Sepsis
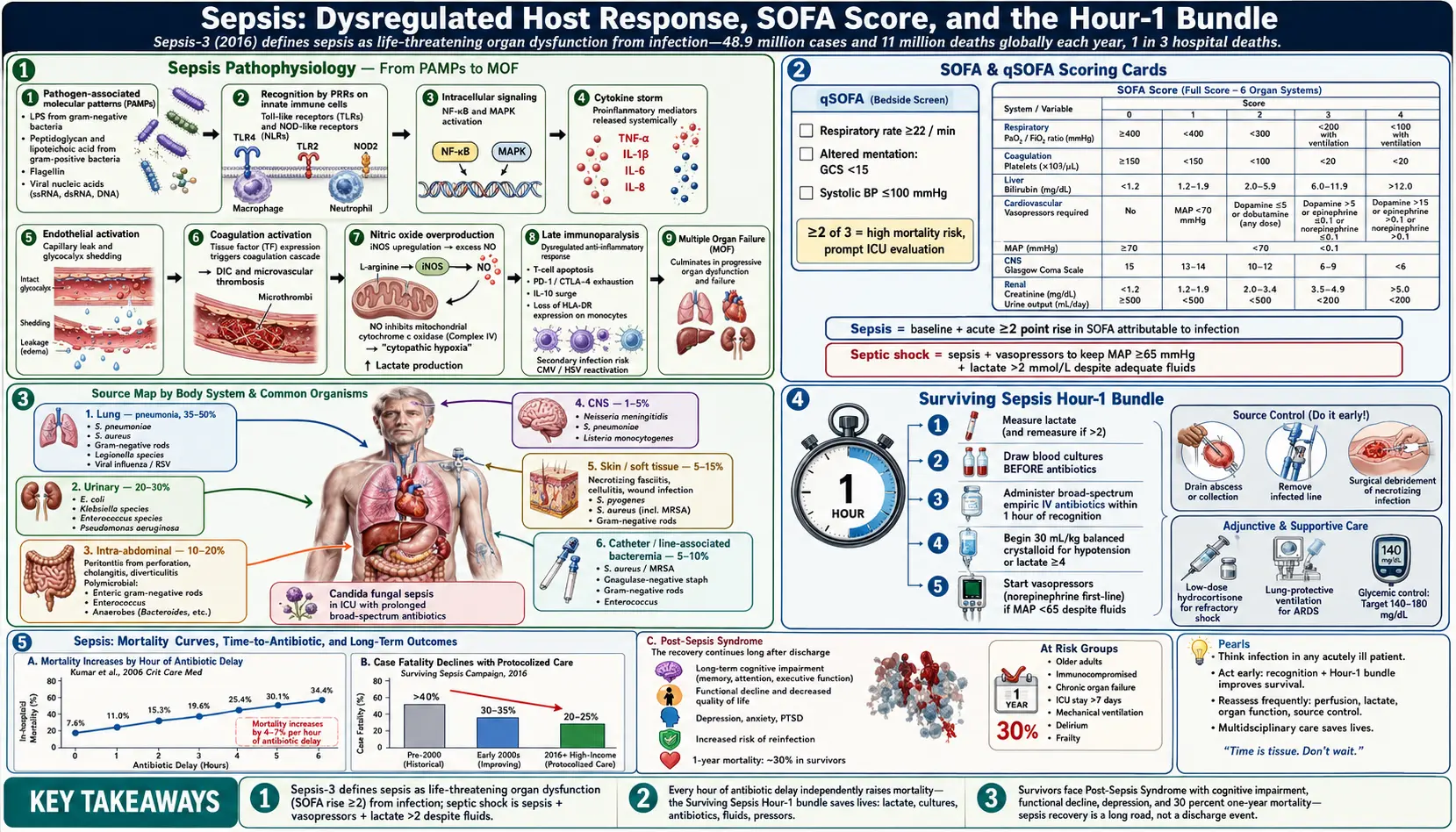
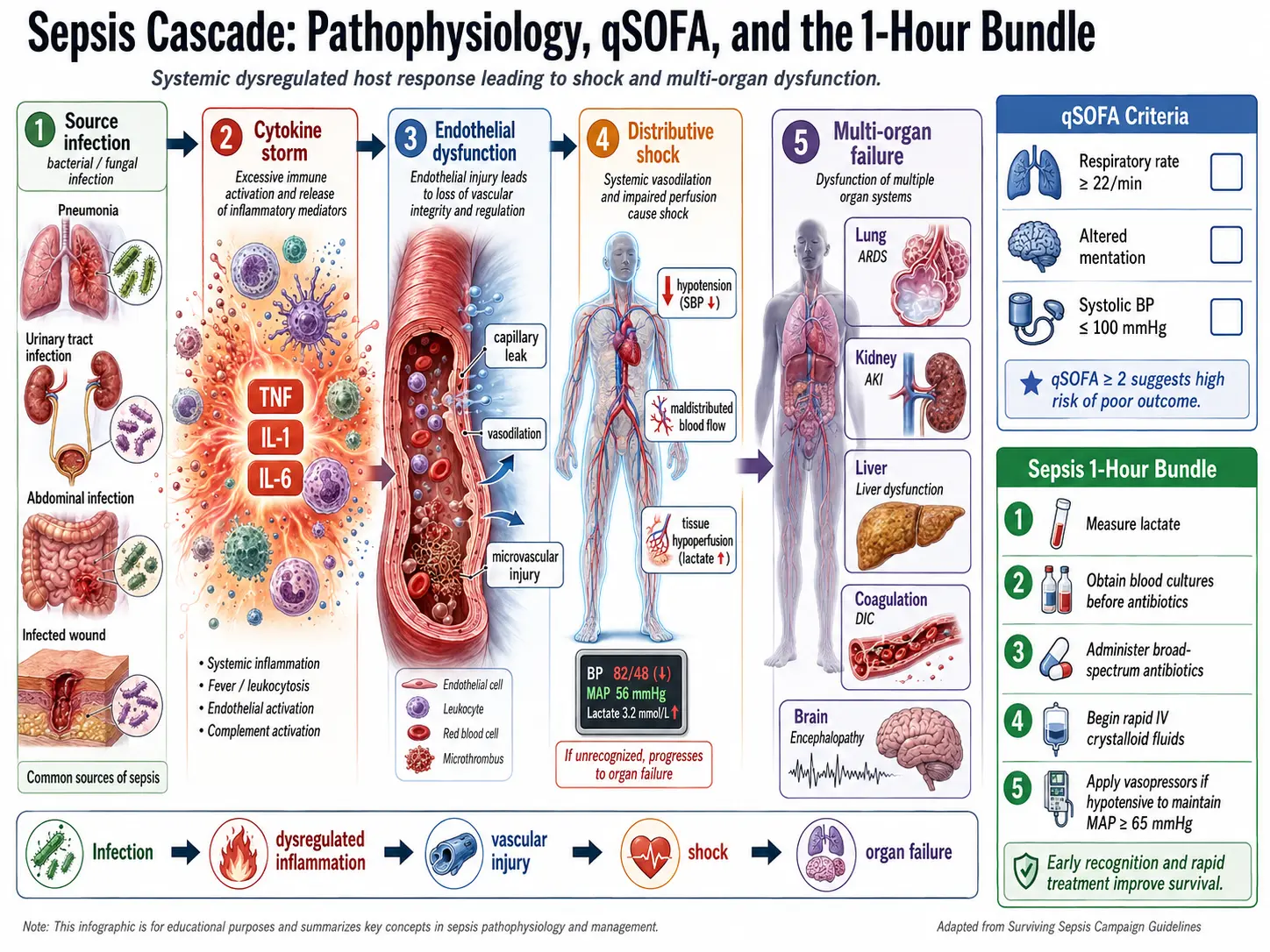
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Sepsis is defined by the Third International Consensus Definitions (Sepsis-3, 2016) as life-threatening organ dysfunction caused by a dysregulated host response to infection. This landmark definition replaced the older systemic inflammatory response syndrome (SIRS)-based criteria, recognizing that the pathologic process in sepsis extends far beyond simple inflammation to encompass immunosuppression, coagulation activation, mitochondrial dysfunction, and cellular metabolic failure.
Septic shock is a subset of sepsis characterized by profound circulatory, cellular, and metabolic abnormalities, operationally defined as vasopressor requirement to maintain mean arterial pressure (MAP) ≥65 mmHg AND serum lactate >2 mmol/L (>18 mg/dL) in the absence of hypovolemia.
