Cellulitis
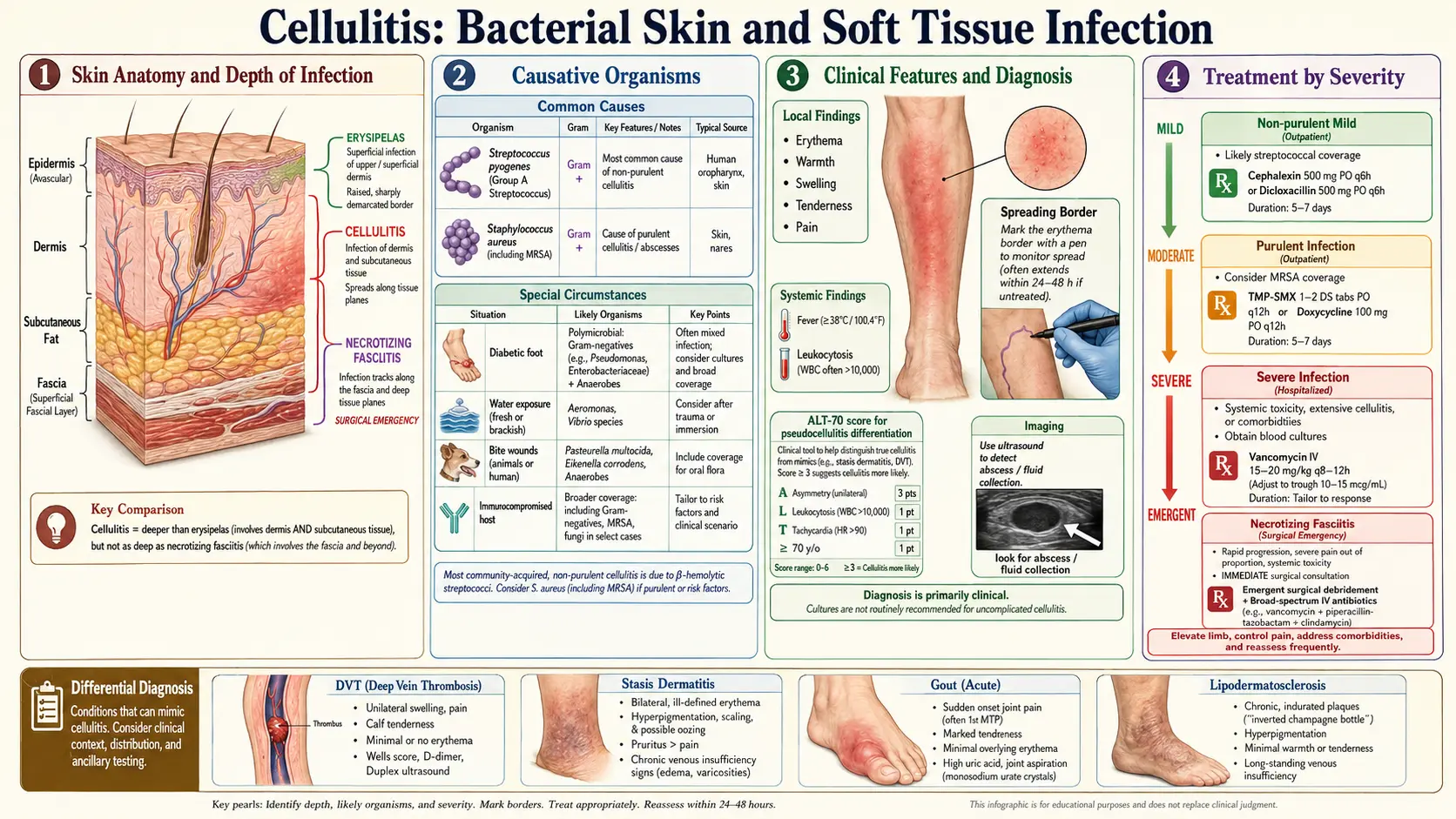
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Cellulitis is an acute bacterial infection of the dermis and subcutaneous tissues characterized by spreading erythema, warmth, edema, and tenderness. It is one of the most common bacterial skin infections encountered in both primary care and emergency medicine settings. The term "cellulitis" should be distinguished from erysipelas, which involves the more superficial dermis and upper subcutaneous tissue and characteristically has a sharply demarcated, raised, and indurated border — though both conditions exist on a spectrum of non-purulent skin and soft tissue infection (SSTI), are predominantly caused by streptococcal species, and are treated similarly.
