Peripheral Artery Disease
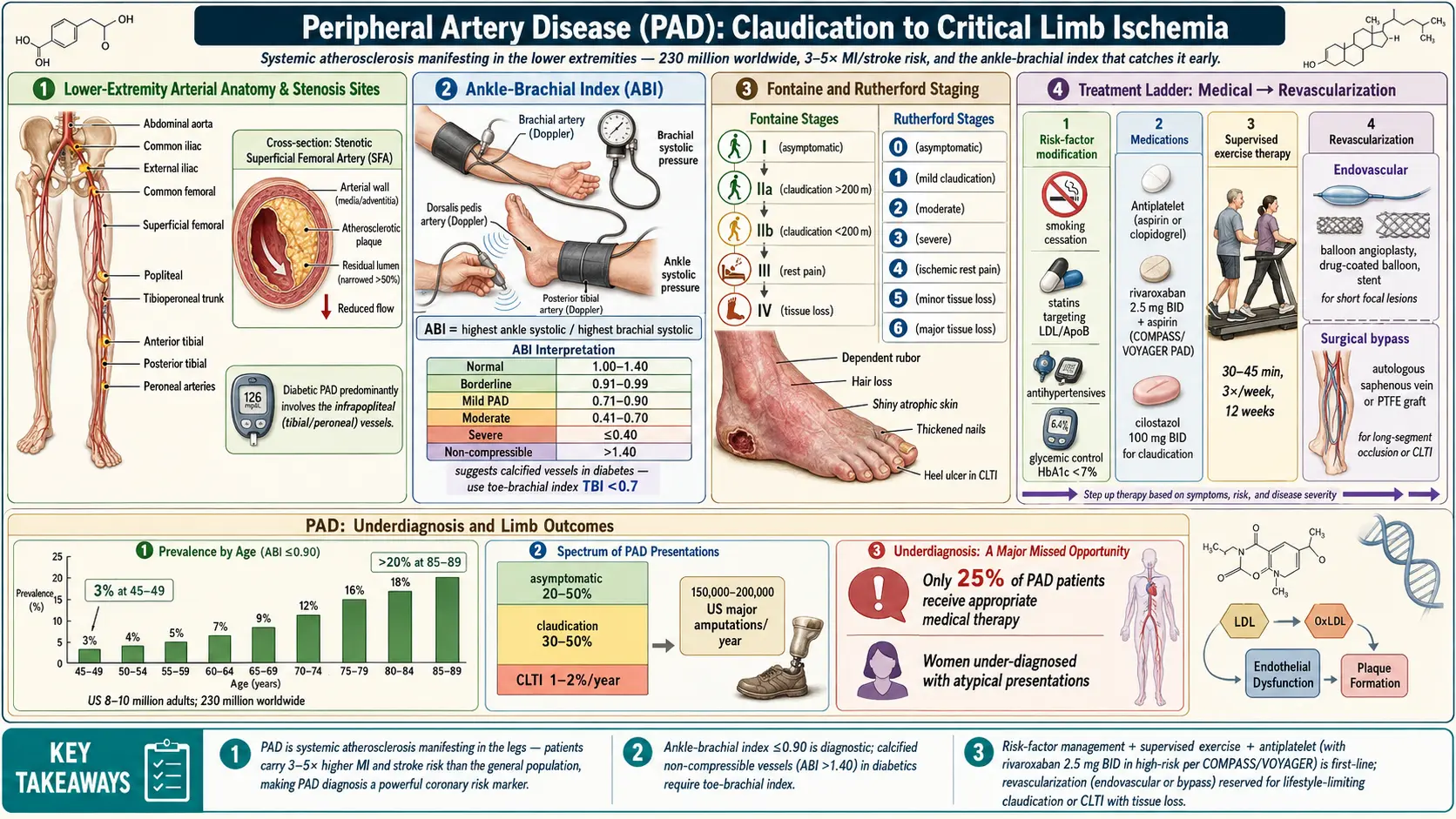
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Peripheral artery disease (PAD) is a manifestation of systemic atherosclerosis characterized by stenosis or occlusion of the arteries supplying the extremities — predominantly the lower limbs — resulting in impaired blood flow and tissue ischemia. PAD is both a significant cause of limb morbidity and a powerful marker of generalized cardiovascular disease, with affected patients carrying a 3- to 5-fold increased risk of myocardial infarction and stroke compared to the general population.
