Pancreatitis
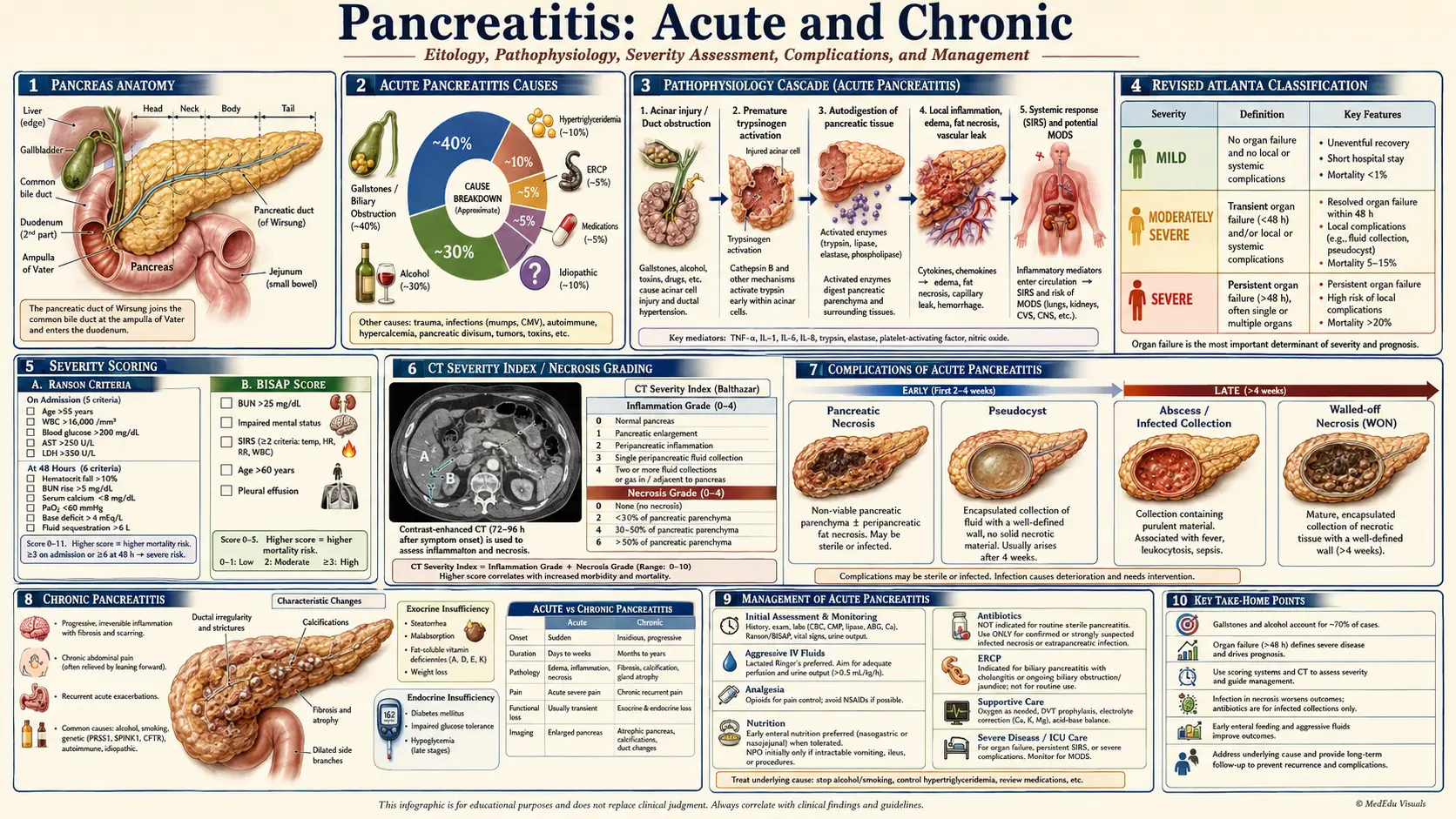
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Pancreatitis is an inflammatory condition of the pancreas caused by premature activation of pancreatic digestive enzymes within the gland, resulting in autodigestion. It encompasses a spectrum from acute pancreatitis (AP) — a discrete episode of pancreatic inflammation that typically resolves completely — to chronic pancreatitis (CP), a progressive fibro-inflammatory disease leading to permanent structural and functional damage of the exocrine and endocrine pancreas.
