Gestational Diabetes Mellitus
Gestational diabetes mellitus (GDM) is defined as glucose intolerance first recognized during pregnancy, encompassing a spectrum from mild insulin resistance to near-overt diabetes. GDM complicates 6–16% of pregnancies worldwide and carries significant maternal and fetal consequences — including fetal macrosomia, birth injury, neonatal hypoglycemia, preeclampsia, cesarean delivery, and long-term maternal risk of type 2 diabetes — that are substantially mitigated by early diagnosis and glycemic management.
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
- Featured Videos
1. Overview
GDM was first formally described by O'Sullivan and Mahan in 1964, who established oral glucose tolerance test (OGTT) thresholds based on maternal risk of developing overt diabetes postpartum — a criterion that has been revised considerably since. The modern understanding of GDM recognizes it as a heterogeneous condition: some women have pre-existing undiagnosed type 2 diabetes or impaired glucose tolerance unmasked by the metabolic demands of pregnancy (overt diabetes in pregnancy), while the majority experience true pregnancy-induced relative insulin resistance that resolves postpartum but confers a 7-fold increased lifetime risk of type 2 diabetes.
Two principal diagnostic frameworks are in current use worldwide, reflecting the central controversy in GDM management — the one-step versus two-step screening approach:
- IADPSG (International Association of the Diabetes and Pregnancy Study Groups) one-step criteria: Universal 75g OGTT at 24–28 weeks; single abnormal value sufficient for diagnosis; based on HAPO study data correlating glucose levels with adverse perinatal outcomes
- Carpenter-Coustan two-step approach: 50g glucose challenge test (GCT) as non-fasting screen; women above threshold undergo 100g 3-hour OGTT; two or more abnormal values required; endorsed by ACOG and widely used in the United States
2. Epidemiology
GDM is among the most common medical complications of pregnancy:
- Global prevalence: approximately 6–16% of all pregnancies; IDF estimates 21.1 million live births (15.8%) affected by some form of hyperglycemia in pregnancy in 2021
- Prevalence is highest in Southeast Asia (27.4%), the Middle East and North Africa (20.4%), and lowest in Europe (7.7%), reflecting underlying type 2 diabetes risk patterns
- In the United States, GDM prevalence increased from approximately 6% in 2000 to over 8% by 2020, driven by rising maternal obesity rates and advanced maternal age trends
- Adoption of IADPSG one-step criteria increases diagnosed prevalence by approximately 2–3-fold versus Carpenter-Coustan criteria, as IADPSG requires only one abnormal OGTT value
- GDM accounts for approximately 86% of all diabetes in pregnancy; pre-gestational type 1 and type 2 diabetes account for the remainder
- Women with prior GDM have a 50–70% cumulative 10-year risk of developing type 2 diabetes
- Offspring of GDM pregnancies have a 3–8-fold increased lifetime risk of obesity and type 2 diabetes compared to offspring of normoglycemic pregnancies
3. Pathophysiology
Normal Metabolic Adaptation to Pregnancy
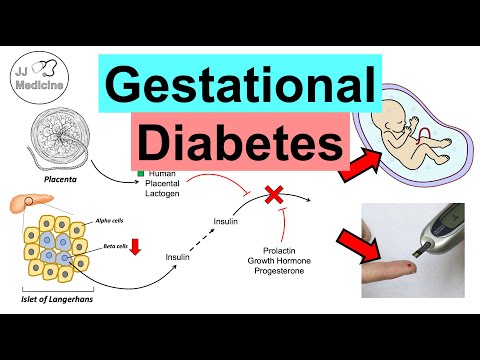
Normal pregnancy induces a progressive state of insulin resistance that peaks in the third trimester, mediated by placental hormones and cytokines — principally human placental lactogen (hPL), placental growth hormone (PGH), prolactin, progesterone, cortisol, and tumor necrosis factor-alpha (TNF-alpha). This physiological insulin resistance ensures preferential glucose availability to the fetus (glucose crosses the placenta via facilitated diffusion down a concentration gradient). To maintain maternal normoglycemia, pancreatic beta-cell mass expands by approximately 10–15% and insulin secretion increases 200–250% in normal pregnancy — a compensatory response to the 2–3-fold increase in insulin resistance measured by hyperinsulinemic euglycemic clamp.