The term "PAD" most commonly refers to lower extremity arterial disease (LEAD), though the condition encompasses atherosclerotic involvement of the renal, mesenteric, and upper extremity arteries as well. The spectrum of lower-extremity PAD ranges from asymptomatic disease (diagnosed by ankle-brachial index) to intermittent claudication, to the most severe form — chronic limb-threatening ischemia (CLTI), formerly termed critical limb ischemia — which encompasses rest pain, non-healing ulcers, and gangrene.
2. Epidemiology
PAD affects approximately 230 million individuals worldwide, with a global prevalence of approximately 6.3% in adults over 25. Prevalence increases sharply with age: from 3% in those aged 45–49 to over 20% in adults aged 85–89. In the United States, approximately 8–10 million adults are affected.
PAD exhibits marked geographic variation. High-income countries account for 45% of global PAD burden, while sub-Saharan Africa and Oceania have the fastest-growing prevalence. Among racial/ethnic groups in the US, non-Hispanic Black individuals have a 2-fold higher prevalence than non-Hispanic White individuals, even after adjusting for traditional cardiovascular risk factors — likely reflecting disparities in hypertension prevalence, access to care, and socioeconomic factors.
Symptomatic claudication affects approximately 2% of adults over 50; however, the majority of PAD patients (20–50%) remain asymptomatic. CLTI affects approximately 1–2% of PAD patients annually, with an estimated 150,000–200,000 major amputations performed per year in the US.
PAD is significantly underdiagnosed: only 25% of patients with PAD receive appropriate medical therapy. Women with PAD often present atypically (less classic claudication, more functional limitations) and are diagnosed and treated less aggressively than men.
3. Pathophysiology
The fundamental pathologic process in PAD is atherosclerosis — a chronic inflammatory disorder of medium and large arteries driven by lipid deposition, endothelial dysfunction, and immune activation.
Atherosclerotic Plaque Formation
Endothelial dysfunction — triggered by oxidative stress, turbulent shear forces, dyslipidemia, hypertension, and smoking-derived toxins — leads to upregulation of adhesion molecules (VCAM-1, ICAM-1, P-selectin) and chemokines (MCP-1). Circulating monocytes adhere, transmigrate into the intima, and differentiate into macrophages. Macrophages engulf oxidized LDL via scavenger receptors (SR-A, CD36), becoming foam cells and forming the lipid-rich necrotic core of the atherosclerotic plaque.
Smooth muscle cells migrate from the media to the intima under PDGF and TGF-β stimulation, proliferating and secreting extracellular matrix proteins (collagen, elastin) that form the fibrous cap. Progressive plaque growth leads to luminal narrowing, and plaque rupture or erosion triggers acute thrombosis, causing acute limb ischemia.
Hemodynamic Consequences
Hemodynamically significant stenosis (>50% luminal diameter reduction, or >75% cross-sectional area) impairs distal blood flow. During exercise, peripheral vasodilation cannot overcome the fixed resistance of the stenosis, creating a pressure gradient that reduces perfusion pressure distal to the lesion — manifesting as claudication. In CLTI, resting perfusion is insufficient to maintain tissue viability, leading to ischemic rest pain (typically at night when legs are elevated) and tissue loss.
Collateral Circulation and Angiogenesis
Chronic ischemia stimulates collateral vessel formation via VEGF, FGF, and angiopoietin signaling pathways. The adequacy of collateral development determines the clinical severity of symptoms for a given degree of stenosis. Diabetic patients have impaired angiogenic responses, partly explaining their disproportionate risk of CLTI and amputation.