Cellulitis must also be differentiated from necrotizing fasciitis — a deep, rapidly progressive, life-threatening infection of the fascial planes requiring emergent surgical debridement. Failure to distinguish necrotizing fasciitis from simple cellulitis can be fatal; any features of systemic toxicity, pain out of proportion to visible findings, skin necrosis, crepitus, or failure to respond to IV antibiotics should prompt urgent surgical consultation and imaging.
Approximately 14–24 million episodes of cellulitis occur annually in the United States, making it the most common indication for hospital admission among SSTIs and a leading cause of antibiotic prescribing.
2. Epidemiology
- Incidence: Cellulitis accounts for approximately 14.5 million visits to healthcare providers annually in the US, with approximately 700,000 hospitalizations per year. It represents the third most common reason for Emergency Department antibiotic prescriptions.
- Healthcare burden: Annual direct medical costs exceed $3.7 billion in the US. Average hospital stay approximately 3–5 days for uncomplicated cellulitis.
- Demographics: Incidence increases with age; higher in men than women; highest in lower-income populations with limited access to care. The lower extremity (predominantly the leg) is affected in 70–80% of cases.
- Recurrence: A significant clinical problem — cumulative recurrence rates of approximately 8–20% within 1 year and 30–50% within 3 years. Lymphedema, chronic venous insufficiency, and obesity are the strongest predictors of recurrence.
- Misdiagnosis: Studies from tertiary centers indicate that approximately 30% of "cellulitis" diagnoses in ED and inpatient settings are incorrect — pseudocellulitis conditions (stasis dermatitis, lipodermatosclerosis, contact dermatitis, DVT, gout, papular urticaria) are misdiagnosed as cellulitis, leading to unnecessary hospitalization and antibiotic use. Clinicians should maintain a high index of suspicion for non-infectious mimics, particularly in bilateral lower extremity presentations.
- Healthcare-associated (nosocomial) cellulitis: Occurs at surgical sites, IV catheter sites, and drain sites; more likely to involve gram-negative or resistant organisms.
3. Pathophysiology
Cellulitis typically results from disruption of the normal epidermal barrier, allowing commensal or environmental bacteria to invade the dermis and subcutaneous tissues. Several pathogenic mechanisms operate:
Portal of Entry and Bacterial Invasion
Common portals include: tinea pedis with interdigital maceration (the most important and commonly overlooked risk factor for lower-leg cellulitis, present in up to 70% of cases), minor traumatic wounds (abrasions, lacerations, punctures, insect bites), chronic skin conditions (eczema, psoriasis), surgical wounds, skin ulcers (venous, diabetic, pressure), lymphedema, and percutaneous catheters. In many cases, no obvious portal is identified at presentation.
Streptococcal species produce multiple virulence factors that facilitate tissue invasion: streptokinase (fibrinolysin), hyaluronidase ("spreading factor" — degrades the extracellular matrix barrier), DNase, and M-protein–mediated resistance to phagocytosis. M-protein elicits potent inflammatory responses and, in certain serotypes, drives immunologic complications (post-streptococcal glomerulonephritis, rheumatic fever).
Staphylococcus aureus produces protein A (inhibits opsonization), coagulase, leukotoxins (Panton-Valentine leukocidin, PVL — associated with tissue necrosis in MRSA strains), and hyaluronidase, enabling both local tissue destruction and systemic dissemination.