GDM Pathogenesis
GDM develops in women who cannot mount sufficient beta-cell compensatory hyperinsulinemia in the face of pregnancy-induced insulin resistance. Two overlapping defects are present:
Insulin Resistance
Women who develop GDM demonstrate greater pre-pregnancy and early pregnancy insulin resistance than controls, suggesting a pre-existing metabolic vulnerability. Molecular mechanisms include: impaired insulin receptor substrate-1 (IRS-1) tyrosine phosphorylation; reduced PI3K/Akt signaling in skeletal muscle and adipose tissue; elevated plasma free fatty acids from increased lipolysis impairing myocellular glucose uptake (lipotoxicity); and inflammatory cytokines (IL-6, TNF-alpha, CRP) from visceral adipose tissue further impairing insulin signaling at the receptor and post-receptor level.
Beta-Cell Dysfunction
Women with GDM have a 50% reduction in the acute insulin response to glucose compared to normoglycemic pregnant women, and reduced disposition index (beta-cell function relative to insulin resistance). Defects include impaired first-phase insulin secretion, reduced glucose-stimulated insulin secretion, and inappropriate glucagon secretion. Beta-cell dysfunction in GDM is partly reversible postpartum but incompletely — residual beta-cell deficit persists and, combined with ongoing insulin resistance risk factors (obesity, sedentary lifestyle), explains the high lifetime T2DM conversion rate.
Placental Mechanisms
The placenta is not merely a passive bystander — GDM placentas exhibit abnormal nutrient transport. Upregulation of GLUT1, GLUT3, and GLUT9 glucose transporters in the syncytiotrophoblast enhances fetal glucose delivery in the setting of maternal hyperglycemia. Leptin resistance, placental insulin-like growth factor (IGF) signaling alterations, and increased placental mTOR pathway activity promote fetal anabolism, contributing to macrosomia. Maternal hyperglycemia stimulates fetal hyperinsulinemia (Pedersen hypothesis), which acts as a fetal growth factor independently of maternal IGF levels.
4. Etiology and Risk Factors
Major Risk Factors
- Pre-pregnancy obesity: BMI above 30 kg/m² confers approximately 4-fold increased GDM risk; morbid obesity (BMI above 40) confers 8–10-fold increased risk; approximately 60% of GDM cases occur in obese women
- Prior GDM: 33–50% recurrence rate in subsequent pregnancies; increases with higher pre-pregnancy BMI and shorter inter-pregnancy interval
- Family history of type 2 diabetes: First-degree relative with T2DM doubles GDM risk; reflects shared genetic susceptibility in beta-cell function loci (TCF7L2, KCNJ11, CDKN2A/B)
- Advanced maternal age: Risk increases approximately 6% per additional year of age after 25; women aged 35–39 have approximately 2-fold risk versus women aged 25–29; declining beta-cell reserve with aging
- Ethnicity: Asian (South, East, Southeast), Middle Eastern, Hispanic, and Black women have substantially higher GDM risk than non-Hispanic White women at equivalent BMI; different body fat distribution (higher visceral-to-subcutaneous fat ratio at lower BMI in Asian women) is a key mechanism
- Polycystic ovary syndrome (PCOS): 2.5–4-fold increased GDM risk; shared underlying insulin resistance, hyperandrogenism, and beta-cell dysfunction; women with PCOS should be offered early GDM screening at first antenatal visit
- Prior delivery of a macrosomic infant (birth weight above 4000–4500 g): Indicates likely prior undiagnosed GDM
- Unexplained stillbirth or recurrent miscarriage: May indicate previously unrecognized dysglycemia
Additional Risk Factors
- Glucocorticoid therapy during pregnancy (betamethasone for fetal lung maturity causes transient but significant hyperglycemia)
- Multiple gestation (higher hPL and placental hormonal burden)
- Polyhdramnios (associated with uncontrolled GDM)
- Pre-pregnancy impaired fasting glucose or impaired glucose tolerance (pre-diabetes)
- Sedentary lifestyle and low physical activity levels
- Short sleep duration and obstructive sleep apnea (insulin resistance mediators)
5. Clinical Presentation
GDM is characteristically asymptomatic — this is the fundamental rationale for universal screening. The absence of classic diabetic symptoms (polyuria, polydipsia, polyphagia, weight loss) means GDM is almost always diagnosed through protocol-driven screening rather than symptomatic presentation.
Maternal Signs Suggestive of GDM
- Polyhydramnios: Excessive amniotic fluid volume (AFI above 24 cm or single deepest pocket above 8 cm on ultrasound) due to fetal osmotic diuresis from hyperglycemia; identified in up to 20% of GDM pregnancies
- Large-for-gestational-age (LGA) fetus: Estimated fetal weight above the 90th percentile for gestational age on ultrasound; abdominal circumference (AC) disproportionately large relative to head circumference suggests fetal macrosomia
- Recurrent candidiasis: Vaginal yeast infections facilitated by glucosuria
- Glucosuria: Present on urine dipstick; however, glucosuria in pregnancy has poor sensitivity (55%) and specificity for GDM due to physiologically reduced renal glucose threshold in pregnancy — not a reliable screening tool but warrants OGTT when detected
Symptomatic hyperglycemia (osmotic symptoms) in pregnancy should prompt immediate fasting glucose or HbA1c to exclude overt diabetes, which carries a substantially higher risk of congenital anomalies (if hyperglycemia was present peri-conceptionally) and pregnancy complications than GDM alone.