Acute pancreatitis is classified by the revised Atlanta criteria (2012) as mild, moderately severe, or severe, based on the presence and duration of organ failure and local complications. Chronic pancreatitis is characterized by irreversible pancreatic parenchymal fibrosis, ductal changes, and loss of acinar and islet tissue, resulting in exocrine insufficiency (malabsorption) and endocrine insufficiency (pancreatogenic diabetes mellitus — Type 3c DM).
2. Epidemiology
Acute pancreatitis is the most common gastrointestinal cause of hospital admission in the United States, with an annual incidence of 13–45 per 100,000 population and approximately 300,000 hospitalizations per year. The incidence is rising globally, likely due to increasing rates of gallstone disease, obesity, and alcohol consumption. Overall mortality from AP is approximately 2–3%, rising to 15–30% in severe necrotizing pancreatitis with infected necrosis.
Chronic pancreatitis affects approximately 50 per 100,000 adults in developed countries, with a prevalence of 10–15 per 100,000. It is more common in men, with a male-to-female ratio of approximately 3:1, reflecting higher rates of heavy alcohol use in men. The prevalence of CP is increasing due to improved survival of AP and better recognition of non-alcoholic etiologies. Chronic pancreatitis carries a standardized mortality ratio of 2–4-fold above the general population.
3. Pathophysiology
Acute Pancreatitis
The central event in AP is the inappropriate intracellular activation of trypsinogen to trypsin within pancreatic acinar cells, before secretion into the duodenum. This triggers a cascade:
- Acinar cell injury: Impaired intracellular calcium signaling, mitochondrial dysfunction, and impaired secretory vesicle trafficking cause co-localization of lysosomal cathepsin B with zymogen granules, activating trypsinogen to trypsin within the acinar cell
- Trypsin activation cascade: Active trypsin activates downstream zymogens — chymotrypsinogen, proelastase, phospholipase A2 — initiating autodigestion of pancreatic parenchyma, peripancreatic fat, and vasculature
- Local inflammatory response: Acinar cell injury triggers release of DAMP signals (HMGB1, ATP), activating pancreatic stellate cells and resident macrophages; NF-κB activation drives production of TNF-α, IL-1β, IL-6, IL-8, and PAF (platelet-activating factor)
- Systemic inflammatory response syndrome (SIRS): In severe AP, overwhelming cytokine release leads to SIRS, causing capillary leak, third-space fluid sequestration, and ultimately multi-organ dysfunction syndrome (MODS) affecting lungs (ARDS), kidneys (AKI), and cardiovascular system (shock)
- Pancreatic necrosis: Microvascular thrombosis and ischemia lead to pancreatic parenchymal and peripancreatic fat necrosis in 15–20% of AP cases; secondary bacterial infection of necrosis (infected necrotizing pancreatitis) dramatically worsens prognosis
Chronic Pancreatitis
The sentinel acute pancreatitis event (SAPE) hypothesis proposes that repeated episodes of acinar cell injury activate pancreatic stellate cells (PSCs). Activated PSCs produce collagen and fibronectin, leading to fibrosis. Multiple mechanisms contribute:
- Oxidative stress from alcohol metabolism activates PSCs via acetaldehyde and reactive oxygen species (ROS)
- Obstruction of small pancreatic ducts by protein plugs (in alcoholic CP) or ductal strictures triggers upstream inflammation
- Genetic mutations (PRSS1, SPINK1, CFTR, CTRC) predispose to trypsin-driven acinar injury
- TGF-β1 is the principal fibrogenic cytokine driving PSC activation and collagen deposition
- Neurogenic inflammation: increased density of pancreatic nociceptive nerve fibers (CGRP+, substance P+) contributes to the hallmark chronic pain of CP
4. Etiology and Risk Factors