Microvascular Disease
In diabetic PAD, concurrent microvascular disease (arteriolar sclerosis, capillary basement membrane thickening) compounds macrovascular occlusion, worsening tissue ischemia and impairing wound healing. Diabetic neuropathy exacerbates foot ulceration by reducing protective pain sensation.
Inflammation and Coagulation
Systemic inflammation (elevated CRP, IL-6, TNF-α, fibrinogen) promotes platelet activation, endothelial dysfunction, and plaque vulnerability. Hypercoagulable states (antiphospholipid syndrome, factor V Leiden) can accelerate thrombotic occlusion in PAD.
4. Etiology and Risk Factors
Traditional Cardiovascular Risk Factors
- Cigarette smoking: Strongest modifiable risk factor for PAD (OR 2.3–4.0); dose-dependent relationship; smoking cessation reduces progression risk by 30–50%
- Diabetes mellitus: 2- to 4-fold increased PAD risk; accelerates atherosclerosis and microvascular disease; HbA1c >7% associated with worse outcomes; diabetic PAD predominantly affects infrapopliteal (tibial/peroneal) vessels
- Hypertension: 2.5-fold increased risk; end-organ damage compounds lower extremity ischemia
- Dyslipidemia: Elevated LDL-C (particularly small dense LDL particles), low HDL-C, elevated triglycerides, and elevated Lp(a) all increase PAD risk
- Chronic kidney disease: Strong independent predictor; eGFR <60 mL/min/1.73m² associated with 2-fold PAD risk
Non-Modifiable Risk Factors
- Age >65 years (strongest predictor)
- Male sex (though female prevalence approaches male in older cohorts)
- Family history of premature atherosclerosis
- African American race
Other Risk Factors
- Hyperhomocysteinemia
- Elevated C-reactive protein and inflammatory markers
- Elevated lipoprotein(a)
- Hypercoagulable states (antiphospholipid antibody syndrome, thrombophilias)
- HIV infection (accelerated atherosclerosis from chronic inflammation and antiretroviral therapy)
Non-Atherosclerotic Causes
- Buerger disease (thromboangiitis obliterans): Inflammatory segmental arteritis in young heavy smokers
- Popliteal artery entrapment syndrome: Congenital anomalous muscle-artery relationship
- Cystic adventitial disease
- Radiation-induced arteriopathy
- Takayasu arteritis and giant cell arteritis (large vessel vasculitis)
- Fibromuscular dysplasia (more common in renal and carotid arteries)
5. Clinical Presentation
Asymptomatic PAD
Approximately 40–50% of PAD patients are asymptomatic, identified only by abnormal ankle-brachial index (ABI). Despite absent symptoms, these individuals carry the same cardiovascular risk as symptomatic patients.
Intermittent Claudication
The classic presentation of symptomatic PAD is intermittent claudication — reproducible cramping, aching, or tightness pain in the exercising muscle groups distal to the stenosis, consistently reproduced by a fixed exercise distance (claudication distance), and relieved within 2–5 minutes of rest. Claudication location identifies the approximate level of disease:
- Calf claudication: Femoropopliteal disease (most common)
- Thigh and buttock claudication: Aortoiliac disease; Leriche syndrome (bilateral claudication, absent femoral pulses, erectile dysfunction)
- Foot claudication: Tibial/peroneal disease
Fontaine and Rutherford Classification
The Fontaine classification stages PAD severity:
- Stage I: Asymptomatic, ABI <0.9
- Stage IIa: Mild claudication (>200 m walking distance)
- Stage IIb: Moderate to severe claudication (≤200 m walking distance)
- Stage III: Ischemic rest pain
- Stage IV: Ulceration or gangrene
The Rutherford classification (Grades 0–III, Categories 0–6) is used in clinical research with greater granularity. The updated WIfI classification (Wound, Ischemia, foot Infection) is specifically designed for CLTI to predict amputation risk and revascularization benefit.
