Cirrhosis
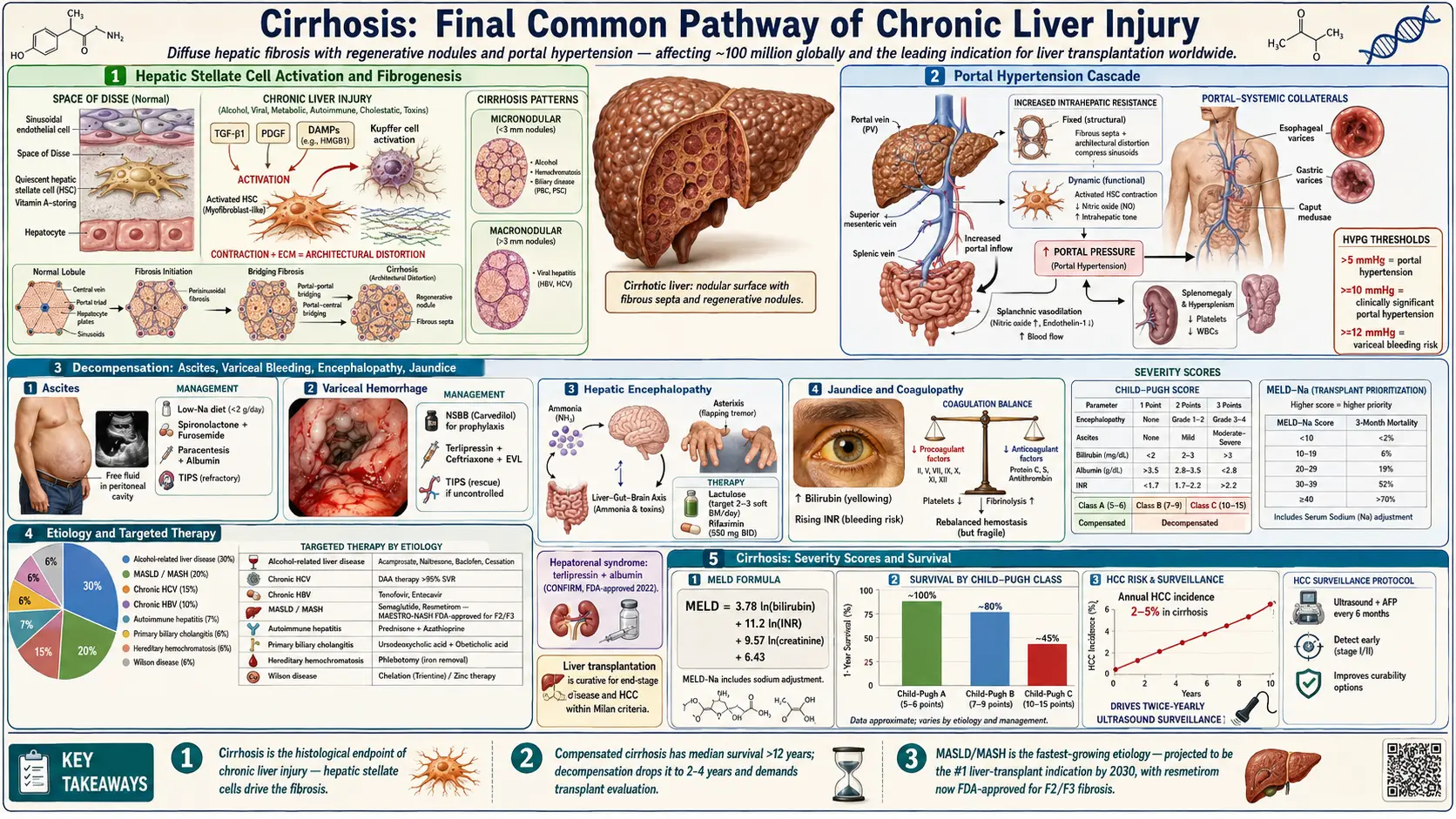
Cirrhosis is the final common pathway of chronic liver injury, characterized by diffuse hepatic fibrosis, nodular regeneration, and disruption of normal hepatic architecture. It results from progressive replacement of functional hepatocytes with fibrous scar tissue, leading to portal hypertension and impaired synthetic, metabolic, and detoxification functions. Cirrhosis is a leading cause of morbidity and mortality worldwide, and the primary indication for liver transplantation.