Inflammatory Response
Bacterial PAMPs (cell wall components, exotoxins, flagellin) activate skin-resident innate immune cells — Langerhans cells, dermal dendritic cells, keratinocytes, and mast cells — via TLRs, triggering chemokine/cytokine release (IL-8, IL-6, TNF-α). Neutrophil recruitment generates the characteristic inflammatory exudate of erythema, heat, swelling, and pain (calor, rubor, tumor, dolor). In most cases, local innate immunity contains the infection; systemic bacteremia is relatively uncommon (<2% of uncomplicated cellulitis) but can occur with virulent organisms or compromised host defenses.
Lymphatic Involvement and Recurrence
Repeated episodes of cellulitis damage local lymphatics, impairing lymphatic drainage and creating progressive lymphedema — which paradoxically increases susceptibility to further cellulitis episodes, establishing a vicious cycle. This mechanism underlies the high recurrence rates in patients with chronic lymphedema and explains why prophylactic antibiotics targeting lymphatic-tropic Streptococcus are effective in preventing recurrence.
4. Etiology and Risk Factors
Causative Organisms
Non-purulent cellulitis (no abscess or purulent drainage) is caused predominantly by:
- Beta-hemolytic streptococci: Group A Streptococcus pyogenes (most common), Group B (S. agalactiae — particularly in neonates, elderly, and immunocompromised), Group C and G streptococci.
- Staphylococcus aureus: Less commonly causes non-purulent cellulitis; more commonly associated with purulent SSTIs (furunculosis, carbuncles, abscesses).
Special circumstances:
- Immunocompromised patients: Gram-negative organisms (E. coli, Pseudomonas aeruginosa, Acinetobacter), atypical organisms (Stenotrophomonas, Cryptococcus).
- Lymphedema/elephantiasis: Non-group A streptococcal species.
- Freshwater exposure: Aeromonas hydrophila — particularly in penetrating injuries near rivers/lakes; often rapidly progressive.
- Saltwater/seafood exposure: Vibrio vulnificus — particularly dangerous in liver disease patients; can progress to necrotizing fasciitis; mortality up to 25%.
- Animal bites (dog/cat): Pasteurella multocida (cat bites especially; rapid progression), Capnocytophaga canimorsus (dog bites, can cause sepsis in asplenic patients), Staphylococcus, streptococci, anaerobes.
- Human bites: Eikenella corrodens, streptococci, anaerobes (Fusobacterium, Bacteroides).
- Periorbital (preseptal) cellulitis: S. aureus, S. pyogenes, Streptococcus pneumoniae, H. influenzae.
- Orbital cellulitis: Polymicrobial; often secondary to sinusitis; S. aureus, Streptococcus spp., anaerobes.
- Diabetic foot cellulitis: Often polymicrobial with gram-negative rods and anaerobes in addition to staphylococci and streptococci.
Host Risk Factors
- Lymphedema (chronic or secondary to cancer treatment, filariasis)
- Chronic venous insufficiency, varicose veins
- Obesity (BMI >30)
- Diabetes mellitus (impaired neutrophil function, peripheral neuropathy, vascular disease)
- Immunosuppression (HIV, corticosteroids, chemotherapy, organ transplant)
- Tinea pedis / interdigital fungal infection (disrupts skin barrier)
- Prior cellulitis (strongest predictor of recurrence)
- Peripheral vascular disease, lower extremity edema
- Intravenous drug use
- Skin trauma, ulcers, surgical wounds
5. Clinical Presentation
Classic Presentation
Typical lower-extremity cellulitis presents with an acute onset of spreading erythema, warmth, swelling (edema), and tenderness — classically beginning near a portal of entry and expanding over hours to days. The advancing border is typically not sharply demarcated (distinguishing it from erysipelas, which has a raised, sharply defined border). The skin surface may have a "peau d'orange" (orange peel) texture from lymphatic obstruction. Systemic symptoms — fever, chills, malaise, tachycardia — are variably present and more pronounced with Group A streptococcal cellulitis or in immunocompromised patients.