6. Diagnosis
Two-Step Approach (Carpenter-Coustan / ACOG)
Step 1 — 50g glucose challenge test (GCT): Non-fasting oral 50g glucose load; plasma glucose measured at 1 hour. Positive screen: glucose at or above 130–140 mg/dL (threshold varies by institution — 130 mg/dL has higher sensitivity, 140 mg/dL higher specificity). Women with a 1-hour glucose at or above 200 mg/dL are presumptively diagnosed with GDM and proceed to treatment without a formal OGTT. Performed at 24–28 weeks gestation; at first prenatal visit for high-risk women.
Step 2 — 100g 3-hour OGTT (Carpenter-Coustan thresholds): Performed after at least 3 days of unrestricted diet (150g carbohydrate per day) and an overnight fast of 8–14 hours. Diagnosis of GDM requires two or more of the following plasma glucose values to be met or exceeded:
- Fasting: 95 mg/dL (5.3 mmol/L)
- 1 hour: 180 mg/dL (10.0 mmol/L)
- 2 hours: 155 mg/dL (8.6 mmol/L)
- 3 hours: 140 mg/dL (7.8 mmol/L)
Note: The older NDDG (National Diabetes Data Group) thresholds are slightly higher and have largely been replaced by Carpenter-Coustan values in the United States.
Women with only one abnormal value on the 100g OGTT have intermediate risk and may benefit from individualized glycemic monitoring and dietary counseling.
One-Step Approach (IADPSG / WHO 2013)
Universal 75g 2-hour OGTT at 24–28 weeks, performed after an overnight fast of at least 8 hours (no prior GCT screen required). GDM is diagnosed if any single glucose value meets or exceeds:
- Fasting: 92 mg/dL (5.1 mmol/L)
- 1 hour: 180 mg/dL (10.0 mmol/L)
- 2 hours: 153 mg/dL (8.5 mmol/L)
The IADPSG thresholds were derived from the Hyperglycemia and Adverse Pregnancy Outcome (HAPO) study — a landmark multicenter prospective observational study of 23,316 pregnant women across 15 centers in 9 countries, which demonstrated a continuous graded association between maternal glucose levels (even below traditional diabetic thresholds) and adverse outcomes including birth weight above the 90th percentile, cord blood C-peptide above the 90th percentile (neonatal hyperinsulinemia), and primary cesarean section rate.
Additionally, the 75g OGTT at 24–28 weeks serves to identify overt diabetes in pregnancy when fasting glucose is at or above 126 mg/dL or 2-hour glucose is at or above 200 mg/dL — these women have pre-gestational diabetes requiring more intensive management.
Early screening (first trimester) is recommended for high-risk women (prior GDM, obesity, PCOS, prior macrosomic infant, family history of T2DM, South/East Asian women) using fasting glucose or HbA1c to identify overt pre-gestational diabetes or very early GDM.
Comparison of Screening Approaches
The two-step Carpenter-Coustan approach diagnoses approximately 5–6% of pregnancies as GDM; the IADPSG one-step approach diagnoses approximately 15–20%. The DECIDE trial (2023) and TOBOGM trial (2022) have provided important prospective randomized data comparing these approaches and their clinical impact on maternal and neonatal outcomes, informing ongoing debate between ACOG (two-step) and ADA/WHO/FIGO (one-step) professional society positions.
HbA1c in GDM
HbA1c is not recommended as a primary GDM diagnostic tool due to physiological hemodilution, increased red cell turnover, and iron deficiency anemia in pregnancy causing spuriously low values. However, an HbA1c at or above 6.5% (48 mmol/mol) in the first trimester indicates probable pre-gestational diabetes.
7. Treatment
Glycemic Targets
Optimal glycemic control is the cornerstone of GDM management. Target plasma glucose levels (consistent across ADA, ACOG, Diabetes UK, and FIGO guidelines):
- Fasting / pre-meal: below 95 mg/dL (5.3 mmol/L)
- 1-hour postprandial: below 140 mg/dL (7.8 mmol/L)
- 2-hour postprandial: below 120 mg/dL (6.7 mmol/L)
Self-monitoring of blood glucose (SMBG) with a calibrated glucometer is required initially four times daily (fasting and 1–2 hours after each main meal) to assess whether dietary measures achieve targets. Women requiring insulin may need more frequent monitoring including bedtime readings.