Acute Pancreatitis — TIGARO Classification
- Toxic: Alcohol (accounts for 30–35% of AP in developed countries); smoking (independent risk factor); drugs (azathioprine, 6-mercaptopurine, valproate, tetracycline, metronidazole, didanosine, sulfonamides, furosemide, corticosteroids, ACE inhibitors, GLP-1 receptor agonists)
- Idiopathic: 10–20% of cases; may include microlithiasis (biliary sludge), sphincter of Oddi dysfunction, genetic mutations, or autoimmune pancreatitis that is not recognized
- Gallstones/metabolic: Gallstone-associated biliary pancreatitis is the most common cause (40–50% of AP); hypertriglyceridemia (serum TG >1,000 mg/dL) accounts for 1–4%; hypercalcemia (hyperparathyroidism)
- Anatomic/autoimmune: Pancreatic divisum, choledochal cyst, ampullary stenosis, autoimmune pancreatitis (IgG4-related)
- Radiation/post-procedural: Post-ERCP pancreatitis (3–5% after ERCP; up to 15% in high-risk patients); radiation-induced
- Obstructive: Pancreatic or ampullary tumors, parasitic infection (Ascaris lumbricoides)
Chronic Pancreatitis — TIGAR-O Classification
- Toxic-metabolic: Alcohol (70–80% of CP in developed countries); tobacco (independent dose-dependent risk factor, synergistic with alcohol); hyperlipidemia; hypercalcemia
- Idiopathic: Early onset (juvenile) and late onset forms
- Genetic:
- PRSS1 gain-of-function mutations (hereditary pancreatitis — autosomal dominant; first episode typically before age 20)
- SPINK1 N34S loss-of-function (reduces trypsin inhibitor activity; risk modifier, not causative alone)
- CFTR mutations (cystic fibrosis and CFTR-related pancreatitis)
- CTRC (chymotrypsinogen C) loss-of-function mutations
- CASR mutations (calcium-sensing receptor)
- Autoimmune: Type 1 AIP (IgG4-related systemic disease — lymphoplasmacytic sclerosing pancreatitis); Type 2 AIP (idiopathic duct-centric pancreatitis — associated with IBD)
- Recurrent/severe acute pancreatitis: Post-necrotizing pancreatitis progression to CP
- Obstructive: Pancreatic ductal adenocarcinoma, intraductal papillary mucinous neoplasm (IPMN), post-traumatic stricture
5. Clinical Presentation
Acute Pancreatitis
- Abdominal pain: Sudden onset, severe, constant, epigastric pain radiating to the back; "boring" or "band-like" quality; worse in supine position; relieved by leaning forward (pancreatitis position)
- Nausea and vomiting (not relieving pain — distinguishes from biliary colic)
- Fever (<38.5°C in uncomplicated AP; higher fever suggests infected necrosis or cholangitis)
- Tachycardia and hypotension (from third-space fluid loss)
- Abdominal tenderness (epigastric; guarding if peritonitis from perforation or retroperitoneal inflammation)
- Abdominal distension (paralytic ileus)
- Cullen's sign: periumbilical ecchymosis (retroperitoneal hemorrhage tracking to umbilicus — <3% of cases, indicates hemorrhagic pancreatitis)
- Grey Turner's sign: flank ecchymosis (retroperitoneal hemorrhage tracking laterally — <3% of cases)
- Jaundice: if biliary obstruction by gallstone or pancreatic head inflammation
Chronic Pancreatitis
- Chronic abdominal pain: The dominant symptom in 80% of patients; epigastric, persistent or intermittent; radiates to back; exacerbated by eating (causing food aversion); neuropathic quality (burning, stabbing) in advanced disease
- Exocrine pancreatic insufficiency (EPI): Develops when >90% of exocrine function is lost; steatorrhea (bulky, oily, pale, floating stools); malabsorption of fat-soluble vitamins (A, D, E, K); weight loss; nutritional deficiencies
- Endocrine insufficiency: Type 3c (pancreatogenic) diabetes mellitus; characterized by insulin deficiency with preserved glucagon secretion — leading to erratic glucose control and high hypoglycemia risk; develops in 40–80% of CP patients over 20 years