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Featured Videos
1. Overview
Cirrhosis represents the histological endpoint of chronic hepatic inflammation and fibrosis, regardless of etiology. The liver loses its normal lobular architecture, replaced by regenerative nodules surrounded by fibrous septa. This structural disruption creates intrahepatic shunting of portal blood, causing portal hypertension — the driver of most major complications including variceal hemorrhage, ascites, spontaneous bacterial peritonitis (SBP), hepatic encephalopathy, and hepatorenal syndrome.
Cirrhosis is clinically divided into two stages with profoundly different prognoses: compensated cirrhosis (absence of decompensation events, median survival >12 years) and decompensated cirrhosis (characterized by ascites, variceal bleeding, encephalopathy, or jaundice; median survival 2–4 years without transplantation). A third state, acute-on-chronic liver failure (ACLF), represents an acute deterioration with multi-organ failure and very high short-term mortality (28-day mortality 30–60%).
2. Epidemiology
Cirrhosis affects an estimated 100 million people globally. It accounts for approximately 1.3 million deaths per year worldwide, representing 2.4% of all global mortality. In the United States, approximately 630,000 adults have compensated cirrhosis and 440,000 have decompensated cirrhosis, with an additional 1.5 million undiagnosed cases estimated. The age-standardized prevalence is 14.9 per 100,000 population.
The epidemiology of cirrhosis has shifted dramatically: while alcohol-related liver disease (ALD) and chronic hepatitis C (HCV) historically dominated, metabolic dysfunction-associated steatotic liver disease (MASLD, formerly NAFLD/NASH) is now the fastest-growing etiology, projected to become the leading indication for liver transplantation by 2030. Hepatitis B remains the most common cause worldwide, particularly in sub-Saharan Africa and East Asia. The male-to-female ratio for cirrhosis is approximately 2:1, reflecting higher rates of alcohol consumption and metabolic risk factors in men.
3. Pathophysiology
Fibrogenesis
Regardless of etiology, hepatic fibrosis is driven by activation of hepatic stellate cells (HSCs). In the normal liver, HSCs exist in a quiescent state, storing vitamin A as retinyl esters. Chronic hepatocyte injury triggers release of damage-associated molecular patterns (DAMPs), activating Kupffer cells (hepatic macrophages) and promoting release of TGF-β1, PDGF, and other profibrogenic mediators. These signals activate HSCs, which transdifferentiate into myofibroblast-like cells, losing vitamin A stores and acquiring contractile and fibrogenic properties.
Activated HSCs produce: extracellular matrix proteins (type I and III collagen, fibronectin, laminin); tissue inhibitors of metalloproteinases (TIMPs) that inhibit matrix degradation; and matrix metalloproteinases (MMPs) that remodel but also perpetuate fibrosis. Portal fibroblasts and bone marrow-derived fibrocytes also contribute to the fibrogenic response. The resulting fibrosis distorts hepatic architecture, creating regenerative nodules, which are classified as:
- Micronodular cirrhosis: Nodules <3 mm; typically caused by alcohol, hemochromatosis, biliary obstruction
- Macronodular cirrhosis: Nodules >3 mm; typically caused by viral hepatitis
- Mixed micronodular and macronodular
Portal Hypertension
Portal hypertension (portal pressure >5 mmHg; clinically significant at >10 mmHg; hepatic venous pressure gradient [HVPG] ≥10 mmHg) results from two mechanisms:
- Increased intrahepatic resistance: Fixed component (fibrous tissue, regenerative nodules) and dynamic component (HSC contraction mediated by reduced endothelial NOS and NO production, increased endothelin-1)
- Increased portal blood flow: Splanchnic arterial vasodilation (mediated by increased NO, prostacyclin, glucagon) leads to increased portal venous inflow, amplifying portal hypertension despite formation of portosystemic collaterals
Portal pressure above 12 mmHg (HVPG threshold) is required for variceal bleeding. Hyperdynamic circulation (increased cardiac output, decreased systemic vascular resistance) is characteristic of advanced cirrhosis, leading to renal vasoconstriction and RAAS activation.