Medical Nutrition Therapy (MNT)
First-line treatment for all women with GDM. Goals: achieve glycemic targets, provide adequate nutrition for maternal and fetal health, achieve appropriate gestational weight gain (based on pre-pregnancy BMI), and avoid ketosis. Key principles:
- Total caloric intake individualized to pre-pregnancy BMI and gestational weight gain goals (IOM guidelines); typically 1800–2500 kcal/day; caloric restriction to 1600–1800 kcal/day in obese women (BMI above 30) reduces hyperglycemia without adverse fetal effects
- Carbohydrate distribution: 40–50% of total calories from carbohydrates, distributed across 3 meals and 2–4 snacks; emphasis on low glycemic index carbohydrates (legumes, whole grains, non-starchy vegetables); avoidance of sugar-sweetened beverages, refined starches, and high-GI foods
- Bedtime snack containing protein and complex carbohydrate (15–30g) reduces fasting glucose by blunting nocturnal glycogenolysis
- Protein: 20–25% of calories; fat: 30–35%; minimize saturated fat and trans-fat
- Structured dietary education by a registered dietitian with expertise in GDM; cultural adaptation of dietary advice improves adherence
Approximately 70–85% of women with GDM achieve glycemic targets on MNT alone and do not require pharmacological therapy.
Physical Activity
Aerobic exercise (150 minutes per week of moderate-intensity activity such as walking, swimming, stationary cycling) reduces postprandial glucose by 10–20% through non-insulin-mediated glucose uptake in skeletal muscle (GLUT4 translocation via AMPK signaling). Resistance training also improves insulin sensitivity. Post-meal walking for 15–30 minutes is particularly effective for postprandial hyperglycemia. Contraindications to exercise in pregnancy should be assessed (placenta previa, preterm labor risk, multiple gestation, severe hypertension).
Pharmacological Management — Indications
Pharmacotherapy is initiated when MNT and exercise fail to achieve glycemic targets within 1–2 weeks. General indications:
- Fasting glucose persistently above 95 mg/dL (5.3 mmol/L)
- 1-hour postprandial glucose persistently above 140 mg/dL or 2-hour postprandial above 120 mg/dL
- Ultrasound evidence of fetal macrosomia or polyhydramnios despite dietary targets being apparently met
Insulin Therapy
Insulin is the gold standard pharmacological agent for GDM — it does not cross the placenta in clinically meaningful amounts, has the longest safety record, and allows precise dose titration. Insulin selection:
- Fasting hyperglycemia: Intermediate-acting (NPH insulin) at bedtime is first-line — NPH peaks 6–8 hours after injection, targeting overnight glucose production; starting dose 0.1–0.2 units/kg; titrate up by 10–15% every 3 days until fasting target achieved. Long-acting analogs (insulin detemir — preferred over glargine due to more extensive pregnancy safety data) are alternatives with lower nocturnal hypoglycemia risk
- Postprandial hyperglycemia: Rapid-acting analogs — insulin aspart and insulin lispro have the most robust pregnancy safety data (Class B); both are FDA-approved in pregnancy. Administered immediately before meals; starting dose 1–2 units per 15g carbohydrate or empirically 2–4 units per meal, titrated based on 1–2-hour postprandial readings. Regular insulin (slower onset) may be used but is less convenient than analogs
- Total daily dose (TDD) calculation: Many clinicians use weight-based TDD (0.7–1.0 units/kg in the second trimester; 0.8–1.2 units/kg in the third trimester due to increasing insulin resistance), splitting 50% as basal and 50% as prandial in a basal-bolus regimen for women with mixed fasting and postprandial hyperglycemia
- Insulin requirements increase across gestation, typically requiring dose adjustments every 1–2 weeks in the third trimester
Metformin in GDM
Metformin (biguanide) is an oral hypoglycemic that reduces hepatic glucose output (via AMPK-mediated LKB1 activation), enhances peripheral insulin sensitivity, and decreases intestinal glucose absorption. It crosses the placenta, reaching fetal concentrations approximately 50% of maternal plasma levels — the long-term safety implications of in utero metformin exposure are an area of active investigation.
Clinical evidence: The MiG (Metformin in Gestational Diabetes) trial — the largest RCT (N=751) — demonstrated that metformin achieves similar glycemic control to insulin with lower maternal hypoglycemia, less gestational weight gain, higher patient satisfaction, and no increase in neonatal adverse outcomes. However, 46% of metformin-assigned women required supplemental insulin to achieve targets. Metformin does not increase congenital anomaly risk when started after organogenesis (14 weeks).