Sepsis represents a medical emergency. Every hour of delay in appropriate antibiotic administration is independently associated with increased mortality. It is the leading cause of death in intensive care units worldwide and a major driver of preventable hospital mortality, long-term disability, and healthcare expenditure.
2. Epidemiology
Sepsis is among the most common and deadly conditions encountered in acute care medicine. Global epidemiological estimates include:
- Incidence: Approximately 48.9 million sepsis cases occur worldwide each year (Rudd et al., Lancet 2020), with 11 million deaths — representing 19.7% of all global deaths.
- United States: Approximately 1.7 million adults develop sepsis annually; nearly 270,000 die — more annual deaths than from prostate cancer, breast cancer, and HIV combined. Sepsis accounts for 1 in 3 hospital deaths and costs the US healthcare system >$24 billion annually.
- ICU burden: Sepsis accounts for approximately 10–30% of ICU admissions across different healthcare settings.
- Demographics: Incidence increases sharply with age; patients >65 years account for approximately 60–85% of sepsis cases. Male sex, Black race, and lower socioeconomic status are independently associated with higher incidence and mortality in US studies.
- Pediatric sepsis: An estimated 3 million neonatal and 4 million pediatric sepsis cases occur annually; leading cause of child mortality globally.
The incidence of sepsis has increased over recent decades — partly reflecting true epidemiological change (aging population, increasing immunocompromised hosts, antimicrobial resistance) and partly improved recognition and coding. Case fatality rates have declined from >40% to 20–25% in high-income settings due to protocolized care, though they remain higher in low- and middle-income countries.
3. Pathophysiology
The pathophysiology of sepsis involves a complex, dynamic interplay between pro-inflammatory and counter-regulatory immune responses that is highly heterogeneous across patients:
Innate Immune Activation
Pattern recognition receptors (PRRs) — including Toll-like receptors (TLRs), NOD-like receptors (NLRs), and RIG-I-like receptors — recognize pathogen-associated molecular patterns (PAMPs) such as lipopolysaccharide (LPS, gram-negative), peptidoglycan, lipoteichoic acid (gram-positive), flagellin, and viral nucleic acids. Activation triggers downstream signaling via NF-κB and MAP kinase pathways, inducing transcription of pro-inflammatory cytokines (TNF-α, IL-1β, IL-6, IL-8, IL-12) and type I interferons.
Cytokine Storm and Endothelial Injury
Systemic cytokine release causes widespread endothelial activation and dysfunction: upregulation of adhesion molecules (ICAM-1, VCAM-1, E-selectin), increased vascular permeability (capillary leak syndrome), glycocalyx shedding, and loss of endothelial barrier integrity. The result is interstitial edema, decreased oncotic pressure, and impaired organ perfusion.
Coagulation Cascade Activation
Tissue factor (TF) expression on activated endothelium and monocytes initiates the extrinsic coagulation pathway. Simultaneous suppression of natural anticoagulants (protein C, antithrombin III, tissue factor pathway inhibitor) and impaired fibrinolysis promote microvascular thrombosis and disseminated intravascular coagulation (DIC), further compromising organ perfusion.
Mitochondrial Dysfunction and Cellular Metabolic Failure
Nitric oxide (NO) overproduction by inducible nitric oxide synthase (iNOS) inhibits cytochrome c oxidase, impairing mitochondrial oxidative phosphorylation — leading to bioenergetic failure and "cytopathic hypoxia" even when oxygen delivery is adequate. This underlies the phenomenon of elevated lactate in the absence of tissue hypoperfusion in some sepsis patients.
Immunosuppression and Immunoparalysis
Concurrent with or following the hyper-inflammatory phase, a profound immunosuppressive state develops — characterized by T-cell apoptosis, T-cell exhaustion (upregulation of inhibitory checkpoints PD-1, CTLA-4, TIM-3), reduced MHC-II expression on antigen-presenting cells, IL-10 and TGF-β overproduction, and functional monocyte deactivation. This creates a state of acquired immunodeficiency that predisposes to secondary infections and reactivation of latent viruses (CMV, HSV, EBV).