Chronic Limb-Threatening Ischemia (CLTI)
CLTI represents the most severe form of PAD and is defined by:
- Ischemic rest pain: Persistent pain at rest (usually forefoot/toes) for >2 weeks, typically worse at night and partially relieved by leg dependency; ABI <0.4, ankle pressure <50 mmHg, or toe pressure <30 mmHg
- Ischemic ulceration: Non-healing wounds persisting >2 weeks despite optimal wound care; punched-out ulcers with pale bases, often on toes, heel, or pressure points
- Gangrene: Dry (mummified) or wet (infected) tissue necrosis; wet gangrene constitutes a surgical emergency
Acute Limb Ischemia (ALI)
The six Ps characterize ALI: Pain, Pallor, Pulselessness, Paresthesias, Paralysis (most ominous), and Poikilothermia (coolness). ALI is a vascular emergency requiring intervention within 6 hours to prevent irreversible ischemia. The Rutherford classification for ALI grades severity from I (viable) to III (irreversible/major tissue loss).
6. Diagnosis
Ankle-Brachial Index (ABI)
The ABI is the primary diagnostic test: ratio of systolic blood pressure at the ankle (posterior tibial or dorsalis pedis) to the higher brachial systolic pressure. Measured using a continuous wave Doppler probe and sphygmomanometer.
- Normal: 1.00–1.40
- Borderline: 0.91–0.99
- PAD: ≤0.90 (sensitivity 79%, specificity 96% for angiographically confirmed ≥50% stenosis)
- Severe ischemia: ≤0.40
- Non-compressible (falsely elevated, >1.40): Due to medial arterial calcification — common in diabetes and CKD; requires toe-brachial index (TBI; normal ≥0.70)
Exercise ABI (post-treadmill 5 minutes at 3.2 km/h, 10% grade): Drop of ≥20% from resting ABI confirms claudication when resting ABI is borderline or normal despite suggestive symptoms.
Additional Vascular Studies
- Segmental limb pressures and pulse volume recordings (PVRs): Localize disease level; PVRs qualitatively assess flow waveform morphology
- Duplex ultrasound: Defines anatomy, stenosis severity (peak systolic velocity ratio >2.0 indicates ≥50% stenosis), plaque morphology; operator-dependent
- CT angiography (CTA): High-resolution 3D arterial mapping; excellent for pre-procedural planning; risk of contrast nephropathy and radiation; artifact from arterial calcification
- Magnetic resonance angiography (MRA): No ionizing radiation; gadolinium-enhanced MRA provides excellent anatomic detail; limited by metallic implants and nephrogenic systemic fibrosis risk in severe CKD
- Digital subtraction angiography (DSA): Gold standard for anatomy definition; reserved for when non-invasive imaging is inconclusive or immediately preceding intervention
- Transcutaneous oxygen pressure (TcPO₂): Measures tissue oxygenation; TcPO₂ <30 mmHg predicts impaired wound healing; useful in patients with non-compressible arteries
- Toe pressures: Absolute toe pressure <50 mmHg (or <30 mmHg with diabetes) defines CLTI
Laboratory Evaluation
- Fasting lipid panel, HbA1c, fasting glucose
- CBC, CMP, coagulation studies
- Creatinine and eGFR (pre-contrast administration)
- Homocysteine, Lp(a) in young patients or atypical presentations
- Inflammatory markers (hsCRP, ESR) if vasculitis suspected
- Hypercoagulable workup when appropriate (antiphospholipid antibodies, protein C/S, antithrombin III)
Differential Diagnosis
- Neurogenic claudication (lumbar spinal stenosis): Pain with walking and standing; relieved by sitting/forward flexion; normal ABI
- Venous claudication: Bursting thigh pain with exercise; associated with post-thrombotic syndrome
- Musculoskeletal pain (osteoarthritis, hip disease): Not consistently reproducible with same exercise distance
- Compartment syndrome (chronic exertional): Young athletes; pain with exercise, rapid relief with rest
- Diabetic neuropathic ulcers: Plantar pressure points; warm foot; bounding pulses (unless concurrent PAD)
- Venous ulcers: Medial malleolus location; associated lipodermatosclerosis, varicosities
7. Treatment
Medical Management (All PAD Patients)
Optimal medical therapy (OMT) reduces cardiovascular events and limb outcomes in all PAD patients regardless of symptom severity.