Classification by IDSA Severity
- Class I (mild): No systemic features; healthy host; suitable for outpatient oral antibiotics.
- Class II (moderate): Systemically ill (fever, tachycardia, hypotension) but no severe immunosuppression or critical illness; may require IV antibiotics but can be managed on medical ward.
- Class III (severe): Markedly systemically ill; rapidly progressing infection; immunocompromise; failure to respond to oral antibiotics; requires IV antibiotics with close monitoring; consider ICU if septic.
- Class IV (critical): Signs suggesting necrotizing infection (pain out of proportion, crepitus, bullae, skin necrosis, septic shock); surgical emergency.
Special Clinical Presentations
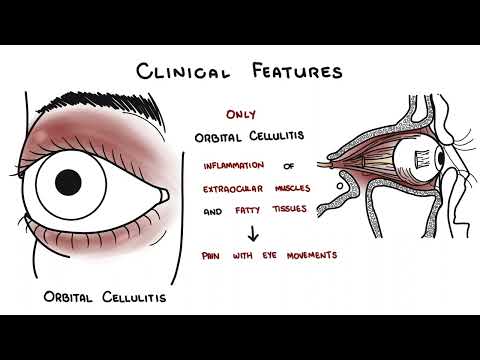
Periorbital (preseptal) cellulitis: Eyelid erythema, edema, tenderness without proptosis, pain with eye movement, or diplopia — confined anterior to the orbital septum; generally manageable with oral or IV antibiotics without surgery.
Orbital cellulitis: Posterior to orbital septum; presents with proptosis, ophthalmoplegia, pain with eye movement, chemosis, reduced visual acuity. Risk of intracranial extension (cavernous sinus thrombosis, meningitis, subdural empyema). Requires urgent CT orbits with contrast, ophthalmology and ENT consultation, IV antibiotics, and often surgical drainage.
Facial/buccal cellulitis: Spreading erythema and swelling of the cheek; may arise from dental source, sinusitis, or trauma; risk of odontogenic origin requires dental evaluation.
Perianal cellulitis (perianal streptococcal dermatitis): Especially in children; bright red, well-defined perianal erythema, pruritus, pain; caused by Group A streptococcal infection; treated with oral penicillin or amoxicillin.
6. Diagnosis
Cellulitis is a clinical diagnosis based on history and physical examination. There is no confirmatory laboratory test. The clinician must: (1) confirm that the presentation is consistent with cellulitis, (2) exclude mimicking conditions ("pseudocellulitis"), and (3) identify features that may indicate necrotizing fasciitis or other emergent diagnoses.
Laboratory Studies
- Complete blood count: Leukocytosis with neutrophilia in moderate-severe cellulitis; may be normal in mild cases. Leukopenia suggests severe immunosuppression or viral etiology.
- CRP and ESR: Elevated; useful for monitoring treatment response but non-specific for diagnosis.
- Blood cultures: Yield is low (<2–4% positivity) in uncomplicated cellulitis; recommended in immunocompromised patients, signs of sepsis, failure to improve on antibiotics, or suspicion of bacteremia. Not routinely indicated in uncomplicated community-acquired cellulitis.
- Wound/swab cultures: Not useful for non-purulent cellulitis (surface swabs yield colonizers, not pathogens). Needle aspiration of advancing border — sensitivity approximately 10–15%; not routinely recommended. Biopsy cultures may be warranted in atypical or refractory cases.
- Creatinine, liver function, glucose: Baseline for antibiotic dosing and to identify diabetes, renal/hepatic impairment.
- D-dimer/duplex ultrasound: If DVT is in the differential diagnosis (common mimicker; unilateral lower extremity swelling with erythema).
Imaging
- MRI with gadolinium: Most sensitive imaging modality for distinguishing cellulitis from necrotizing fasciitis (key finding: deep fascial enhancement and fat stranding in NF); also demonstrates orbital/periorbital disease extent.