Child-Pugh Classification
The Child-Pugh score (originally Child-Turcotte, modified by Pugh) assesses severity using five parameters: bilirubin, albumin, INR/prothrombin time, ascites, and hepatic encephalopathy. Each parameter is scored 1–3 (best to worst):
- Child-Pugh A (5–6 points): Compensated cirrhosis; 1-year survival ~100%
- Child-Pugh B (7–9 points): Significant dysfunction; 1-year survival ~80%
- Child-Pugh C (10–15 points): Decompensated cirrhosis; 1-year survival ~45%
The Child-Pugh score guides transplant listing, surgical risk assessment, and prediction of variceal bleeding risk.
4. Etiology and Risk Factors
Common Causes (responsible for 90% of cases)
- Alcohol-related liver disease (ALD): Most common cause in Western countries; threshold dose approximately 40 g/day in men, 20 g/day in women, for ≥5 years; acetaldehyde toxicity, oxidative stress, gut dysbiosis, and immune activation drive fibrosis
- Chronic hepatitis C (HCV): Cirrhosis develops in 15–30% of chronically infected patients over 20–30 years; now largely preventable with DAA therapy
- Chronic hepatitis B (HBV): Cirrhosis risk 15–40% over 5 years in untreated HBV; viral replication (HBV DNA levels) is the primary driver
- MASLD/MASH (metabolic-associated steatohepatitis): Obesity, type 2 diabetes, dyslipidemia, hypertension as risk factors; fibrosis progression rate 0.07–0.14 stages/year; now termed MASLD per 2023 multi-society nomenclature update
Less Common Causes
- Autoimmune hepatitis (AIH): ANA, anti-SMA, anti-LKM antibodies; responsive to corticosteroids + azathioprine
- Primary biliary cholangitis (PBC): Anti-mitochondrial antibodies (AMA); portal inflammation → biliary cirrhosis; ursodeoxycholic acid ± obeticholic acid therapy
- Primary sclerosing cholangitis (PSC): Multifocal biliary strictures; association with IBD; no proven medical therapy; transplantation curative
- Hereditary hemochromatosis: HFE gene mutations (C282Y homozygosity); iron overload-induced oxidative injury; phlebotomy therapy
- Wilson's disease: ATP7B mutations; copper accumulation; chelation or zinc therapy
- Alpha-1 antitrypsin deficiency: PiZZ phenotype; liver disease from intracellular polymerization; transplantation curative
- Drug-induced liver injury (DILI): Methotrexate, amiodarone, isoniazid, nitrofurantoin
- Cardiac cirrhosis (congestive hepatopathy): Right heart failure causing hepatic venous congestion; sinusoidal congestion and centrilobular fibrosis
- Cryptogenic cirrhosis: Often represents unrecognized or burned-out MASLD (metabolic risk factors without current steatosis)
5. Clinical Presentation
Compensated Cirrhosis
- Often asymptomatic, discovered incidentally on imaging or blood tests
- Non-specific symptoms: fatigue, malaise, right upper quadrant discomfort, mild weight loss
- Physical examination: spider angiomata (>5 on upper body — suggestive of cirrhosis), palmar erythema, leukonychia, parotid enlargement (alcoholic), digital clubbing, gynecomastia (estrogen excess), testicular atrophy, loss of body hair
- Hepatomegaly (smooth, firm edge) or small firm liver in late cirrhosis; splenomegaly (portal hypertension)
Decompensated Cirrhosis
- Ascites: Flank dullness, shifting dullness, fluid wave (tense ascites); most common decompensating event
- Jaundice: Scleral icterus, tea-colored urine, pale stools
- Hepatic encephalopathy: Confusion, asterixis (liver flap), altered sleep-wake cycle, disorientation; grade I–IV (West Haven criteria)
- Variceal bleeding: Hematemesis, melena; hemodynamic instability