Organ Dysfunction Mechanisms
- Acute kidney injury: Renal microvascular thrombosis, tubular epithelial cell apoptosis, inflammation-mediated tubular injury (not primarily ischemic).
- Acute respiratory distress syndrome (ARDS): Diffuse alveolar damage from neutrophil influx, reactive oxygen species, and loss of alveolar-capillary barrier integrity.
- Septic cardiomyopathy: TNF-α and IL-1β–mediated myocardial depression, NO-mediated reduced calcium sensitivity, and mitochondrial dysfunction causing reversible biventricular dysfunction.
- Hepatic dysfunction: Kupffer cell activation, intrahepatic cholestasis, sinusoidal microthrombosis.
- Encephalopathy: BBB disruption, neuroinflammation, microvascular thrombosis, neurotransmitter dysregulation.
4. Etiology and Risk Factors
Common Infectious Sources
- Lung (pneumonia): Most common source (approximately 35–50% of cases); bacteria include S. pneumoniae, S. aureus, gram-negative bacilli, and Legionella; also influenza and respiratory syncytial virus.
- Urinary tract: Second most common (approximately 20–30%); E. coli, Klebsiella, Enterococcus, Pseudomonas.
- Abdominal/intra-abdominal: Peritonitis (perforated viscus, appendicitis), cholangitis, diverticulitis; often polymicrobial.
- Primary bacteremia/line sepsis: S. aureus (including MRSA), coagulase-negative staphylococci (catheter-associated), gram-negative rods.
- Skin/soft tissue: Necrotizing fasciitis, cellulitis; S. pyogenes, S. aureus, polymicrobial.
- CNS: N. meningitidis, S. pneumoniae, Listeria monocytogenes.
- Fungal sepsis: Candida spp. (predominantly ICU patients, broad-spectrum antibiotic exposure, TPN, renal replacement therapy).
Host Risk Factors
- Advanced age (>65 years), neonates/young infants
- Immunosuppression: HIV/AIDS, solid organ or hematopoietic stem cell transplant, chemotherapy, corticosteroids, biologic agents
- Chronic diseases: diabetes mellitus, CKD, cirrhosis, COPD, heart failure, malignancy
- Indwelling devices: urinary catheters, central venous catheters, mechanical ventilation
- Recent surgery or invasive procedures; burns
- Genetic polymorphisms in TLR4, TNF-α, IL-1 receptor antagonist, protein C pathway
5. Clinical Presentation
SOFA Score (Sequential Organ Failure Assessment)
Sepsis-3 defines organ dysfunction as an acute change in total SOFA score ≥2 points from baseline, attributable to infection. SOFA quantifies dysfunction across six organ systems:
- Respiratory: PaO₂/FiO₂ ratio (P:F ratio)
- Coagulation: Platelet count
- Liver: Serum bilirubin
- Cardiovascular: MAP and vasopressor requirement
- CNS: Glasgow Coma Scale score
- Renal: Serum creatinine and urine output
Each domain scored 0–4; total score 0–24. Score ≥2 above baseline associated with hospital mortality >10%.
qSOFA (Quick SOFA)
Bedside screening tool for identifying high-risk patients outside the ICU. One point each for: altered mental status (GCS <15), respiratory rate ≥22 breaths/min, systolic BP ≤100 mmHg. Score ≥2 identifies patients at increased risk for prolonged ICU stay or in-hospital death. Sensitivity lower than SOFA for sepsis diagnosis but useful as a rapid alert.
Clinical Signs and Symptoms
Early sepsis: Fever (>38°C) or hypothermia (<36°C), tachycardia (>90 bpm), tachypnea (>20 breaths/min), altered mental status, hypotension (SBP <90 mmHg or MAP <70 mmHg), decreased urine output, skin mottling, delayed capillary refill.
Septic shock: Refractory hypotension requiring vasopressors, cold/clammy extremities (cold shock) or warm vasodilated periphery (warm distributive shock), oliguria/anuria, markedly elevated lactate, declining consciousness, petechiae/purpura (DIC), jaundice, respiratory distress (ARDS).
Neonatal/pediatric sepsis: Temperature instability, poor feeding, lethargy, respiratory distress, bulging fontanelle, petechial rash (meningococcemia), hypoglycemia, jaundice — often lacking classic fever.
