Diverticulitis
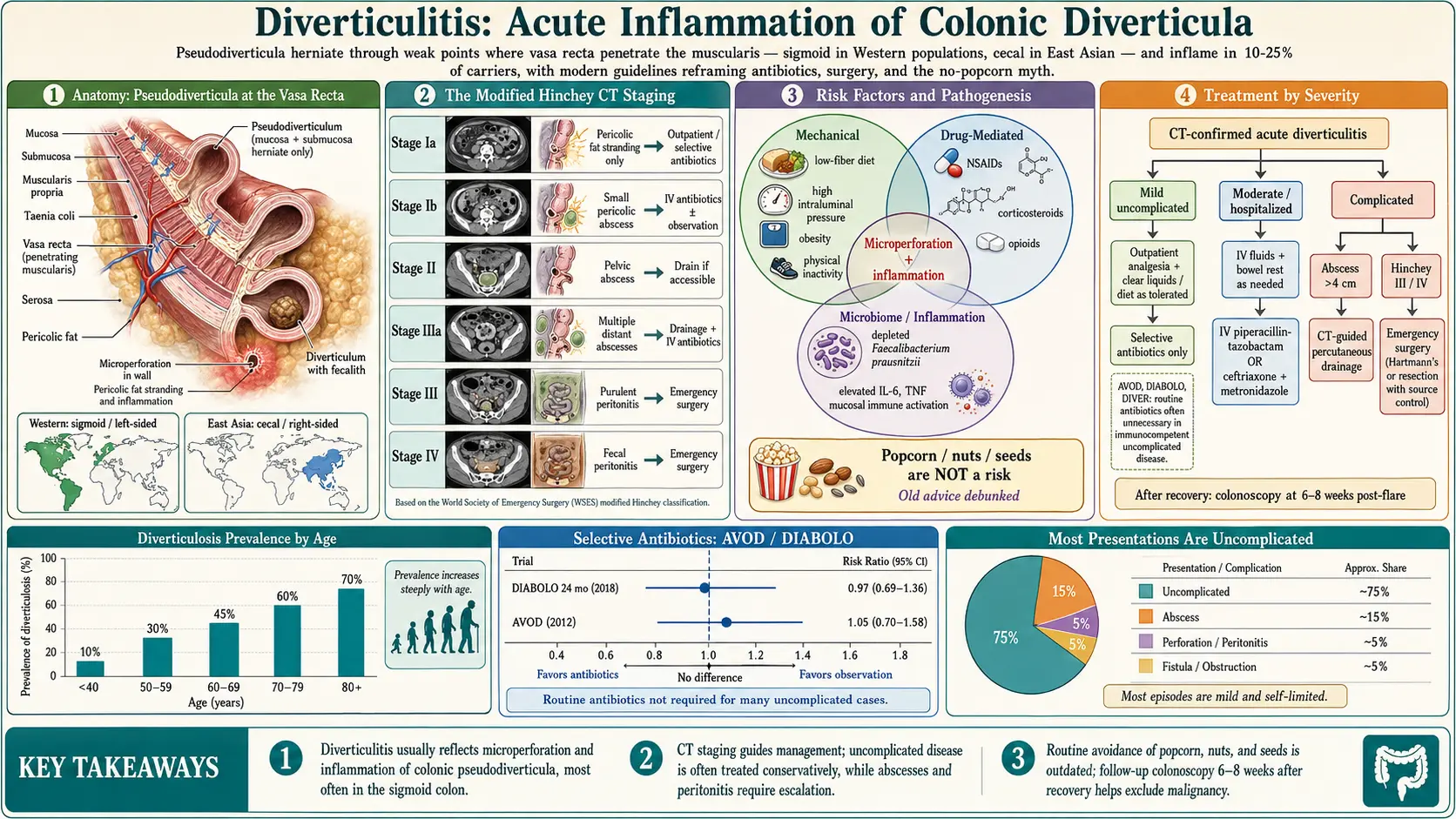
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Diverticular disease of the colon encompasses a spectrum of conditions arising from colonic diverticula — acquired pulsion herniae of the colonic mucosa and submucosa through weaknesses in the muscularis propria at sites where vasa recta penetrate the bowel wall. The spectrum includes:
- Diverticulosis: The asymptomatic presence of diverticula (the anatomical substrate); affects the majority of individuals with diverticula
- Symptomatic uncomplicated diverticular disease (SUDD): Chronic or recurrent abdominal symptoms attributable to diverticula in the absence of macroscopic inflammation or complications
- Diverticulitis: Macroscopic inflammation and/or infection of one or more diverticula, with a spectrum from mild uncomplicated diverticulitis to complicated disease (abscess, perforation, fistula, obstruction)
- Diverticular hemorrhage: Acute lower gastrointestinal hemorrhage from rupture of a vas rectum at the neck or dome of a diverticulum
The sigmoid colon is the predominant site in Western populations due to high intraluminal pressures generated in this segment. Right-sided diverticulitis predominates in East Asian populations.