- Caput medusae: Dilated periumbilical veins (portosystemic shunting)
- Fetor hepaticus: Sweet musty breath odor (dimethyl sulfide)
- Muscle wasting (sarcopenia): Reduced temporal, thenar, hypothenar muscle bulk; common and prognostically important
6. Diagnosis
Liver Function Tests
- Bilirubin: Elevated in decompensated disease; total and direct fractions; conjugated hyperbilirubinemia predominates
- Albumin: Reduced (<3.5 g/dL) — reflects synthetic dysfunction; half-life 21 days
- Prothrombin time / INR: Elevated INR reflects reduced hepatic synthesis of clotting factors (II, V, VII, IX, X); half-life 6–24 hours makes INR more sensitive than albumin for acute changes
- Aminotransferases (AST, ALT): May be normal or only mildly elevated in cirrhosis (late stages have few remaining hepatocytes); AST:ALT ratio >2:1 suggests alcoholic etiology
- Alkaline phosphatase (ALP) and GGT: Elevated in cholestatic diseases (PBC, PSC, ALD)
- Platelet count: Thrombocytopenia (<150,000/µL) due to hypersplenism and reduced thrombopoietin synthesis; most sensitive CBC marker for significant fibrosis
- Serum sodium: Hyponatremia (dilutional; ADH excess; <130 mEq/L is a MELD-Na modifier and prognostic marker)
Non-Invasive Fibrosis Assessment
- FIB-4 index: [Age × AST] / [Platelet × √ALT]; <1.30 excludes advanced fibrosis; >2.67 predicts advanced fibrosis (F3–F4)
- APRI score: AST/upper limit of normal / platelet count × 100; >2.0 predicts cirrhosis
- Enhanced Liver Fibrosis (ELF) test: Serum markers (HA, PIIINP, TIMP-1)
- Transient elastography (FibroScan): Liver stiffness measurement (LSM) in kPa; >12.5 kPa suggests cirrhosis in MASLD; >17.5 kPa in ALD; >14 kPa in HCV; ≥20 kPa predicts clinically significant portal hypertension (CSPH)
- MR elastography: More accurate than ultrasound-based methods; measures liver stiffness in 3D; less affected by ascites and obesity
Liver Biopsy
Still considered the gold standard for diagnosis and staging of fibrosis. Staged by the METAVIR score (F0–F4, where F4 = cirrhosis) or Ishak score (0–6, where 5–6 = cirrhosis). Biopsy provides information on activity grade (A0–A3) independently of fibrosis stage. Increasingly replaced by non-invasive methods in HBV/HCV, but remains essential in uncertain etiology, suspected overlap syndromes, and before initiating immunosuppression in AIH.
Imaging
- Ultrasound: Heterogeneous echogenicity, nodular liver surface, caudate lobe hypertrophy, splenomegaly, ascites, portal vein diameter >13 mm; limited sensitivity for early cirrhosis
- CT/MRI with contrast: Regenerative nodules, heterogeneous enhancement, signs of portal hypertension (varices, splenomegaly); gadoxetate-enhanced MRI for hepatocellular carcinoma (HCC) surveillance
- Upper endoscopy: Assessment for esophageal/gastric varices; required in all newly diagnosed cirrhosis patients (Baveno VII criteria); may be deferred if LSM <20 kPa AND platelet count >150,000/µL
- Hepatic venous pressure gradient (HVPG): Gold standard for portal pressure measurement; ≥10 mmHg = clinically significant portal hypertension; ≥12 mmHg = variceal bleeding threshold
MELD Score
Model for End-Stage Liver Disease: MELD = 3.78 × ln(bilirubin mg/dL) + 11.2 × ln(INR) + 9.57 × ln(creatinine mg/dL) + 6.43. MELD-Na incorporates serum sodium (MELD-Na = MELD + 1.32 × [137 − Na] − [0.24 × MELD × (137 − Na)]). MELD-Na is used for organ allocation on the transplant waiting list in the United States; MELD 15–20 represents the threshold above which transplantation improves survival versus continued medical management.