- Antiplatelet therapy:
- Aspirin 75–100 mg/day: Reduces MI, stroke, and cardiovascular death; class I recommendation for symptomatic PAD
- Clopidogrel 75 mg/day: Marginally superior to aspirin in PAD (CAPRIE trial: 23.8% relative risk reduction vs. aspirin); preferred in aspirin-intolerant patients or those with concurrent coronary artery disease
- Aspirin + rivaroxaban 2.5 mg twice daily (COMPASS regimen): Reduces major adverse limb events (MALE) by 46% and major adverse cardiovascular events (MACE) by 28% compared to aspirin alone (COMPASS trial, 2017); FDA-approved for PAD; increases bleeding risk
- Ticagrelor: Not superior to clopidogrel in PAD (EUCLID trial, 2017)
- Statin therapy: High-intensity statin therapy (atorvastatin 40–80 mg, rosuvastatin 20–40 mg); LDL-C target <55 mg/dL (ESC 2019) or <70 mg/dL (AHA/ACC); reduces MACE and MALE; PCSK9 inhibitors (evolocumab, alirocumab) for patients not at LDL-C goal on maximally tolerated statin
- Blood pressure control: Target <130/80 mmHg; ACE inhibitors or ARBs preferred (HOPE trial: ramipril 10 mg reduced MACE by 22% in high-risk patients including PAD); beta-blockers are not contraindicated in PAD and may be used for compelling indications (post-MI, HF)
- Glycemic control: HbA1c <7–8% in most diabetic PAD patients; SGLT2 inhibitors (empagliflozin, canagliflozin) reduce cardiovascular events and may have specific limb benefit (though canagliflozin associated with amputation signal in CANVAS trial)
- Smoking cessation: Most impactful single intervention; reduces claudication progression by 30–50% and major amputation risk; pharmacotherapy (varenicline, bupropion, nicotine replacement) and behavioral support
- Cilostazol (phosphodiesterase III inhibitor): 100 mg twice daily; improves maximum and pain-free walking distance by 40–60% in claudication (FDA-approved); contraindicated in heart failure of any severity; drug interactions with CYP3A4/CYP2C19 inhibitors
- Pentoxifylline: 400 mg three times daily; modest benefit; generally second-line to cilostazol
Supervised Exercise Therapy
Structured, supervised exercise programs (SET) — typically 30–60 minutes of treadmill walking 3 times/week for 12 weeks — improve maximum walking distance by 100–150% and are guideline-recommended as first-line therapy for claudication (Class I, Level A). The CLEVER trial demonstrated SET superior to stenting alone for quality-of-life improvement in aortoiliac PAD at 6 months (though revascularization + SET may be synergistic).
Revascularization
Revascularization is indicated for: (1) lifestyle-limiting claudication refractory to OMT + SET; (2) CLTI (urgent/emergent in ALI). The BEST-CLI trial (2022) compared surgical bypass vs. endovascular therapy in CLTI patients and found surgical bypass (specifically with single-segment great saphenous vein conduit) was superior in patients with adequate conduit.