- CT with IV contrast: Faster than MRI; identifies gas in tissue (crepitus) — pathognomonic for gas-forming infections including type 1 necrotizing fasciitis; preferred for initial evaluation when NF is suspected and MRI is not immediately available.
- Plain radiographs: May show subcutaneous gas in gas-forming infections; less sensitive than CT.
- Ultrasound: Can identify coexisting abscess (hypoechoic collection, compressibility, cobblestone sign) — important to identify since abscesses require incision and drainage, not just antibiotics.
LRINEC Score (Laboratory Risk Indicator for Necrotizing Fasciitis)
A scoring system incorporating CRP, WBC, hemoglobin, sodium, creatinine, and glucose to stratify risk for necrotizing fasciitis. Score ≥6 warrants urgent surgical evaluation; score ≥8 associated with >75% probability of NF. However, LRINEC has limited sensitivity (~70%) and should not substitute for clinical judgment or imaging when clinical suspicion for NF is high.
7. Treatment
Non-Purulent Cellulitis (Streptococcal-Predominant)
Mild (Class I) — Outpatient oral therapy:
- Cephalexin 500 mg PO four times daily × 5–7 days — first-line.
- Dicloxacillin 500 mg PO four times daily × 5–7 days — alternative.
- Amoxicillin-clavulanate 875/125 mg PO twice daily × 5–7 days — for animal bites or polymicrobial risk.
- Clindamycin 300–450 mg PO three times daily × 5–7 days — for penicillin allergy (verify susceptibility; up to 30% clindamycin resistance in MRSA).
- TMP-SMX is NOT recommended for non-purulent cellulitis — poor streptococcal activity demonstrated in randomized trials (Stevens et al., N Engl J Med 2017).
Moderate-Severe (Classes II–III) — Inpatient IV therapy:
- Cefazolin 1–2 g IV every 8 hours — first-line.
- Oxacillin 1–2 g IV every 4 hours — alternative.
- Nafcillin 1–2 g IV every 4 hours — alternative.
- Clindamycin 600–900 mg IV every 8 hours — for significant penicillin allergy.
- Vancomycin (target AUC/MIC 400–600) — add if MRSA is suspected (healthcare-associated, prior MRSA colonization, penetrating trauma, failure to respond to beta-lactams) or for septic patients pending cultures.
MRSA-directed therapy (if purulent or MRSA-suspected):
- Outpatient: TMP-SMX 1–2 DS tablets PO twice daily × 5–7 days; doxycycline 100 mg PO twice daily × 5–7 days.
- Inpatient: Vancomycin IV; daptomycin 4–6 mg/kg IV daily; linezolid 600 mg IV/PO twice daily; ceftaroline 600 mg IV every 8–12 hours (broad-spectrum cephalosporin with MRSA activity).
Supportive measures: Elevation of affected extremity above heart level (reduces edema and pain); warm compresses; outline of erythema border with skin-safe marker for monitoring progression; adequate analgesia (NSAIDs for pain and anti-inflammatory benefit; acetaminophen).
Treatment of Special Circumstances
Orbital cellulitis: Broad-spectrum IV antibiotics (ampicillin-sulbactam, ceftriaxone ± metronidazole); ENT and ophthalmology; CT scan; surgical drainage if abscess, optic nerve compromise, or failure to respond within 24–48 hours.
Diabetic foot infection: Polymicrobial coverage (piperacillin-tazobactam, ampicillin-sulbactam, or carbapenems for severe/limb-threatening); wound care; vascular assessment; surgical debridement as needed.
Animal bites: Amoxicillin-clavulanate; wound irrigation; tetanus prophylaxis; rabies post-exposure prophylaxis assessment.
Duration of Therapy
Typically 5–7 days for uncomplicated cellulitis; extend to 10–14 days if slow response or large area involved. Stepdown from IV to oral therapy when systemic signs resolve and patient tolerating oral medications.