7. Treatment
Treatment of Underlying Etiology
- ALD: Alcohol cessation (most important intervention; reverses early fibrosis, reduces portal pressure); AUDIT-C questionnaire; pharmacological support (acamprosate, naltrexone, baclofen for alcohol use disorder); management of alcohol-associated hepatitis (prednisolone for severe disease with Maddrey discriminant function ≥32)
- HCV: All-oral DAA regimens (sofosbuvir-based, glecaprevir/pibrentasvir); achieve sustained virological response (SVR) >95%; SVR reduces risk of decompensation and HCC but does not eliminate HCC surveillance requirement in cirrhosis
- HBV: Nucleos(t)ide analogues (tenofovir disoproxil fumarate, tenofovir alafenamide, entecavir); suppresses HBV DNA, reduces fibrosis progression and HCC risk
- MASLD/MASH: Weight loss 7–10% improves histology; GLP-1 receptor agonists (semaglutide, liraglutide) reduce liver fat and inflammation; resmetirom (thyroid hormone receptor-β agonist) FDA-approved for MASH with liver fibrosis F2/F3 (MAESTRO-NASH trial)
- PBC: Ursodeoxycholic acid (UDCA) 13–15 mg/kg/day; obeticholic acid for inadequate UDCA response; elafibranor, seladelpar (PPAR agonists) recently approved for PBC
- AIH: Prednisone 40–60 mg/day + azathioprine 50–100 mg/day; achieve biochemical remission in 80–85%; maintenance indefinitely to prevent relapse
- Hemochromatosis: Weekly phlebotomy until serum ferritin <50 ng/mL; maintenance phlebotomy every 2–4 months; chelation (deferoxamine, deferasirox) for severe cases
- Wilson's disease: D-penicillamine or trientine (copper chelation); zinc acetate for maintenance and asymptomatic patients
Management of Portal Hypertension and Complications
Primary prophylaxis of variceal bleeding: Non-selective beta-blockers (NSBBs — propranolol, nadolol, carvedilol) for medium/large varices; target heart rate reduction 25% or to 55–60 bpm; carvedilol preferred for its additional anti-alpha1-adrenergic vasodilatory effect; endoscopic variceal ligation (EVL) is equivalent for primary prophylaxis in patients intolerant of NSBBs.
Acute variceal bleeding: IV fluid resuscitation (target hemoglobin 7–8 g/dL; liberal transfusion worsens outcomes); vasoactive agents (terlipressin 2 mg IV q4h × 5 days, or octreotide/somatostatin analogues for 3–5 days); urgent endoscopy within 12 hours for EVL (esophageal) or injection sclerotherapy/tissue adhesive (gastric); broad-spectrum antibiotics (norfloxacin or IV ceftriaxone 1 g/day × 7 days); short-term albumin (1.5 g/kg on day 1, 1 g/kg on day 3) per INFECIR trial; TIPS (transjugular intrahepatic portosystemic shunt) for refractory bleeding or high-risk patients (Child-Pugh B with active bleeding, Child-Pugh C).
Ascites management: Low-sodium diet (<88 mEq/day); spironolactone (starting 100 mg/day, max 400 mg/day) ± furosemide (starting 40 mg/day, max 160 mg/day); maintain Na:K excretion ratio >1 or gradual weight loss 300–500 g/day (without peripheral edema); large-volume paracentesis (LVP) with IV albumin 6–8 g/L ascites removed for >5 L paracentesis (prevents post-paracentesis circulatory dysfunction); TIPS for refractory ascites in selected patients.