- Jaundice (biliary stricture from fibrosis of the pancreatic head)
- Signs of portal hypertension (splenic vein thrombosis)
6. Diagnosis
Acute Pancreatitis — Diagnostic Criteria
Diagnosis requires 2 of 3 criteria (revised Atlanta Classification, 2012):
- Characteristic abdominal pain (epigastric, radiating to back)
- Serum lipase or amylase ≥3× upper limit of normal (ULN)
- Characteristic findings on cross-sectional imaging (CT or MRI)
Note: Cross-sectional imaging is not required if criteria 1 and 2 are met. CT with intravenous contrast (CECT) is reserved for patients with diagnostic uncertainty, failure to improve after 48–72 hours, or suspected complications.
Ranson Criteria (Severity Assessment at Admission and 48 hours)
At admission:
- Age >55 years
- WBC >16,000/mm³
- Blood glucose >200 mg/dL
- Serum LDH >350 IU/L
- AST >250 IU/L
Within 48 hours:
- Hematocrit fall >10%
- BUN rise >5 mg/dL
- Serum calcium <8 mg/dL
- PaO2 <60 mmHg
- Base deficit >4 mEq/L
- Fluid sequestration >6 L
Interpretation: 0–2 criteria: mild (1% mortality); 3–4 criteria: moderate (15% mortality); 5–6 criteria: severe (40% mortality); ≥7 criteria: near 100% mortality.
Bedside Index of Severity in Acute Pancreatitis (BISAP) Score
Simpler bedside tool (1 point each, measured within 24 hours):
- BUN >25 mg/dL
- Impaired mental status (GCS <15)
- SIRS (≥2 of 4 SIRS criteria)
- Age >60 years
- Pleural effusion on imaging
Score 0–2: low mortality (<1%); Score 3–5: high mortality (5–22%). BISAP is now preferred to Ranson criteria in many guidelines due to its simplicity.
CT Severity Index (CTSI) — Balthazar Score
Assessed on CECT at 48–72 hours after onset:
- Grade A: Normal pancreas (0 points)
- Grade B: Focal/diffuse enlargement (1 point)
- Grade C: Peripancreatic fat stranding (2 points)
- Grade D: Single peripancreatic fluid collection (3 points)
- Grade E: Two or more fluid collections or gas in/adjacent to pancreas (4 points)
- Necrosis score: No necrosis (0), <30% necrosis (2), 30–50% necrosis (4), >50% necrosis (6)
- CTSI = CT Grade score + Necrosis score; maximum 10 points; score ≥7 correlates with 17% mortality and 92% morbidity
Diagnosis of Chronic Pancreatitis
No single gold standard test; diagnosis relies on morphological evidence (imaging) and/or functional testing in the appropriate clinical context:
- CT abdomen: Pancreatic calcifications (pathognomonic), parenchymal atrophy, ductal dilation; sensitivity 80%, specificity 85% for advanced CP
- MRI/MRCP: Superior to CT for ductal changes, side branch disease, and early CP; secretin-enhanced MRCP improves ductal and functional assessment
- Endoscopic ultrasound (EUS): Most sensitive for early CP; Rosemont criteria use parenchymal and ductal features to classify CP likelihood; allows FNA if mass lesion suspected
- ERCP: Cambridge classification grades ductal changes (I–IV); largely replaced by MRCP for diagnosis but used therapeutically
- Fecal elastase-1: Pancreatic exocrine function marker; <200 µg/g indicates EPI; false positives with liquid stools
- 72-hour fecal fat coefficient of fat absorption: Gold standard for EPI (<93% fat absorption is abnormal); rarely performed clinically
7. Treatment
Acute Pancreatitis — Medical Management
- Aggressive IV fluid resuscitation: Goal-directed, early aggressive hydration (250–500 mL/hour in first 12–24 hours) is the cornerstone of AP management; lactated Ringer's solution (LR) is preferred over normal saline — LR reduces SIRS and organ failure risk (reduced calcium-driven acinar activation; anti-inflammatory); target urine output >0.5 mL/kg/hour, heart rate <120, mean arterial pressure >65 mmHg, and BUN decline
- Pain management: IV opioids (morphine, hydromorphone, fentanyl); NSAIDs if no contraindication; epidural analgesia for severe cases