8. Complications
- Necrotizing fasciitis: Most feared complication; may begin as apparent cellulitis; constitutes a surgical emergency. Type I (polymicrobial) and Type II (Group A streptococcal, "flesh-eating" disease) necrotizing fasciitis carry mortality of 20–40% even with surgery.
- Bacteremia and sepsis: Relatively rare (<2–4% of non-purulent cellulitis) but life-threatening; risk higher with immunosuppression, rapidly progressing infection, Group A streptococcal disease.
- Osteomyelitis: Contiguous spread to underlying bone, particularly in diabetic foot infections with peripheral neuropathy; diagnosis by MRI.
- Septic arthritis: Spread to adjacent joint, particularly elbow, knee; urgent joint aspiration required.
- Lymphangitis: Red linear streaking proximally along lymphatic vessels; indicates lymphatic spread and systemic dissemination risk.
- Lymphedema: Permanent lymphatic injury from recurrent cellulitis; perpetuates cycle of recurrence.
- Antibiotic adverse effects: C. difficile colitis (fluoroquinolones, clindamycin highest risk); drug rash; nephrotoxicity (vancomycin); bone marrow suppression (linezolid with prolonged use).
- Orbital cellulitis complications: Cavernous sinus thrombosis, intracranial abscess, meningitis, vision loss.
- Recurrent cellulitis: Chronic lymphedema, functional impairment, repeated healthcare utilization.
9. Prognosis
The prognosis of uncomplicated cellulitis treated promptly with appropriate antibiotics is generally excellent:
- Clinical response: Most patients show improvement within 24–72 hours of appropriate antibiotics. Failure to improve within 48–72 hours should prompt reassessment of diagnosis (is this truly cellulitis?), antibiotics (adequate spectrum, dose, route), and surgical evaluation for abscess or necrotizing infection.
- Outpatient cure rates: Approximately 85–95% for Class I cellulitis with appropriate oral antibiotics.
- Hospitalization rates: Approximately 30–40% of ED cellulitis patients require hospitalization for IV antibiotics.
- Recurrence: As noted, 30–50% 3-year cumulative recurrence rate, particularly in patients with lymphedema, obesity, or chronic venous insufficiency. Prophylactic antibiotics significantly reduce recurrence.
- Prognostic factors for poor outcome: Immunosuppression, diabetes, peripheral vascular disease, delay in treatment, presence of necrotizing component, bacteremia, elderly age.
- Necrotizing fasciitis: Mortality 20–40%; extremity amputation may be required; outcomes dependent on speed of surgical intervention.
10. Prevention
- Treat tinea pedis and interdigital maceration: Most important preventive intervention for lower-leg recurrent cellulitis; topical antifungals (clotrimazole, terbinafine) cure active infection; miconazole powder for prevention of moisture/maceration.
- Wound care: Prompt and thorough cleaning of all skin wounds with soap, water, and antiseptic; appropriate wound closure; timely debridement of necrotic tissue.
- Compression therapy: Medical-grade compression stockings for chronic venous insufficiency and lymphedema reduce edema, improving lymphatic function and skin barrier integrity.
- Moisturization: Regular emollient application to skin affected by eczema, xerosis, or stasis dermatitis to restore epidermal barrier.
- Limb hygiene: Meticulous skin care in patients with lymphedema; lymphedema massage (manual lymphatic drainage); avoid trauma and constriction of affected limbs.
- Prophylactic antibiotics: For patients with ≥3–4 episodes of cellulitis per year: benzathine penicillin G 1.2 million units IM every 2–4 weeks OR oral phenoxymethylpenicillin (penicillin V) 250 mg twice daily. The UK PATCH trial demonstrated penicillin prophylaxis reduces recurrence rate by approximately 47%. Continue for at least 6 months; many patients require indefinite suppressive therapy while underlying risk factors persist.
- Weight management: Obesity reduction reduces lymphedema and susceptibility to cellulitis.
- Glycemic control: Optimizing HbA1c in diabetic patients reduces infection susceptibility and wound healing impairment.