Spontaneous bacterial peritonitis (SBP): Diagnose by ascitic fluid PMN >250 cells/mm³; empiric IV ceftriaxone 2 g/day × 5 days; IV albumin 1.5 g/kg on day 1 and 1 g/kg on day 3 reduces AKI and mortality (Sort et al. NEJM 1999 landmark trial); secondary prophylaxis with norfloxacin 400 mg/day or trimethoprim-sulfamethoxazole.
Hepatic encephalopathy (HE): Identify and treat precipitants (infection, GI bleed, constipation, electrolyte abnormalities, sedatives); lactulose 30–45 mL q6–8h titrated to 2–3 soft stools/day (reduces intestinal ammonia production); rifaximin 550 mg twice daily as adjunct or alternative for secondary prevention (Bajaj et al. NEJM 2010); branched-chain amino acids; zinc supplementation; TIPS occlusion if TIPS-induced refractory HE.
Hepatorenal syndrome (HRS): Volume challenge with albumin 1 g/kg/day × 2 days; terlipressin + albumin is first-line (CONFIRM trial, FDA-approved 2022 for HRS-AKI); norepinephrine + albumin (ICU setting); TIPS for HRS-CKD; liver transplantation (± simultaneous liver-kidney transplantation for HRS-CKD >4–6 weeks).
Liver Transplantation
Curative therapy for end-stage liver disease, HCC within Milan criteria (single lesion ≤5 cm or ≤3 lesions each ≤3 cm), and selected cases of ACLF. Absolute contraindications include extrahepatic malignancy, active alcohol or substance use, severe cardiopulmonary disease, and sepsis. Five-year post-transplant survival exceeds 75–80% in carefully selected candidates.
8. Complications
- Variceal hemorrhage: Life-threatening; 20–30% mortality per acute bleeding episode; secondary prevention with NSBBs + EVL
- Ascites and spontaneous bacterial peritonitis: SBP occurs in 8–30% of cirrhotic inpatients; 1-year mortality after first SBP episode 30–50%
- Hepatic encephalopathy: Overt HE in 30–45% of cirrhotic patients; covert/minimal HE impairs driving ability and quality of life
- Hepatorenal syndrome: HRS-AKI (type 1, rapid onset) and HRS-CKD (type 2, progressive); median survival without transplantation 2–4 weeks (HRS-AKI) and 6 months (HRS-CKD)
- Hepatopulmonary syndrome (HPS): Intrapulmonary vascular dilation causing hypoxemia; PaO2 <80 mmHg on room air; contrast echocardiography (bubble study) is diagnostic; liver transplantation is the only cure
- Portopulmonary hypertension (PoPH): Mean PAP >25 mmHg with portal hypertension; prostacyclin analogues, PDE-5 inhibitors, endothelin antagonists; transplantation if mPAP <35 mmHg
- Hepatocellular carcinoma (HCC): Annual incidence 2–5% in cirrhosis; HCV/HBV highest risk; diagnosed by imaging (LI-RADS criteria on contrast CT/MRI) without biopsy requirement; curative treatments (resection, ablation, transplantation) applicable only to early-stage disease
- Coagulopathy: "Rebalanced hemostasis" — reduced synthesis of both procoagulant and anticoagulant factors; INR overestimates bleeding risk as thrombomodulin-modified thrombin generation is relatively preserved; routine FFP transfusion not recommended before procedures without bleeding
- Malnutrition and sarcopenia: Present in 50–90% of patients with advanced cirrhosis; associated with increased mortality, infections, and HE; late evening snacks (branched-chain amino acids) recommended
9. Prognosis
Prognosis in cirrhosis is primarily determined by the degree of portal hypertension and hepatic synthetic dysfunction. The transition from compensated to decompensated cirrhosis markedly worsens prognosis: 5-year mortality is approximately 20% in compensated and 70% in decompensated cirrhosis. After a first decompensating event, median survival is 2–4 years without transplantation.