2. Epidemiology
Diverticulosis is one of the most prevalent gastrointestinal conditions in the Western world. Autopsy studies estimate prevalence of approximately 5% in individuals under 40, rising to 30% by age 60 and 65–70% by age 85 in Western populations. The overall prevalence in the United States is estimated at 35–50% in adults over 50, corresponding to approximately 70 million Americans with diverticulosis.
Diverticulitis develops in 10–25% of individuals with diverticulosis over a lifetime. Annually, diverticulitis accounts for approximately 300,000 hospitalizations and 1.5 million outpatient visits in the US, with direct healthcare costs exceeding $2 billion. The incidence of acute diverticulitis is increasing, particularly in younger adults (under 50), where it now accounts for 25–30% of all diverticulitis hospitalizations.
Complicated diverticulitis accounts for 15–20% of all acute diverticulitis episodes. Perforation carries significant mortality (5–20%), particularly in elderly and immunocompromised patients and when surgery is required.
3. Pathophysiology
Diverticulosis Formation
Colonic diverticula in Western populations are pseudodiverticula (false diverticula) — containing only mucosa and submucosa, lacking the muscularis propria layer. They form at anatomical weak points where vasa recta (terminal branches of the marginal artery) penetrate the circular muscle layer to supply the submucosa and mucosa. High intraluminal pressures — generated by segmental colonic contractions against formed stool in the narrow sigmoid — drive mucosal herniation through these points.
The "low-fiber hypothesis" proposes that reduced dietary fiber leads to smaller, denser stools requiring greater phasic contraction pressures for propulsion. Structural changes in the colonic wall (increased elastin deposition, collagen cross-linking, and reduced smooth muscle compliance with aging) amplify susceptibility. Microbiome dysbiosis and increased intestinal permeability may perpetuate low-grade mucosal inflammation even in "asymptomatic" diverticulosis.
Diverticulitis Pathogenesis
The exact initiating event is debated. The traditional erosion hypothesis proposes that inspissated fecal material (fecalith) within a diverticulum causes pressure-mediated erosion of the diverticular wall, leading to micro- or macro-perforation and pericolic inflammation. Alternatively, increased intraluminal pressure from segmental contraction may directly cause microperforations.
Colonic microbiome dysbiosis — with relative depletion of Faecalibacterium prausnitzii and overgrowth of Bacteroides and Fusobacterium species — leads to mucosal immune dysregulation and susceptibility to inflammation. Mast cell activation and increased mucosal pro-inflammatory cytokines (IL-6, TNF-α) have been demonstrated in SUDD and are implicated in the transition to overt diverticulitis. The degree of pericolic involvement determines classification into uncomplicated (confined) versus complicated (abscess, free perforation) disease.