6. Diagnosis
Sepsis diagnosis is fundamentally clinical, supported by laboratory and microbiological data. No single biomarker is diagnostic.
Microbiology
- Blood cultures: Obtain ≥2 sets (aerobic and anaerobic) from separate venipuncture sites before antibiotic administration — without delaying antibiotic administration by >45 minutes. Blood cultures positive in approximately 30–40% of sepsis cases.
- Source-directed cultures: Urine (midstream or catheter), sputum (respiratory sepsis), wound/abscess drainage, CSF (if meningitis suspected), bronchoalveolar lavage, pleural/peritoneal fluid as clinically indicated.
- Rapid diagnostic platforms: PCR-based blood pathogen identification (e.g., BioFire FilmArray BCID2, T2Bacteria) can identify organisms and resistance genes within hours from positive blood cultures — reducing time to targeted therapy.
Biomarkers
- Serum lactate: Elevated lactate (≥2 mmol/L) reflects tissue hypoperfusion (or metabolic derangement) and is both a diagnostic criterion for septic shock and a prognostic marker. Serial lactate measurement guides resuscitation; lactate clearance ≥10% at 2 hours is a target. Lactate ≥4 mmol/L is associated with mortality >40%.
- Procalcitonin (PCT): Acute-phase reactant released by parenchymal cells in response to bacterial (not viral) infection. Useful for antibiotic de-escalation decisions; rise and fall pattern more clinically informative than single value. PCT-guided protocols reduce antibiotic exposure without worsening outcomes.
- C-reactive protein (CRP): Non-specific marker of systemic inflammation; less useful for diagnosing bacterial vs. non-infectious inflammation than PCT.
- Complete blood count: Leukocytosis (WBC >12,000/mm³), leukopenia (<4,000/mm³), or >10% immature neutrophils (band forms); thrombocytopenia indicates DIC or organ involvement.
- Comprehensive metabolic panel: Elevated creatinine (AKI), elevated bilirubin (hepatic dysfunction), hyperglycemia (stress response), hypoalbuminemia.
- Coagulation studies: Elevated PT/INR, PTT, D-dimer; decreased fibrinogen — DIC profile.
- Arterial blood gas: Metabolic acidosis (low HCO₃, negative base excess), elevated anion gap, PaO₂/FiO₂ ratio for ARDS assessment.
Imaging
Chest radiograph or CT chest (pneumonia, pleural effusion, ARDS); CT abdomen/pelvis (intra-abdominal source, abscess); bedside point-of-care ultrasound (POCUS) for cardiac function, IVC collapsibility, pleural effusions, biliary pathology.
7. Treatment
Sepsis management is time-critical. The Surviving Sepsis Campaign (SSC) 2021 guidelines form the cornerstone of evidence-based management.
Hour-1 Bundle (SSC 2021)
- Measure lactate. Re-measure lactate if initial lactate >2 mmol/L.
- Obtain blood cultures before antibiotics (without delaying antibiotic administration).
- Administer broad-spectrum antibiotics within 1 hour of recognition.
- IV crystalloid resuscitation: 30 mL/kg IV crystalloid (normal saline or lactated Ringer's) for sepsis-induced hypotension or lactate ≥4 mmol/L.
- Vasopressors if patient remains hypotensive during/after fluid resuscitation: maintain MAP ≥65 mmHg.
Antibiotic Therapy
Empiric broad-spectrum therapy should be initiated immediately, guided by suspected source, local antibiogram, and patient risk factors for resistant organisms:
- Community-acquired sepsis (no MRSA/Pseudomonas risk): Piperacillin-tazobactam 4.5 g IV q6h OR ceftriaxone 2 g IV q24h + metronidazole (if abdominal source).
- Hospital/healthcare-associated or high MRSA risk: Add vancomycin (target AUC/MIC 400–600 via AUC-based monitoring) OR daptomycin 6–10 mg/kg IV q24h.
- Pseudomonas/MDR gram-negative risk (immunocompromised, prior colonization, healthcare-associated): Meropenem 1–2 g IV q8h OR cefepime 2 g IV q8h OR piperacillin-tazobactam 4.5 g IV q6h (extended infusion 4h).
