapentaenoic acid (Vascepa) potentially pro-arrhythmic in high doses per REDUCE-IT analyses
11. Recent Research and Advances
- Pulsed-field ablation (PFA): Non-thermal electroporation technology for AF ablation selectively targeting myocardial tissue while sparing adjacent structures (esophagus, phrenic nerve). MANIFEST-17K registry (2023) and PULSED-AF trial demonstrate comparable efficacy to thermal ablation with superior safety profile. FDA-approved PFA system (Farapulse) available since 2023.
- EAST-AFNET 4 trial (2020): Landmark trial demonstrating early rhythm control reduces composite of cardiovascular death, stroke, and HF hospitalization by 21% vs. usual care, establishing that rhythm control within 12 months of AF diagnosis improves outcomes.
- CASTLE-AF trial: Catheter ablation superior to medical therapy in AF patients with heart failure and reduced ejection fraction, with 38% reduction in all-cause mortality and HF hospitalization.
- Wearable and digital health technologies: Apple Watch, Fitbit, and AliveCor KardiaMobile demonstrate 71–98% sensitivity and 87–99% specificity for AF detection; integrated AI algorithms improve accuracy. Digital health platforms enable remote monitoring and telehealth-based AF management.
- Convergent procedure: Combined epicardial (surgical) and endocardial (catheter) ablation for persistent/longstanding persistent AF; CONVERGE trial (2020) demonstrated superiority over endocardial ablation alone.
- Left atrial appendage closure advances: Lariat suture-based device, Amulet occluder (non-inferior to DOAC in AMULET-IDE trial, 2021); LAA surgical ligation (LAAOS III trial, 2021: 33% stroke reduction with surgical LAA occlusion at time of cardiac surgery).
- Novel anticoagulants: Factor XIa inhibitors (asundexian, abelacimab) in phase III trials (OCEANIC-AF, AZALEA-TIMI 71) offer potential for stroke prevention with lower bleeding risk than current DOACs.
- Genetic and biomarker-guided therapy: Polygenic risk scores for AF identification; circulating biomarkers (fibrosis markers: galectin-3, ST2) to identify substrate for ablation outcome prediction.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Atrial fibrillation epidemiology
- Atrial fibrillation anticoagulation
- Atrial fibrillation catheter ablation
- CHA2DS2-VASc score
- Atrial fibrillation rate vs rhythm control
- Atrial fibrillation stroke prevention
- DOAC atrial fibrillation
- Atrial fibrillation guidelines
- Left atrial appendage closure
- Atrial fibrillation pathophysiology
- Paroxysmal atrial fibrillation
- Atrial fibrillation screening
Connections
- Stroke
- Heart Failure
- Hypertension
- Arrhythmia
- Valvular Heart Disease
- Deep Vein Thrombosis
- Heart Palpitations
- Fatigue
- Lightheadedness
- Magnesium
- Potassium
- Calcium
- Omega-3 Fatty Acids
- Cardiomyopathy
- Diabetes
- Obstructive Sleep Apnea
- Aspirin and Stroke Prevention
- Edema
Featured Videos

Atrial Fibrillation (AFib) | Q&A
Atrial Fibrillation Overview - ECG, types, pathophysiology, treatment, complications

Atrial Fibrillation (A-Fib) ECG, Treatment, Causes, Nursing NCLEX Review ACLS

Living with Atrial Fibrillation (AFib)

Atrial Fibrillation | Clinical Medicine

Atrial Fibrillation: New Solutions for an Old Problem

Atrial Fibrillation can be treated best by changing your diet and lifestyle.

An Atrial Fibrillation Cure? A Cardiologist reveals how to REVERSE and prevent atrial fibrillation!

Acute Atrial Fibrillation
Atrial Fibrillation and Catheter Ablation | NEJM

Atrial Fibrillation: What is it? - Plain English, no Doctor language!

Atrial Fibrillation: From A to Z
Understanding Atrial Fibrillation

Triggers for Atrial Fibrillation

Atrial Fibrillation: ALCOHOL, CAFFEINE, APPLE WATCHES

Atrial fibrillation (A-fib, AF) - causes, symptoms, treatment & pathology