11. Recent Research and Advances
- PATCH trials: UK-based multicenter trials demonstrating efficacy of prophylactic low-dose penicillin (250 mg twice daily) in reducing cellulitis recurrence; updated PATCH III examining duration of prophylaxis and patient selection criteria.
- Cellulitis misdiagnosis and stewardship: Systematic implementation of cellulitis diagnostic criteria (IDSA classification, psoriasis/DVT/stasis dermatitis exclusion) in academic centers has reduced unnecessary inpatient admissions by 15–30% and antibiotic use (Thomas et al., JAMA Intern Med 2022).
- Outpatient parenteral antimicrobial therapy (OPAT): Increasing use of OPAT and hospital-in-the-home programs for Class II–III cellulitis; non-inferior outcomes to inpatient care at substantially lower cost.
- SSTI microbiome: 16S rRNA and metagenomic sequencing studies revealing the complex polymicrobial ecology of skin infections; implications for empiric therapy and diagnostics.
- Novel diagnostics: Lateral flow assays for rapid streptococcal antigen detection from skin swabs; PCR-based multiplex panels for wound pathogens — under clinical validation.
- Biomarkers for NF vs. cellulitis discrimination: Serum CK (elevated in NF due to muscle involvement), lactate, IL-6, and PCT in combination have better discriminatory performance than LRINEC alone for detecting necrotizing infections.
- Anti-inflammatory adjuncts: Small trials examining NSAIDs, systemic corticosteroids, and doxycycline (anti-inflammatory properties) as adjuncts for reducing inflammatory swelling and accelerating resolution — inconclusive results; not standard of care.
12. References
- Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the IDSA. Clin Infect Dis. 2014;59:e10–e52.
- Moran GJ, Abrahamian FM, Lovecchio F, et al. Acute bacterial skin infections: developments since the 2005 IDSA guidelines. J Emerg Med. 2013;44:e397–e412.
- Drekonja DM, Trautner B, Amula K, et al. Effect of 5 vs 10 days of antibiotic therapy for urinary tract infection in outpatient women. JAMA Intern Med. 2020. [Cellulitis duration comparator referenced in SSTI guidelines]
- Baddour LM, Bisno AL. Recurrent cellulitis after coronary bypass surgery. Association with superficial fungal infection in saphenous venectomy limbs. JAMA. 1984;251:1049–1052.
- Thomas KS, Crook AM, Nunn AJ, et al. Penicillin to prevent recurrent leg cellulitis (PATCH I and PATCH II). N Engl J Med. 2013;368:1695–1703.
- Stevens DL, Bryant AE. Impetigo, erysipelas and cellulitis. In: Ferretti JJ, Stevens DL, Fischetti VA, eds. Streptococcus pyogenes: Basic Biology to Clinical Manifestations. University of Oklahoma Health Sciences Center, 2016. https://www.ncbi.nlm.nih.gov/books/NBK333408/
- Weng QY, Raff AB, Cohen JM, et al. Costs and consequences associated with misdiagnosed lower extremity cellulitis. JAMA Dermatol. 2017;153:141–146.
- Eron LJ, Lipsky BA, Low DE, et al. Managing skin and soft tissue infections: expert panel recommendations on key decision points. J Antimicrob Chemother. 2003;52(Suppl 1):i3–i17.
- Raff AB, Kroshinsky D. Cellulitis: a review. JAMA. 2016;316:325–337.
- Wong CH, Khin LW, Heng KS, et al. The LRINEC (Laboratory Risk Indicator for Necrotizing Fasciitis) score: a tool for distinguishing necrotizing fasciitis from other soft tissue infections. Crit Care Med. 2004;32:1535–1541.
- Swartz MN. Clinical practice. Cellulitis. N Engl J Med. 2004;350:904–912.
- Miller LG, Daum RS, Creech CB, et al. Clindamycin versus trimethoprim-sulfamethoxazole for uncomplicated skin infections. N Engl J Med. 2015;372:1093–1103.
- Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department (EMERGEncy ID Net). N Engl J Med. 2006;355:666–674.