- Carbapenem-resistant Enterobacterales (CRE): Ceftazidime-avibactam, meropenem-vaborbactam, or imipenem-cilastatin-relebactam based on susceptibility.
- Fungal sepsis (Candida risk): Echinocandin (micafungin 100 mg IV q24h, caspofungin 70 mg loading then 50 mg q24h) as first-line; transition to fluconazole if susceptible.
De-escalation to targeted therapy based on culture and sensitivity results as soon as possible, guided by clinical improvement and biomarker trends. Antibiotic duration: generally 7–10 days; shorter courses (5–7 days) adequate for many sources (community-acquired pneumonia, uncomplicated UTI). PCT-guided de-escalation is SSC-recommended.
Hemodynamic Resuscitation
- Vasopressors: Norepinephrine is first-line (0.01–3 mcg/kg/min IV); maintain MAP ≥65 mmHg. Add vasopressin 0.03 units/min for norepinephrine-sparing or refractory shock. Dopamine no longer recommended due to increased arrhythmia risk.
- Epinephrine: Second-line adjunct for refractory shock.
- Angiotensin II (Giapreza): Approved for high-dose vasopressor-dependent vasodilatory shock.
- Fluid resuscitation: Balanced crystalloids (Lactated Ringer's or Plasma-Lyte) preferred over normal saline (reduced AKI risk per SMART and SALT-ED trials). Avoid starches (HES) due to increased AKI and mortality. Albumin may be considered for patients requiring substantial volumes of crystalloid. Avoid fluid overload — daily fluid balance and POCUS-guided titration.
- Corticosteroids: Hydrocortisone 200–300 mg/day IV (as 50 mg q6h or continuous infusion) for septic shock requiring high vasopressor doses (norepinephrine ≥0.25 mcg/kg/min for ≥4 hours). ADRENAL and APROCCHSS trials support use in vasopressor-dependent septic shock. Taper, do not abruptly stop.
Source Control
Early and definitive source control (drainage of abscess, debridement of infected/necrotic tissue, removal of infected device) is essential — ideally within 6–12 hours of identification. Percutaneous drainage preferred over open surgical drainage when feasible.
Organ Support
- Mechanical ventilation (ARDS): Low tidal volume strategy (6 mL/kg ideal body weight), PEEP titration, plateau pressure <30 cmH₂O, prone positioning for severe ARDS (P:F <150), conservative fluid strategy after initial resuscitation.
- Renal replacement therapy (RRT): Continuous RRT (CRRT) preferred in hemodynamically unstable patients with AKI (indications: refractory hyperkalemia, acidosis, uremia, fluid overload).
- Transfusion: Red blood cell transfusion threshold: hemoglobin <7 g/dL (restrictive strategy, TRICC and TRISS trials); platelet transfusion for <10,000/mm³ or <20,000/mm³ with bleeding risk.
- Glycemic control: Target glucose 140–180 mg/dL; avoid hypoglycemia (strict normoglycemia target of 80–110 mg/dL shown harmful in NICE-SUGAR trial).
- DVT prophylaxis: Low-molecular-weight heparin preferred; add mechanical prophylaxis (IPC devices).
- Stress ulcer prophylaxis: For patients with risk factors (mechanical ventilation >48h, coagulopathy); proton pump inhibitors or H₂ receptor antagonists.
- Nutrition: Early enteral nutrition preferred over parenteral; avoid complete caloric starvation in first 7 days but avoid overfeeding.
8. Complications
- Acute respiratory distress syndrome (ARDS): Diffuse alveolar damage; develops in 30–40% of ICU sepsis patients.
- Acute kidney injury (AKI): Occurs in 40–50% of septic shock; approximately 5–6% require RRT; risk of permanent CKD.
- Disseminated intravascular coagulation (DIC): Microvascular thrombosis and simultaneous hemorrhage; associated with purpura fulminans.
- Septic cardiomyopathy: Reversible biventricular dysfunction in 40–50% of septic shock; echocardiographic monitoring essential.
- Septic encephalopathy: Most common organ dysfunction in sepsis; ranging from delirium to coma; associated with long-term cognitive impairment.
- Hepatic dysfunction: Cholestatic jaundice, transaminase elevation, acute liver failure.