- Endovascular therapy (EVT):
- Percutaneous transluminal angioplasty (PTA): Balloon dilation of stenotic/occlusive lesions
- Bare-metal stents (BMS) and drug-eluting stents (DES): Nitinol self-expanding stents for femoropopliteal disease; DES (paclitaxel, sirolimus-eluting) reduce restenosis vs. BMS
- Drug-coated balloons (DCB): Paclitaxel-coated; improve patency in femoropopliteal disease
- Atherectomy (directional, rotational, orbital, laser): Debulking prior to balloon/stent; role in calcified lesions
- Covered stent-grafts: For SFA occlusions; VIBRANT trial
- Surgical bypass:
- Aortobifemoral bypass: Gold standard for aortoiliac occlusive disease (Leriche syndrome); 5-year patency 85–90%
- Femoropopliteal bypass: Above-knee (prosthetic or vein, 5-year patency 70–75%) vs. below-knee (vein preferred, 5-year patency 65–70%)
- Tibial bypasses: For CLTI with infrapopliteal disease; autologous vein essential
- Hybrid procedures: Combination of surgical inflow reconstruction and endovascular outflow treatment in multilevel disease
- Amputation: Major amputation (below-knee or above-knee) is the procedure of last resort when revascularization is not technically feasible or patient is not a candidate; multidisciplinary limb salvage programs aim to reduce major amputation rates
8. Complications
- Major adverse cardiovascular events (MACE): Myocardial infarction is the leading cause of death in PAD patients; 3- to 5-fold increased risk; annual cardiovascular death rate 5–7%
- Major adverse limb events (MALE): Acute limb ischemia, major amputation, urgent revascularization; annual risk 5–10% in claudicants; much higher in CLTI
- Major amputation: 25–40% of CLTI patients require major amputation within 1 year without revascularization; 1-year mortality after major amputation is 30–50%
- Acute limb ischemia (ALI): Sudden thrombotic or embolic arterial occlusion; surgical emergency; 30-day mortality 15–20%; 30-day amputation rate 10–30%
- Wound complications: Non-healing ulcers, osteomyelitis, gas gangrene (wet gangrene) requiring emergency amputation
- Restenosis: Following endovascular intervention; rates of 30–50% at 1 year for femoropopliteal disease with plain balloon angioplasty; reduced with DCB and DES
- Graft failure: Prosthetic graft infection is a devastating complication requiring explantation
- Contrast nephropathy: Following angiographic procedures, particularly in diabetic or CKD patients
- Functional decline and depression: Claudication significantly limits physical activity, social participation, and quality of life
9. Prognosis
PAD is a marker of systemic atherosclerosis and portends significant cardiovascular morbidity and mortality. The "rule of thirds" for claudication (over 5 years): one-third improve, one-third remain stable, one-third worsen. However, 20–30% of claudicants progress to CLTI over 10 years, particularly those who continue to smoke or have diabetes.
- Cardiovascular mortality: Annual cardiovascular death rate 5–7% in symptomatic PAD; 10-year mortality approximately 50%, predominantly from MI and stroke
- CLTI outcomes: Without revascularization, 1-year mortality is 25%, 1-year amputation rate is 25%; together (death or amputation) affecting 50% of CLTI patients at 1 year
- Post-revascularization: Successful revascularization in CLTI improves limb salvage to 75–85% at 1 year; long-term survival remains poor due to cardiovascular comorbidities
- ABI as prognostic marker: Each 0.10 decrease in ABI below normal is associated with a 10% increase in cardiovascular event risk; ABI <0.40 is associated with the highest cardiovascular mortality
- Prognostic factors: Continued smoking, diabetes, renal insufficiency, low serum albumin (nutritional status), and absence of revascularization are associated with poorer outcomes
10. Prevention
Primary Prevention
- Smoking cessation is paramount — the single greatest modifiable risk factor
- Aggressive dyslipidemia management; statin therapy in all high-risk patients
- Optimal blood pressure control (<130/80 mmHg)
- Diabetes screening and glycemic optimization
- Regular physical activity (150+ minutes/week of moderate-intensity exercise)
- Healthy dietary patterns (Mediterranean or DASH diet)
- Weight management (BMI <30 kg/m²)
- Annual foot examination in diabetic patients for early ulcer detection
Secondary Prevention
- Antiplatelet therapy in all symptomatic PAD patients
- Anticoagulation (aspirin + low-dose rivaroxaban) consideration per COMPASS criteria
- High-intensity statin therapy with LDL-C target <55–70 mg/dL
- ACE inhibitor or ARB therapy
- Supervised exercise rehabilitation
- Diabetic foot care programs: Protective footwear, podiatric surveillance, patient education
- Multidisciplinary wound care teams for early ulcer management
- Cardiac risk factor optimization and cardiology co-management
11. Recent Research and Advances
- COMPASS trial (2017): Aspirin 100 mg + rivaroxaban 2.5 mg twice daily vs. aspirin alone demonstrated 46% reduction in MALE and 28% reduction in MACE in PAD patients; led to FDA approval of this combination regimen and transformed PAD medical management.