Prognostic scoring systems:
- MELD-Na: Primary organ allocation tool; each 1-point increase corresponds to ~2–3% increase in 90-day mortality; MELD-Na >25 indicates urgent listing priority
- Child-Pugh: Class C associated with 45% 1-year survival without transplantation; used for surgical risk assessment
- CLIF-C ACLF score: Predicts 28-day mortality in ACLF; organ failure count most predictive (3 organ failures → 90-day mortality ~75%)
- HVPG: Reduction to <12 mmHg prevents variceal bleeding; <10 mmHg prevents all decompensation events; reduction ≥10% from baseline correlates with improved outcomes
Key predictors of survival: MELD-Na, serum sodium, presence and grade of HE, Child-Pugh class, presence of HCC, renal function (creatinine), and response to treatment of underlying etiology (SVR in HCV, HBV suppression).
10. Prevention
- Alcohol reduction: Primary prevention of ALD through public health interventions; minimum unit pricing policies shown to reduce alcohol-related liver disease hospitalizations
- HCV screening and treatment: Universal one-time HCV RNA or antibody testing in adults 18–79 years (USPSTF recommendation); eradication with DAAs prevents cirrhosis development
- Metabolic risk factor management: Weight loss, physical activity, treatment of diabetes and dyslipidemia to prevent MASLD progression
- HCC surveillance: Liver ultrasound ± AFP every 6 months in all cirrhotic patients; annual MRI in high-risk patients (Child-Pugh B/C, HBV, elevated AFP)
- Secondary prophylaxis of complications: NSBBs for portal hypertension, norfloxacin/rifaximin for SBP secondary prophylaxis, lactulose/rifaximin for HE prevention
11. Recent Research and Advances
- Resmetirom (Rezdiffra): First FDA-approved pharmacotherapy specifically for MASH with liver fibrosis (F2/F3); thyroid hormone receptor-β agonist reduces hepatic fat and reverses fibrosis in MAESTRO-NASH trial (2024); a landmark advance in MASH management
- GLP-1 receptor agonists: Semaglutide 2.4 mg weekly achieves histological improvement in MASH; ESSENCE trial ongoing for cirrhosis outcomes; weight loss of 10–15% achieves fibrosis regression
- Terlipressin for HRS-AKI: CONFIRM trial (2021) led to FDA approval of terlipressin in the United States; 30-day reversal of HRS-AKI significantly superior to placebo
- TIPS optimization: Small-diameter covered TIPS (8 mm vs. 10 mm) reduces HE while maintaining portal pressure reduction; "preemptive TIPS" within 72 hours of variceal bleeding in high-risk patients (Child-Pugh C or B with active bleeding) reduces rebleeding and mortality (Garcia-Pagan et al.)
- Antifibrotic therapy: No currently approved antifibrotic; FXR agonists (obeticholic acid beyond PBC), galectin-3 inhibitors, and CCR2/CCR5 antagonists (cenicriviroc — did not meet endpoints in CENTAUR/AURORA trials) are being investigated
- ACLF pathobiology: CANONIC study defined ACLF as a distinct syndrome with systemic inflammation as central driver; therapeutic targets include immunomodulation, G-CSF (hepatic stem cell mobilization), microbiome modulation
- Baveno VII consensus: 2022 updated guidance allows non-invasive avoidance of screening endoscopy in compensated advanced chronic liver disease using LSM + platelet count thresholds; expands role of carvedilol for portal hypertension management
12. References
- European Association for the Study of the Liver. EASL Clinical Practice Guidelines for the management of patients with decompensated cirrhosis. Journal of Hepatology. 2018;69(2):406–460.
- Baveno VII Faculty. Expanding consensus in portal hypertension: report of the Baveno VII Consensus Workshop. Journal of Hepatology. 2022;76(4):959–974.
- Harrison SA, Bedossa P, Guy CD, et al. A phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis (MAESTRO-NASH). New England Journal of Medicine. 2024;390(6):497–509.