4. Etiology and Risk Factors
Risk factors for diverticulosis development:
- Advancing age (most important — strongly age-associated)
- Low dietary fiber intake (relative risk approximately 1.3–2.0 for highest vs. lowest fiber quintile)
- Western dietary pattern (high in processed foods, red meat, refined carbohydrates)
- Physical inactivity — sedentary lifestyle
- Obesity (BMI >30) — particularly visceral adiposity
- Connective tissue disorders: Marfan syndrome, Ehlers-Danlos syndrome (associated with right-sided diverticulosis)
Risk factors for diverticulitis in individuals with diverticulosis:
- NSAID use (OR ~1.5–2.5; impairs mucosal prostaglandin defenses and promotes microperforation)
- Corticosteroid use (OR ~2.0; impairs immune response and mucosal repair)
- Opioid use (reduces colonic motility, increases intraluminal pressure)
- Immunosuppression (transplant recipients, HIV/AIDS, chemotherapy): increased severity and atypical presentations
- Smoking (OR ~1.4)
- Physical inactivity and obesity
- Red and processed meat consumption
- Low dietary fiber intake — controversial for diverticulitis specifically (some studies show no independent protective effect beyond diverticulosis prevention)
5. Clinical Presentation
Uncomplicated diverticulitis (75–80% of acute episodes):
- Acute onset left lower quadrant (LLQ) pain (most common) — constant, dull, or cramping; right-sided or periumbilical pain in right-sided diverticulitis (more common in East Asian patients) or cecal diverticulitis (may mimic appendicitis)
- Low-grade fever (38–38.5°C)
- Localized tenderness in LLQ on palpation; voluntary guarding; bowel sounds typically present
- Altered bowel habits (constipation more common than diarrhea; either may occur)
- Nausea, anorexia
- Urinary symptoms (dysuria, frequency) if pericolic inflammation involves the bladder or ureter
Complicated diverticulitis (20–25% of acute episodes):
- Pericolic abscess: Fever >38.5°C, more severe pain, tender palpable mass (if large), WBC elevation
- Free perforation with peritonitis: Sudden severe generalized abdominal pain, peritoneal signs (board-like rigidity, rebound tenderness), absent bowel sounds, septic shock — surgical emergency
- Fistula: Colovesical fistula presents with pneumaturia, fecaluria, recurrent UTIs; colovaginal fistula with vaginal flatus or feculent vaginal discharge; colocutaneous fistula with skin drainage
- Colonic obstruction: Progressive abdominal distension, obstipation, vomiting — from acute inflammation, abscess compression, or chronic fibrous stricture after recurrent episodes
Differential diagnosis: Colorectal carcinoma (may be indistinguishable clinically and radiologically), irritable bowel syndrome, ischemic colitis, inflammatory bowel disease (Crohn's colitis), appendicitis (right-sided), ovarian cyst, tubo-ovarian abscess, endometriosis, gynecologic pathology.