- BEST-CLI trial (2022): First randomized trial comparing bypass surgery vs. endovascular therapy for CLTI; demonstrated superiority of surgical bypass (using single-segment great saphenous vein) for the primary endpoint of major adverse limb events, death, or major amputation in cohort 1 (adequate single-segment GSV); opened important discussions about patient selection for revascularization strategy.
- VOYAGER-PAD trial (2020): Rivaroxaban 2.5 mg twice daily + aspirin vs. aspirin alone post-PAD revascularization; 15% relative risk reduction in MACE/MALE; established DOAC role in post-revascularization patients.
- Drug-coated balloons (DCB) advances: Multiple randomized trials (IN.PACT ADMIRAL, ILLUMENATE) demonstrate DCB superiority over plain balloon angioplasty for femoropopliteal disease; ongoing investigation of safety (paclitaxel mortality signal subsequently addressed by larger meta-analyses).
- Cell-based therapies and gene therapy: Angiogenic gene therapy (HIF-1α, VEGF, FGF) and autologous cell therapies (bone marrow-derived mononuclear cells, CD34+ cells) in phase II/III trials for no-option CLTI patients; TAMARIS trial (non-rAd-VEGF165 for CLTI) was negative; newer approaches with plasmid-based delivery (VM202/pVGI.1) show promise in early trials.
- Intravascular lithotripsy (IVL): Shockwave Medical device for heavily calcified PAD lesions; disrupts vascular calcification without wire trauma; improving EVT outcomes in calcified vessels.
- Artificial intelligence and PAD: AI algorithms for automated ABI interpretation, wound classification (WIfI scoring), and 3D vessel analysis from CT angiography are improving diagnostic accuracy and procedural planning.
- SGLT2 inhibitor effects on PAD: Post-hoc analyses of EMPA-REG, CANVAS, CREDENCE, and DECLARE trials evaluating specific limb outcomes; conflicting signals between agents warrant dedicated PAD trials.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Peripheral artery disease epidemiology
- Ankle brachial index
- Intermittent claudication
- Critical limb ischemia
- Peripheral artery disease revascularization
- Peripheral artery disease supervised exercise
- Peripheral artery disease guidelines
- Cilostazol claudication
- Peripheral artery disease statin
- Peripheral artery disease smoking
- Peripheral artery disease diabetes
- Peripheral artery disease mortality
Connections
- Stroke
- Hypertension
- Cardiovascular Disease
- Coronary Artery Disease
- Atherosclerosis
- Aortic Aneurysm
- Deep Vein Thrombosis
- Cold Hands and Feet
- Lipid Panel
- Inflammatory Markers
- Garlic
- Omega-3 Fatty Acids
- Diabetes
- Erectile Dysfunction
- Aspirin
- Heart Failure
- Ginkgo Biloba
- Lipoprotein a
Featured Videos
Understanding Peripheral Arterial Disease

Peripheral Vascular Disease

Peripheral Arterial Disease | Clinical Medicine

Avoiding Leg Amputations Due to Peripheral Arterial Disease | Q&A

PAD: Get the facts on peripheral artery disease

Peripheral Artery Disease (PAD): Symptoms & Treatments - Ask A Nurse | @LevelUpRN
Peripheral Arterial Disease for the Internist

Are you at risk for Peripheral Artery Disease?

Peripheral Arterial Disease & Peripheral Bypass Graft - Medical-Surgical | @LevelUpRN
Peripheral vascular disease, Causes, Signs and Symptoms, Diagnosis and Treatment.

Peripheral artery disease: Symptoms

Peripheral Artery Disease (Full Lecture)

How to treat leg Pain & Peripheral Artery Disease.

Dark Chocolate and Peripheral Artery Disease with Mary McDermott, MD

Treatment for Peripheral Artery Disease
Peripheral Artery Disease Treatment | Advanced Cutting-Edge Options #pad #peripheralarterydisease