- Wong F, Pappas SC, Boyer TD, et al. Terlipressin plus albumin for the treatment of type 1 hepatorenal syndrome (CONFIRM). New England Journal of Medicine. 2021;384(9):818–828.
- Garcia-Tsao G, Abraldes JG, Berzigotti A, Bosch J. Portal hypertensive bleeding in cirrhosis: risk stratification, diagnosis, and management — 2016 practice guidance by the AASLD. Hepatology. 2017;65(1):310–335.
- Sort P, Naveau S, Gaudreault P, et al. Effect of intravenous albumin on renal impairment and mortality in patients with cirrhosis and spontaneous bacterial peritonitis. New England Journal of Medicine. 1999;341(6):403–409.
- Moreau R, Jalan R, Gines P, et al. Acute-on-chronic liver failure is a distinct syndrome that develops in patients with acute decompensation of cirrhosis (CANONIC study). Gastroenterology. 2013;144(7):1426–1437.
- Sanyal AJ, Chalasani N, Kowdley KV, et al. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis (PIVENS). New England Journal of Medicine. 2010;362(18):1675–1685.
- Bajaj JS, Heuman DM, Hylemon PB, et al. Randomised clinical trial: lactulose versus rifaximin for preventing minimal hepatic encephalopathy episodes. Alimentary Pharmacology and Therapeutics. 2011;33(11):1250–1258.
- Friedman SL. Mechanisms of hepatic fibrogenesis. Gastroenterology. 2008;134(6):1655–1669.
- Mallet V, van Bömmel F, Doerig C, et al. Management of viral hepatitis in patients with haematological malignancy and in patients undergoing haemopoietic stem cell transplantation. Lancet Infectious Diseases. 2016;16(8):e139–e151.
- D'Amico G, Garcia-Tsao G, Pagliaro L. Natural history and prognostic indicators of survival in cirrhosis: a systematic review of 118 studies. Journal of Hepatology. 2006;44(1):217–231.
- Cotter TG, Rinella M. Nonalcoholic fatty liver disease 2020: the state of the disease. Gastroenterology. 2020;158(7):1851–1864.
- Ginès P, Krag A, Abraldes JG, et al. Liver cirrhosis. Lancet. 2021;398(10308):1359–1376.
Research Papers
Explore current literature on cirrhosis via PubMed topic searches. These links open live PubMed searches for the listed keywords — results update as new studies are indexed.
- Clinical trials on cirrhosis — PubMed search
- Clinical trials on alcoholic liver disease — PubMed search
- Clinical trials on hepatorenal syndrome — PubMed search
- Clinical trials on portal hypertension — PubMed search
- Clinical trials on hepatic encephalopathy — PubMed search
- Clinical trials on liver fibrosis non-invasive assessment — PubMed search
- Clinical trials on MELD score transplant — PubMed search
Connections
- Liver Disease
- Hemochromatosis
- Hepatitis C
- Hepatitis B
- Non-Alcoholic Fatty Liver Disease
- Acute Kidney Injury
- Gallbladder Disease
- Hypertension
- Hepatitis
- Jaundice
- Obesity
- Diabetes
- Zinc
- Milk Thistle
- GGT
- Thrombocytopenia
- Edema
- Urinalysis
Featured Videos

Cirrhosis - causes, symptoms, diagnosis, treatment, pathology

Living with Cirrhosis: Tips, Symptoms, Treatments | Mass General Brigham

Liver Cirrhosis: Signs, Symptoms, Treatment | Max Hospital

Cirrhosis of the Liver Simplified: Pathophysiology, Symptoms, & NCLEX Nursing Tips

Can Liver Cirrhosis be cured? | Symptoms and Treatment explained

Cirrhosis of the liver: Diet and nutrition | Ohio State Medical Center

Liver Cirrhosis | Types | Pathophysiology | Causes | Sign & Symptoms | Diagnose | Treatment

What Is the Fastest Way to Treat Liver Cirrhosis?

Cirrhosis of the Liver Nursing Care Management Symptoms NCLEX