- Lipsky BA, Berendt AR, Cornia PB, et al. 2012 IDSA clinical practice guideline for the diagnosis and treatment of diabetic foot infections. Clin Infect Dis. 2012;54:e132–e173.
- Skin and Soft Tissue Infections. In: Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases. 9th ed. Elsevier, 2020. Chapter 95.
Research Papers
The following PubMed topic searches retrieve current peer-reviewed literature on Cellulitis. Each link opens a live PubMed query so you always see the most recent publications.
- Cellulitis diagnosis and management review
- Cellulitis randomized controlled trial antibiotic
- Erysipelas versus cellulitis clinical features
- Cellulitis Staphylococcus aureus
- Cellulitis Streptococcus pyogenes
- Recurrent cellulitis prophylaxis
- Cellulitis lymphedema risk factor
- Necrotizing fasciitis diagnosis
- Skin soft tissue infection IDSA guideline
- Cellulitis MRSA treatment
- Cellulitis misdiagnosis pseudocellulitis
Connections
- Sepsis
- Staphylococcus Aureus
- Diabetes
- Garlic
- Manuka Honey
- Turmeric
- Vitamin C
- Silver Nanoparticles
- Edema
- Meningitis
- Obesity
- Eczema
- Psoriasis
- Complete Blood Count
- Pseudomonas Aeruginosa
- Creatinine
- Clostridium Difficile
- Immune Boosting
Featured Videos
Orbital VS Preorbital Cellulitis

Cellulitis & Wounds: What We Need to Know - George Perdrizet, MD, PhD - Patient Symposium 2019

Cellulitis Made Simple

Understand Cellulitis in Cancer Patients: What You Need to Know to Stay Safe

Cellulitis

Balat Basics ni Doktora Derma: May Cellulitis si Cesar (by Dr Cecile Clemente-Ocampo

From Cellulitis to Necrotizing Fasciitis | EM & Acute Care Course

CELLULITIS: Everything You Need To Know! Symptoms. Cause. Risk Factors. Treatment. Prevention.
How to Treat Cellulitis (Common Mistakes to Avoid)
Cellulitis vs Erysipelas | Bacterial Causes, Risk Factors, Signs and Symptoms, Treatment

3 Ways to Stop Recurring Cellulitis and Skin Infections Naturally
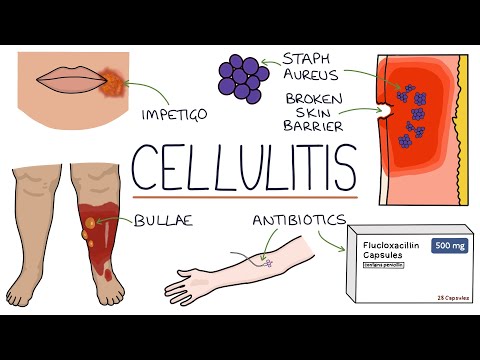
Understanding Cellulitis: Skin and Soft Tissue Infections
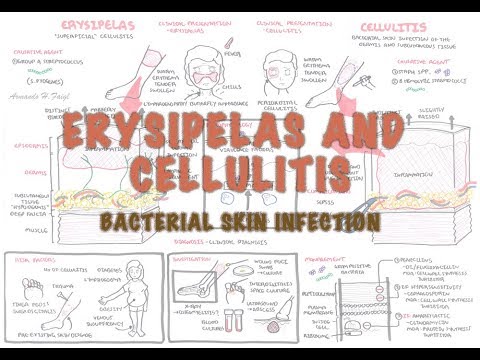
Bacterial Skin Infection - Cellulitis and Erysipelas (Clinical Presentation, Pathology, Treatment)

What is Cellulitis & its symptoms? By Dr. Akhilesh Kumar | Apollo Hospitals

Cellulitis - Tips for Care and Prevention (Tamil)

Is this Cellulitis? 8 Clinical Scenarios | USMLE Step 2 CK Success #8