- Secondary infections: Ventilator-associated pneumonia, catheter-associated UTI, Clostridioides difficile infection — driven by immunosuppression and antibiotic exposure.
- Post-intensive care syndrome (PICS): A cluster of new or worsening impairments in physical function, cognitive function, and mental health (PTSD, anxiety, depression) persisting after ICU discharge; affects up to 50% of sepsis survivors.
9. Prognosis
Prognosis varies widely by severity, source, organism, host factors, and timeliness of intervention:
- Sepsis without shock: Hospital mortality approximately 10–20%.
- Septic shock: Hospital mortality 30–50%; up to 80% in elderly or immunocompromised with multiorgan failure.
- Lactate as prognostic marker: Lactate 2–4 mmol/L: mortality ~25%; lactate >4 mmol/L: mortality >40%.
- SOFA score: Each 1-point increase in SOFA score associated with approximately 10% increase in mortality risk.
- Long-term outcomes: Sepsis survivors face substantially increased 1–5-year mortality compared with matched controls, driven by persistent immune dysfunction, recurrent infections, organ failure, frailty, and cognitive impairment.
- Lactate clearance: Failure to clear lactate (reduction <10% at 2 hours) is associated with significantly worse outcomes.
Earlier recognition and initiation of treatment are the most modifiable determinants of outcome. Studies demonstrate that each hour of delay in appropriate antibiotics is associated with an approximately 7–10% increase in mortality from septic shock.
10. Prevention
- Infection control and prevention: Central line-associated bloodstream infection (CLABSI) bundles (hand hygiene, maximal sterile barriers, chlorhexidine skin prep, optimal site selection, daily review of line necessity); catheter-associated UTI (CAUTI) prevention; VAP prevention bundles (head-of-bed elevation, oral decontamination, minimizing sedation, spontaneous breathing trials).
- Antimicrobial stewardship: Appropriate antibiotic use reduces selective pressure for resistant organisms that drive healthcare-associated sepsis.
- Sepsis screening tools: Electronic health record–based early warning systems (e.g., EPIC Sepsis Model, Modified Early Warning Score) to facilitate earlier recognition.
- Public awareness: The Sepsis Alliance "TIME" mnemonic (Temperature, Infection, Mental decline, Extremely ill) and national sepsis campaigns.
- Post-sepsis follow-up: Survivorship clinics addressing PICS — physical rehabilitation, psychological support, medication reconciliation, and monitoring for recurrent infection.
11. Recent Research and Advances
- Sepsis-3 phenotyping: Machine-learning–derived clinical phenotypes (α, β, γ, δ; Seymour et al., JAMA 2019) identify biologically distinct subgroups with differential treatment responses — moving toward precision sepsis medicine.
- Immune modulation: Anti-PD-1/PD-L1 checkpoint blockade (nivolumab, pembrolizumab) to reverse immunoparalysis is in Phase 2 trials. IL-7 to restore T-cell proliferation is also under investigation.
- Endotype-guided therapy: SRS1 (immunosuppressed) vs. SRS2 (pro-inflammatory) endotypes may respond differently to corticosteroids — the VANISH trial demonstrated endotype-specific harm and benefit from vasopressin vs. norepinephrine.
- Host-response biomarkers: Angiopoietin-2/angiopoietin-1 ratio (endothelial injury), soluble thrombomodulin, cell-free DNA, and protein biomarker panels (e.g., InSep) are being validated for diagnosis and treatment guidance.
- Rapid microbiological diagnosis: Metagenomic next-generation sequencing (mNGS) of blood/plasma (e.g., Karius test) identifies pathogens including rare and atypical organisms within 24 hours without culture.
- IV vitamin C: CITRIS-ALI and VITAMINS trials did not show mortality benefit for high-dose IV ascorbic acid; LOVIT-ICU found harm. Routine use not supported by current evidence.
- Thiamine: May benefit thiamine-deficient septic shock patients; low cost and low risk support empiric use in high-risk populations.
- Balanced vs. saline resuscitation: SMART trial and BaSICS trial data support balanced crystalloids; ongoing PLUS trial.
- AI-driven early warning: Prospective validation of machine-learning EHR-based sepsis alerts in improving time-to-treatment and outcomes is ongoing.
12. References
- Singer M, Deutschman CS, Seymour CW, et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315:801–810.