6. Diagnosis
Laboratory Studies
- Full blood count: leukocytosis (WBC >11,000/mm³) in 65–75%; absence does not exclude diverticulitis
- C-reactive protein (CRP): elevated (>50 mg/L typical; >150 mg/L suggests complicated disease); most useful for severity assessment and monitoring response
- Serum creatinine and electrolytes: baseline renal function; hyperkalemia may suggest obstruction
- Urinalysis: mild pyuria or hematuria may occur from ureteral or bladder proximity to inflamed colon; does not necessarily indicate UTI
- Blood cultures: in febrile patients with suspected complicated disease or sepsis
Imaging
CT abdomen and pelvis with IV contrast is the imaging modality of choice (sensitivity 94%, specificity 99% for diverticulitis):
- Characteristic findings: pericolic fat stranding, diverticular thickening of the sigmoid colon wall, presence of diverticula, pericolic abscess (if complicated)
- CT allows classification of severity (Hinchey/modified Hinchey classification) and guides management decisions regarding interventional radiology drainage or surgery
- CT colonography (virtual colonoscopy) can be performed after acute episode resolution to evaluate the colon if optical colonoscopy is not feasible
Abdominal ultrasound: Can identify pericolic inflammation and diverticula in experienced hands; sensitivity approximately 92%, specificity 90%; preferred in pregnancy, young patients, and when limiting radiation exposure; operator-dependent
MRI abdomen/pelvis: Excellent sensitivity and specificity without radiation; reserved for pregnant patients, young adults, and equivocal CT cases; less available in acute settings
Hinchey Classification (Modified — CT-Based)
- Stage Ia: Pericolic fat stranding (mesenteric inflammation)
- Stage Ib: Pericolic or mesenteric abscess (<4–5 cm)
- Stage II: Pelvic or retroperitoneal abscess
- Stage IIIa: Small (<5 cm) or multiple distant abscesses
- Stage III: Purulent peritonitis (ruptured pericolic or pelvic abscess into peritoneum)
- Stage IV: Fecal peritonitis (free fecal perforation)
Colonoscopy
Colonoscopy is contraindicated during acute diverticulitis (perforation risk). It is recommended 6–8 weeks after resolution of the acute episode in all patients to:
- Exclude colorectal carcinoma (CT may not distinguish diverticulitis from perforated colon cancer)
- Evaluate for IBD, ischemic colitis, or other colonic pathology
- Assess extent and distribution of diverticulosis
7. Treatment
Uncomplicated Diverticulitis
Outpatient management (mild uncomplicated, hemodynamically stable, able to tolerate oral fluids, no significant comorbidities):
- Clear liquid diet progressing to low-fiber diet as tolerated; evidence does not support routine bowel rest in mild uncomplicated disease
- Antibiotics — evolving paradigm: Multiple RCTs (AVOD trial, DIABOLO trial, DIVER trial) have demonstrated that antibiotics do not improve outcomes in mild uncomplicated diverticulitis compared to analgesics and dietary modification in terms of hospital admissions, complications, recurrence, or need for surgery. Current guidelines (ACG, AGA, EAES) recommend selective antibiotic use — reserving antibiotics for immunocompromised patients, significant comorbidities, fever >38.5°C, leukocytosis >15,000/mm³, or significant CRP elevation (>150 mg/L).
- When antibiotics are indicated (oral): amoxicillin-clavulanate 875/125 mg BID × 7–10 days (first-line); alternatively ciprofloxacin 500 mg BID + metronidazole 500 mg TID; trimethoprim-sulfamethoxazole DS BID + metronidazole in penicillin-allergic patients
- Analgesics: paracetamol (preferred); avoid NSAIDs and opioids if possible (NSAIDs worsen mucosal integrity; opioids increase colonic pressure)
Inpatient management (fever >38.5°C, WBC >15,000/mm³, inability to tolerate oral intake, CRP >150 mg/L, elderly or immunocompromised, failed outpatient management, or uncertain diagnosis):
- IV fluids, bowel rest (clear liquids advancing as tolerated)
- IV antibiotics: piperacillin-tazobactam 3.375 g IV q6h; or ceftriaxone 2 g IV daily + metronidazole 500 mg IV q8h; or ertapenem 1 g IV daily (for penicillin allergy or polymicrobial coverage)