- Nutrition: Early enteral nutrition (within 24–48 hours) is strongly recommended for severe AP and moderate AP; nasojejunal (NJ) or nasogastric (NG) feeding are equally safe and superior to parenteral nutrition (reduced infectious complications, gut barrier preservation, reduced mortality); oral feeding as tolerated in mild AP
- Antibiotics: Not recommended prophylactically in acute pancreatitis (no mortality benefit); indicated for infected necrotizing pancreatitis (CT-guided FNA with Gram stain and culture, or empirical treatment with carbapenems or fluoroquinolones + metronidazole if FNA not possible)
- ERCP: Urgent ERCP within 24–48 hours for gallstone AP with concurrent cholangitis or persistent biliary obstruction; not recommended in mild biliary AP without cholangitis
Management of Local Complications
- Peripancreatic fluid collections: Acute peripancreatic fluid collections (APFC) usually resolve spontaneously; pseudocysts (develop after 4 weeks) require intervention if symptomatic, infected, or >6 cm with failed resolution
- Walled-off necrosis (WON): Mature necrotic collection with a fibrous wall (develops after 4 weeks); infected WON treated by step-up approach: percutaneous drainage first → endoscopic ultrasound-guided transmural drainage (endoscopic necrosectomy) → minimally invasive surgical necrosectomy; open surgery reserved for failure of minimally invasive approaches
- Cholecystectomy: Recommended during index hospitalization (or within 4–6 weeks) for mild gallstone pancreatitis to prevent recurrence
Chronic Pancreatitis Management
- Pain management: Step-up analgesic ladder: non-opioids (paracetamol, NSAIDs) → weak opioids (tramadol) → strong opioids (oxycodone, morphine); adjuvant neuromodulators (pregabalin, amitriptyline); celiac plexus block (EUS-guided or CT-guided) for refractory pain; total pancreatectomy with islet autotransplantation (TPIAT) for refractory pain in selected patients without significant diabetes
- Exocrine pancreatic insufficiency: Pancreatic enzyme replacement therapy (PERT): standard dose 40,000–80,000 Ph.Eur. units lipase per main meal; taken with first bite of meal and mid-meal; pH-sensitive enteric-coated microspheres (Creon, Zenpep, Pancreaze); fat-soluble vitamin supplementation
- Diabetes management (Type 3c DM): Insulin therapy is the mainstay; metformin may be used if residual beta-cell function present and no contraindications; sulfonylureas carry hypoglycemia risk; DPP-4 inhibitors and GLP-1 agonists are under investigation; islet cell transplantation in TPIAT patients
- Endoscopic therapy: EUS-guided celiac plexus block; ERCP with ductal stenting, stone extraction (pancreatic duct stones), and stricture dilation for obstructive CP
- Extracorporeal shock wave lithotripsy (ESWL): Fragmentation of large pancreatic duct calculi before ERCP extraction; highly effective for stones >5 mm in the main pancreatic duct
- Surgical management: Drainage procedures (Puestow/Partington-Rochelle lateral pancreaticojejunostomy) for ductal hypertension and dilated duct (>6–8 mm); resection procedures (Whipple/pancreaticoduodenectomy for head-dominant disease; distal pancreatectomy for tail-dominant disease; Beger procedure, Frey procedure)
8. Complications
Acute Pancreatitis Complications
- Local: Acute peripancreatic fluid collections; pancreatic pseudocyst; acute necrotic collection; walled-off necrosis; splenic vein thrombosis; gastric outlet obstruction; colonic necrosis (rare)
- Systemic (severe AP): ARDS (most common organ failure — 20–40% of severe AP); acute kidney injury (AKI); shock; disseminated intravascular coagulation (DIC); hypocalcemia (saponification of peripancreatic fat); hyperglycemia; hemorrhage from pseudoaneurysm
- Infected necrotizing pancreatitis: Develops in 30–40% of necrotizing AP, typically in the second or third week; causative organisms include enteric gram-negatives, Enterococcus, Staphylococcus, and Candida; mortality 30%+