- Rudd KE, Johnson SC, Agesa KM, et al. Global, regional, and national sepsis incidence and mortality, 1990–2017. Lancet. 2020;395:200–211.
- Evans L, Rhodes A, Alhazzani W, et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock 2021. Intensive Care Med. 2021;47:1181–1247.
- Seymour CW, Kennedy JN, Wang S, et al. Derivation, validation, and potential treatment implications of novel clinical phenotypes for sepsis. JAMA. 2019;321:2003–2017.
- Shankar-Hari M, Phillips GS, Levy ML, et al. Developing a new definition and assessing new clinical criteria for septic shock. JAMA. 2016;315:775–787.
- Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006;34:1589–1596.
- Annane D, Renault A, Brun-Buisson C, et al. Hydrocortisone plus Fludrocortisone for Adults with Septic Shock (APROCCHSS). N Engl J Med. 2018;378:809–818.
- Venkatesh B, Finfer S, Cohen J, et al. Adjunctive glucocorticoid therapy in patients with septic shock (ADRENAL). N Engl J Med. 2018;378:797–808.
- Semler MW, Self WH, Wanderer JP, et al. Balanced crystalloids versus saline in critically ill adults (SMART). N Engl J Med. 2018;378:829–839.
- Hotchkiss RS, Monneret G, Payen D. Immunosuppression in sepsis: a novel understanding of the disorder and a new therapeutic approach. Lancet Infect Dis. 2013;13:260–268.
- Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369:840–851.
- PRISM Investigators. Early, goal-directed therapy for septic shock — a patient-level meta-analysis (ProCESS, ARISE, ProMISe). N Engl J Med. 2017;376:2223–2234.
- Prescott HC, Angus DC. Enhancing recovery from sepsis: a review. JAMA. 2018;319:62–75.
- Bone RC, Balk RA, Cerra FB, et al. Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis (ACCP/SCCM Consensus). Chest. 1992;101:1644–1655.
- Shankar-Hari M, Saha R, Wilson J, et al. Rate and risk factors for rehospitalisation after a critical illness — a population-level retrospective cohort study. Crit Care. 2020;24:314.
Research Papers
The following PubMed topic searches retrieve current peer-reviewed literature on Sepsis. Each link opens a live PubMed query so you always see the most recent publications.
- Sepsis review pathophysiology
- Surviving Sepsis Campaign guideline
- Sepsis-3 definitions
- qSOFA sepsis screening
- Early goal directed therapy sepsis
- Sepsis mortality ICU
- Septic shock vasopressor norepinephrine
- Procalcitonin sepsis biomarker
- Sepsis antibiotic timing outcome
- Vitamin C thiamine sepsis trial
- Pediatric sepsis review
- Post-sepsis syndrome long term
Connections
- Cellulitis
- Meningitis
- Vitamin C
- Immune Boosting
- Staphylococcus Aureus
- Klebsiella Pneumoniae
- Escherichia Coli
- Vitamin B1
- Pneumonia
- Clostridium Difficile
- Acute Kidney Injury
- Acute Respiratory Distress Syndrome
- Disseminated Intravascular Coagulation
- Complete Blood Count
- Comprehensive Metabolic Panel
- Heart Failure
- Creatinine
- Jaundice
Featured Videos
Sepsis: Systemic inflammatory response syndrome (SIRS) to multiple organ dysfunction syndrome (MODS)

Septic Shock Nursing (Sepsis) Treatment, Pathophysiology, Symptoms Distributive

Septic (Sepsis) Pathway

Sepsis and Septic Shock

Sepsis: Emergency™

What is Sepsis?

Sepsis: First Response - Educational Video

Community-Acquired Pneumonia and Sepsis

SEPSIS PATHOPHYSIOLOGY IN 7 SIMPLE STEPS (2018)

What Are The Signs Of Sepsis? | This Morning

The Turning Point -- Surviving Sepsis

Sepsis & Septic Shock - Symptoms, Pathophysiology, Diagnosis, Treatment

Faces of Sepsis: Survivor Panel

Sepsis & Septic Shock Explained | Pathophysiology, Phenotypes & More 🦠
Understanding Sepsis (Sepsis Explained Clearly)

What YOU Need to Know about Sepsis