- Transition to oral antibiotics after clinical improvement (48–72 hours afebrile, WBC normalizing, tolerating oral intake)
Complicated Diverticulitis
- Pericolic abscess (Hinchey Ia/Ib — <4 cm): IV antibiotics alone; high success rate (85–90%) without drainage
- Larger abscess (Hinchey II/IIIa — ≥4 cm): CT-guided or ultrasound-guided percutaneous drainage + IV antibiotics; drainage of >4 cm abscesses reduces failure rate and avoids emergent surgery; elective sigmoid resection planned 4–6 weeks after resolution in selected patients
- Free perforation with peritonitis (Hinchey III/IV): Emergency surgery; Hartmann's procedure (sigmoid resection, end colostomy, closure of rectal stump) or primary resection and anastomosis with or without diverting ileostomy (in select hemodynamically stable patients with experienced surgeon); laparoscopic lavage and drainage has been shown inferior to resection for Hinchey III in randomized trials (LADIES, LOLA trial)
- Fistula: Surgical resection with fistula takedown and anastomosis; one-stage procedures preferred in elective settings
- Colonic obstruction: Endoscopic stenting as bridge to surgery vs. emergency resection depending on degree of obstruction and patient condition
Elective Sigmoid Colectomy
The contemporary view (supported by American Society of Colon and Rectal Surgeons guidelines, 2020) is that elective sigmoid resection should be individualized rather than based on number of episodes:
- Recommended after complicated diverticulitis (abscess, fistula, stricture)
- Considered in patients with frequent recurrences severely affecting quality of life
- Considered in immunocompromised patients
- Laparoscopic sigmoid colectomy is the preferred approach when technically feasible — reduced complications, shorter hospital stay, faster recovery
- The previous guideline of "elective resection after 2 uncomplicated episodes" is no longer supported — recurrence risk after second episode (<20%) does not justify routine resection
8. Complications
- Pericolic abscess: Most common complication; 10–15% of acute diverticulitis episodes; may complicate uncomplicated diverticulitis not treated with antibiotics
- Free perforation with peritonitis: Fecal peritonitis carries 20–30% mortality; purulent peritonitis 5–10% mortality; highest risk in elderly and immunocompromised
- Colovesical fistula: Most common diverticular fistula (65%); predominantly affects men (uterus protects posterior bladder in women); presents with pneumaturia, fecaluria, recurrent polymicrobial UTIs
- Colovaginal fistula: Second most common; occurs in women; post-hysterectomy uterine absence increases risk
- Colonic stricture: From fibrosis following recurrent inflammation; may require resection; must exclude malignancy
- Diverticular hemorrhage: Complicates 5% of diverticulosis; most common cause of acute lower GI bleeding in the elderly; right-sided diverticula bleed more frequently than left-sided despite lower prevalence; 80% stop spontaneously; managed with colonoscopic hemostasis, angiographic embolization, or surgical resection
- Post-diverticulitis syndrome: Persistent abdominal symptoms after resolution of acute diverticulitis, attributable to visceral hypersensitivity and altered gut microbiome; overlaps clinically with IBS and may require management as a functional GI disorder
9. Prognosis
The prognosis of uncomplicated diverticulitis is excellent with appropriate management; over 95% of patients with mild uncomplicated diverticulitis recover without sequelae. The recurrence rate after a first episode of uncomplicated diverticulitis is approximately 10–20% at 5 years, with most recurrences also being uncomplicated. The risk of complications at recurrence is not significantly higher than at first presentation.
After complicated diverticulitis (abscess, fistula), the risk of recurrent complicated diverticulitis is higher, supporting elective resection in these patients. Perforation with fecal peritonitis carries significant mortality (20–30%), particularly in elderly and immunocompromised patients requiring emergency surgery. Long-term complications of Hartmann's procedure (irreversible colostomy in a significant proportion due to non-reversal) substantially affect quality of life.