- Post-AP progression: Recurrent AP (17–36%); chronic pancreatitis (10–30% after first AP; up to 50% after recurrent AP); exocrine and endocrine insufficiency
Chronic Pancreatitis Complications
- Pancreatic ductal adenocarcinoma: lifetime risk 4–8% in hereditary pancreatitis (PRSS1); 1.8% in non-hereditary CP; 20-fold above general population risk at 20 years
- Biliary stricture (common bile duct compression by fibrotic pancreatic head)
- Duodenal obstruction
- Splenic vein thrombosis with gastric varices (sinistral portal hypertension)
- Pancreatic fistula and pleural effusion
- Osteoporosis and metabolic bone disease (from malabsorption and Type 3c DM)
- Nutritional deficiencies: fat-soluble vitamins, B12, zinc, selenium
- Narcotic addiction (chronic opioid use for pain management)
9. Prognosis
Approximately 80–85% of acute pancreatitis episodes are mild and self-limited, resolving within 3–7 days without local complications or organ failure. The mortality of mild AP is <1%. Moderately severe AP (transient organ failure <48 hours or local complications without persistent organ failure) carries a mortality of 1–3%. Severe AP (persistent organ failure >48 hours) has a mortality of 15–30%, rising to 30–40% in infected necrotizing pancreatitis, particularly when multiple organ systems are involved.
The prognosis of chronic pancreatitis is significantly affected by etiology. Continued alcohol consumption and tobacco use substantially worsen outcomes. Life expectancy is reduced by 10–20 years compared to age-matched controls. The 10-year survival rate is approximately 70%, with leading causes of death being cardiovascular disease, malnutrition, diabetes-related complications, pancreatic cancer, and alcohol-related multi-organ disease.
Autoimmune pancreatitis (Type 1 AIP) has an excellent prognosis with steroid therapy (prednisone 40 mg/day tapering over 3–4 months) — response rates >95% — but relapse occurs in 30–40%.
10. Prevention
- Gallstone disease management: Early cholecystectomy after mild gallstone pancreatitis (ideally same admission or within 4 weeks) is the most impactful preventive strategy; reduces recurrence risk from 30–60% to <5%
- Alcohol cessation: Complete abstinence is the single most important modifiable risk factor for preventing progression from acute to chronic pancreatitis; reduces pain frequency and rate of endocrine/exocrine insufficiency
- Tobacco cessation: Independent risk factor for both AP and CP progression; cessation reduces CP risk and may slow progression
- Hypertriglyceridemia management: Triglycerides >1,000 mg/dL require urgent reduction with insulin infusion (AP setting), fibrates, omega-3 fatty acids, and dietary fat restriction; target TG <500 mg/dL to prevent recurrence
- Post-ERCP pancreatitis prevention: Rectal indomethacin 100 mg (PR) immediately before or after ERCP in all patients (number needed to treat ~20); prophylactic pancreatic duct stenting in high-risk patients; adequate hydration with LR
- NSAID-sparing strategies: Avoid NSAIDs in patients with history of AP when possible
- Genetic counseling: Recommended for hereditary pancreatitis (PRSS1 mutations) and patients with identified genetic risk factors (SPINK1, CFTR, CTRC)
11. Recent Research and Advances
- Aggressive vs. non-aggressive fluid resuscitation: The WATERFALL trial (NEJM, 2022) demonstrated that aggressive IV hydration with LR (20 mL/kg bolus then 3 mL/kg/hour) did not improve outcomes compared to moderate hydration (10 mL/kg bolus then 1.5 mL/kg/hour) and was associated with increased fluid overload-related complications (ARDS, compartment syndrome). Current guidelines recommend moderate, goal-directed fluid resuscitation.
- Rectal indomethacin prophylaxis for post-ERCP pancreatitis: The landmark APAT trial (2020) confirmed routine rectal indomethacin for all patients undergoing ERCP reduces post-ERCP pancreatitis from 4.4% to 2.1%; now universally recommended regardless of individual risk stratification.
- Step-up approach for necrotizing pancreatitis: Long-term follow-up of the PANTER trial has validated the minimally invasive step-up approach (percutaneous drainage → video-assisted retroperitoneal debridement (VARD) → laparotomy only if needed) as equivalent in efficacy to open necrosectomy with significantly fewer complications.
- Endoscopic transmural drainage vs. percutaneous drainage for WON: Multiple RCTs and the TENSION trial demonstrated superiority of endoscopic ultrasound-guided transmural drainage for infected WON over percutaneous drainage in pancreatic protocol-eligible patients (reduced need for surgery, shorter hospital stay, lower mortality).
- TPIAT (Total Pancreatectomy with Islet Autotransplantation): Expanding indications for TPIAT in patients with hereditary pancreatitis and refractory pain before advanced fibrosis; long-term data show 40–60% insulin independence at 5 years in pediatric patients; adult outcomes less favorable.
- Acinar cell biomarkers: Serum CXCL10, IL-6, and angiopoietin-2 are under investigation as early predictors of severity in AP, potentially outperforming BISAP and Ranson criteria in prospective validation studies.
- Microbiome in AP: The gut microbiome undergoes rapid dysbiosis in severe AP, contributing to bacterial translocation and infected necrosis; selective intestinal decontamination and specific probiotic protocols are under investigation (following the negative outcomes of the PROPATRIA probiotic trial in severe AP in 2008, which increased mortality).
12. References
- Banks PA, Bollen TL, Dervenis C, et al. Classification of acute pancreatitis — 2012: revision of the Atlanta classification and definitions by international consensus. Gut. 2013;62(1):102–111.
- Leppäniemi A, Tolonen M, Tarasconi A, et al. 2019 WSES guidelines for the management of severe acute pancreatitis. World J Emerg Surg. 2019;14:27.
- Crockett SD, Wani S, Gardner TB, et al. American Gastroenterological Association Institute Guideline on Initial Management of Acute Pancreatitis. Gastroenterology. 2018;154(4):1096–1101.
- de-Madaria E, Buxbaum JL, Maisonneuve P, et al. Aggressive or Moderate Fluid Resuscitation in Acute Pancreatitis. N Engl J Med. 2022;387(11):989–1000.
- Besselink MG, van Santvoort HC, Nieuwenhuijs VB, et al. Minimally invasive 'step-up approach' versus maximal necrosectomy in patients with acute necrotising pancreatitis (PANTER trial). Lancet. 2010;375(9711):294.
- van Brunschot S, van Grinsven J, van Santvoort HC, et al. Endoscopic or surgical step-up approach for infected necrotising pancreatitis: a multicentre randomised trial. Lancet. 2018;391(10115):51–58.