10. Prevention
- Dietary fiber: High-fiber diet (25–38 g/day from fruits, vegetables, legumes, whole grains) reduces the risk of diverticulosis development and may reduce SUDD; evidence specifically for preventing diverticulitis is less consistent
- Physical activity: Regular vigorous physical activity is associated with approximately 20–40% lower risk of diverticulitis in prospective cohort studies (Nurses' Health Study, Health Professionals Follow-up Study)
- Red meat restriction: High red meat consumption (particularly unprocessed red meat >4 servings/week) is associated with increased diverticulitis risk (HR ~1.3–1.5)
- Avoidance of nuts, seeds, popcorn: Historical advice to avoid these foods to prevent diverticulitis has been refuted by prospective data (Health Professionals Follow-up Study); these foods are not associated with increased diverticulitis risk and may actually be protective
- Mesalazine (mesalamine): The DIVA trial and meta-analyses have shown that mesalazine (5-aminosalicylic acid, 1.6–3.0 g/day) reduces recurrence risk and SUDD symptoms after acute uncomplicated diverticulitis, with a number needed to treat of approximately 8–10 to prevent one recurrence; use is not universally endorsed in guidelines but is widely used in clinical practice
- Rifaximin: Cyclic rifaximin (400 mg BID × 7 days/month) reduces symptoms in SUDD and may reduce diverticulitis recurrence; more robust evidence in Italian guidelines
- NSAID avoidance: Substituting acetaminophen for NSAIDs in patients with known diverticulosis
11. Recent Research and Advances
- Paradigm shift on antibiotics: The AVOD (Sweden, 2012), DIABOLO (Netherlands, 2017), and DIVER (Spain, 2017) trials collectively established that routine antibiotic therapy for mild uncomplicated acute diverticulitis provides no benefit over analgesics and observation in immunocompetent patients with respect to complications, recurrence, or hospital readmission. This has fundamentally changed management guidelines globally.
- Laparoscopic lavage rejection: The LADIES and SCANDIV trials (2015–2016) demonstrated that laparoscopic lavage was inferior to sigmoid resection for Hinchey III perforated diverticulitis, with higher reoperation rates, leading to withdrawal of this approach from standard guidelines.
- Gut microbiome and diverticular disease: Metagenomic profiling has consistently identified dysbiosis in diverticulosis and diverticulitis, including depletion of butyrate-producing species (Roseburia intestinalis, Faecalibacterium prausnitzii) and enrichment of pro-inflammatory species. FMT and targeted probiotic strategies for SUDD and post-diverticulitis syndrome are under investigation.
- Post-diverticulitis syndrome phenotyping: Recognition that 20–30% of patients have persistent symptoms after acute diverticulitis ("post-diverticulitis irritable bowel" or functional overlay) has prompted studies of neuromodulators, low-FODMAP diet, and gut-directed psychotherapy in this population.
- Mesalamine for recurrence prevention: A 2021 systematic review and meta-analysis confirmed a significant reduction in diverticulitis recurrence with cyclic mesalamine therapy; ongoing RCTs aim to define optimal dosing and duration.
- Personalized elective surgery decision-making: Risk calculators incorporating age, BMI, CRP, and comorbidity score are being developed to individualize elective sigmoid colectomy recommendations beyond the traditional episode-count-based approach.
12. References
- Feuerstein JD, Falchuk KR. Diverticulosis and Diverticulitis. Mayo Clin Proc. 2016;91(8):1094–1104.
- Strate LL, Morris AM. Epidemiology, Pathophysiology, and Treatment of Diverticulitis. Gastroenterology. 2019;156(5):1282–1298.
- Schultz JK, Azhar N, Binda GA, et al. European Society of Coloproctology: guidelines for the management of diverticular disease of the colon. Colorectal Dis. 2020;22(Suppl 2):5–28.
- Feingold D, Steele SR, Lee S, et al. Practice parameters for the treatment of sigmoid diverticulitis. Dis Colon Rectum. 2014;57(3):284–294.
- Hall J, Hardiman K, Lee S, et al. The American Society of Colon and Rectal Surgeons Clinical Practice Guidelines for the Treatment of Left-Sided Colonic Diverticulitis. Dis Colon Rectum. 2020;63(6):728–747.
- Jaung R, Nisbet S, Gosselink MP, et al. Antibiotics Do Not Reduce Length of Hospital Stay for Uncomplicated Diverticulitis in a Randomized Controlled Trial. Clin Gastroenterol Hepatol. 2021;19(3):503–510.
- Daniels L, Unlü Ç, de Korte N, et al. Randomized clinical trial of observational versus antibiotic treatment for a first episode of CT-proven uncomplicated acute diverticulitis. Br J Surg. 2017;104(1):52–61.