- Elta GH, Enestvedt BK, Sauer BG, Lennon AM. ACG Clinical Guideline: Diagnosis and Management of Pancreatic Cysts. Am J Gastroenterol. 2018;113(4):464–479.
- Kleeff J, Whitcomb DC, Shimosegawa T, et al. Chronic pancreatitis. Nat Rev Dis Primers. 2017;3:17060.
- Löhr JM, Dominguez-Munoz E, Rosendahl J, et al. United European Gastroenterology evidence-based guidelines for the diagnosis and therapy of chronic pancreatitis (HaPanEU). United European Gastroenterol J. 2017;5(2):153–199.
- Yadav D, Whitcomb DC. The role of alcohol and smoking in pancreatitis. Nat Rev Gastroenterol Hepatol. 2010;7(3):131–145.
- Whitcomb DC, Frulloni L, Garg P, et al. Chronic pancreatitis: An international draft consensus proposal for a new mechanistic definition. Pancreatology. 2016;16(2):218–224.
- Dumonceau JM, Kapral C, Aabakken L, et al. ERCP-related adverse events: European Society of Gastrointestinal Endoscopy (ESGE) Guideline. Endoscopy. 2020;52(2):127–149.
- Balthazar EJ, Robinson DL, Megibow AJ, Ranson JH. Acute pancreatitis: value of CT in establishing prognosis. Radiology. 1990;174(2):331–336.
- Ranson JH, Rifkind KM, Roses DF, Fink SD, Eng K, Spencer FC. Prognostic signs and the role of operative management in acute pancreatitis. Surg Gynecol Obstet. 1974;139(1):69–81. PMID: 4834279
- Singh VK, Wu BU, Bollen TL, et al. A prospective evaluation of the bedside index for severity in acute pancreatitis score in assessing mortality and intermediate markers of severity in acute pancreatitis. Am J Gastroenterol. 2009;104(4):966–971.
- Gardner TB, Vege SS, Chari ST, et al. Faster rate of initial fluid resuscitation in severe acute pancreatitis diminishes in-hospital mortality. Pancreatology. 2009;9(6):770–776.
- Drewes AM, Bouwense SAW, Campbell CM, et al. Guidelines for the understanding and management of pain in chronic pancreatitis. Pancreatology. 2017;17(5):720–731.
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Pancreatitis. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Acute pancreatitis management
- PubMed: Chronic pancreatitis treatment
- PubMed: Gallstone pancreatitis
- PubMed: Alcoholic pancreatitis
- PubMed: Severity scoring
- PubMed: ERCP in pancreatitis
- PubMed: Enzyme replacement therapy
- PubMed: Hypertriglyceridemic pancreatitis
- PubMed: Necrotizing pancreatitis
- PubMed: Autoimmune pancreatitis
- PubMed: Pain management
- PubMed: Enteral nutrition
Connections
- Gallbladder Disease
- SIBO
- Diabetes
- Turmeric
- Milk Thistle
- Magnesium
- Vitamin D3
- Gut Healing
- Abdominal Pain
- Nausea and Vomiting
- Jaundice
- Hypertension
- Obesity
- Calcium
- Zinc
- Celiac Disease
- Crohn's Disease
- GERD
Featured Videos

Doctor explains Acute Pancreatitis | Causes, Symptoms, Treatment

Pancreatitis - Symptoms and Diagnosis

Chronic pancreatitis - causes, symptoms, diagnosis, treatment, pathology
Acute Pancreatitis: Causes, Symptoms & Treatment | Dr. Neeraj Goel
Acute Pancreatitis - Overview (signs and symptoms, pathophysiology, investigations, treatment)

Pancreatitis | Acute and Chronic Pancreatitis Nursing Lecture Symptoms, Treatment, Pathophysiology

What symptoms did you experience that lead to your diagnosis of acute pancreatitis?

Pancreatitis | 9 Tips to Help with Pancreatitis Naturally | Dr. J9 Live
Understanding Pancreatitis