- Schultz JK, Yaqub S, Wallon C, et al. Laparoscopic Lavage vs Primary Resection for Acute Perforated Diverticulitis: The SCANDIV Randomized Clinical Trial. JAMA. 2015;314(13):1364–1375.
- Strate LL, Liu YL, Aldoori WH, Syngal S, Giovannucci EL. Obesity increases the risks of diverticulitis and diverticular bleeding. Gastroenterology. 2009;136(1):115–122.
- Strate LL, Liu YL, Syngal S, Aldoori WH, Giovannucci EL. Nut, corn, and popcorn consumption and the incidence of diverticular disease. JAMA. 2008;300(8):907–914.
- Aldoori WH, Giovannucci EL, Rockett HR, Sampson L, Rimm EB, Willett WC. A prospective study of dietary fiber types and symptomatic diverticular disease in men. J Nutr. 1998;128(4):714–719.
- Binda GA, Cuomo R, Laghi A, et al. Practice parameters for the treatment of colonic diverticular disease: Italian Society of Colon and Rectal Surgery (SICCR) guidelines. Tech Coloproctol. 2015;19(10):615–626.
- Tursi A, Brandimarte G, Elisei W, et al. Randomised clinical trial: mesalazine and/or probiotics in maintaining remission of symptomatic uncomplicated diverticular disease — a double-blind, randomised, placebo-controlled study. Aliment Pharmacol Ther. 2013;38(7):741–751.
- Peery AF, Crockett SD, Murphy CC, et al. Burden and Cost of Gastrointestinal, Liver, and Pancreatic Diseases in the United States: Update 2018. Gastroenterology. 2019;156(1):254–272.
- Tursi A, Papa A, Danese S. Review article: the pathophysiology and medical management of diverticulosis and diverticular disease of the colon. Aliment Pharmacol Ther. 2015;42(6):664–684.
- Andeweg CS, Knobben L, Hendriks JC, Bleichrodt RP, van Goor H. How to diagnose acute left-sided colonic diverticular disease: proposal for a clinical scoring system. Ann Surg. 2011;253(5):940–946.
Research Papers
Curated PubMed topic searches of peer-reviewed literature on Diverticulitis. Each link opens a live PubMed query so you always see the most current studies.
- PubMed: Diverticulitis treatment guidelines
- PubMed: Antibiotic therapy in diverticulitis
- PubMed: Dietary fiber and diverticular disease
- PubMed: CT imaging of diverticulitis
- PubMed: Surgery for complicated diverticulitis
- PubMed: Recurrent diverticulitis
- PubMed: Colonoscopy after diverticulitis
- PubMed: Sigmoid diverticulitis
- PubMed: Perforation and abscess
- PubMed: Mesalamine and rifaximin trials
- PubMed: Epidemiology of diverticular disease
- PubMed: Outpatient management
Connections
- Inflammatory Bowel Disease
- Irritable Bowel Syndrome
- Crohn's Disease
- SIBO
- Magnesium
- Gut Healing Protocol
- Lentils
- Turmeric
- Resistant Starches and Gut Microbiome
- Probiotics
- Constipation
- Abdominal Pain
- Sepsis
- Cancer
- Gallbladder Disease
- Ulcerative Colitis
- Peptic Ulcer Disease
- Surgery Decisions Resection J Pouch Ostomy
Featured Videos

Diverticulosis & Diverticulitis: Symptoms & Treatments - Ask A Nurse | @LevelUpRN

How To Treat Diverticulitis - NEW DATA

What is diverticulitis? Causes, symptoms, treatment & more

Diverticulitis | Diverticular Disease Nursing | Diverticulosis Symptoms, Diet, Treatment NCLEX

How To Treat Diverticulitis in 2026

Diverticular Disease (diverticulitis) - Overview

What To Avoid with Diverticulitis | Risk Factors & Ways to Reduce Risk

Foods To Eat and Avoid With Diverticulitis

8 Things to Help Heal Diverticulitis | Dr. J9 Live