Current guidelines: ADA accepts metformin as an alternative to insulin for GDM when insulin is refused or unavailable; ACOG acknowledges metformin as a reasonable alternative. FIGO supports metformin as a first-line oral agent. Metformin is not approved by the FDA specifically for GDM but is used off-label widely. Concerns about long-term offspring metabolic outcomes (increased adiposity observed in MiG offspring follow-up at 2 and 9 years) have tempered enthusiasm and warrant ongoing study.
Glyburide (Glibenclamide) in GDM
Glyburide is a second-generation sulfonylurea that crosses the placenta (umbilical cord:maternal ratio approximately 0.7) and stimulates pancreatic insulin secretion. Once used widely in the United States as an oral alternative to insulin, glyburide has fallen out of favor following meta-analyses demonstrating higher rates of neonatal hypoglycemia, macrosomia, and NICU admission versus insulin; it is no longer recommended as a first-line oral agent by ADA, ACOG, or FIGO guidelines.
Obstetric Management and Fetal Surveillance
- Ultrasound surveillance: Fetal growth scan at 28–32 weeks to assess for macrosomia (estimated fetal weight, AC above 90th percentile) and polyhydramnios; additional growth scan at 34–36 weeks in women with poor glycemic control or evidence of macrosomia
- Fetal well-being: Non-stress testing (NST) or biophysical profile (BPP) weekly from 32–34 weeks for women requiring pharmacotherapy or with suboptimal glycemic control; practices vary by institution
- Timing of delivery: GDM well-controlled on diet/exercise alone: delivery at 39 0/7 to 40 6/7 weeks recommended (risk of stillbirth does not begin to exceed risk of neonatal complications from delivery before 39 weeks). GDM requiring insulin/metformin: delivery at 38 0/7 to 39 6/7 weeks. Delivery before 38 weeks for obstetric indications (preeclampsia, poor glycemic control with macrosomia, non-reassuring fetal surveillance)
- Route of delivery: Cesarean delivery is recommended when estimated fetal weight exceeds 4500g (4000g in some guidelines) to reduce shoulder dystocia risk; otherwise, vaginal delivery is appropriate
- Intrapartum glucose management: Maintain maternal glucose 70–110 mg/dL during labor with hourly glucose monitoring; insulin drip protocol for women on insulin therapy; glucose-containing IV fluids only if needed for hypoglycemia prevention
Postpartum Management
Insulin requirements drop dramatically at delivery with expulsion of the placenta and resolution of placental hormone-driven insulin resistance. Insulin is typically discontinued immediately postpartum in diet-controlled or insulin-requiring GDM. Fasting glucose should be checked 24–48 hours postpartum — persistent hyperglycemia indicates pre-gestational type 2 or type 1 diabetes.
Postpartum OGTT: The 75g 2-hour OGTT at 4–12 weeks postpartum (ADA, WHO) is the gold standard test to reclassify glucose tolerance status and detect persistent type 2 diabetes, impaired fasting glucose (IFG), or impaired glucose tolerance (IGT) in women with prior GDM. HbA1c is an alternative when OGTT is not feasible but has lower sensitivity for detecting pre-diabetes and early T2DM postpartum. Categories:
- Normal: fasting below 100 mg/dL and 2-hour below 140 mg/dL
- Impaired fasting glucose (IFG): fasting 100–125 mg/dL
- Impaired glucose tolerance (IGT): 2-hour 140–199 mg/dL
- Type 2 diabetes: fasting at or above 126 mg/dL or 2-hour at or above 200 mg/dL
Unfortunately, postpartum OGTT completion rates are poor (30–50% in most health systems). Women who have normal postpartum OGTT should undergo repeat glucose assessment every 1–3 years using fasting glucose or HbA1c (or OGTT for highest risk women) given the 50–70% lifetime T2DM conversion rate.
Breastfeeding should be encouraged — it reduces maternal insulin resistance, promotes weight loss, improves infant microbiome composition, and is associated with 40–50% lower T2DM risk in women with prior GDM in observational studies.
Lifestyle modification: Women with pre-diabetes diagnosed postpartum should be offered intensive lifestyle modification programs (modeled on the Diabetes Prevention Program — 7% weight loss, 150 minutes/week physical activity) and consideration of metformin if high-risk (BMI above 35, HbA1c above 6.0%, prior GDM diagnosed before 24 weeks, or prior rapid conversion to T2DM).
8. Complications
Maternal Complications
- Preeclampsia: GDM increases preeclampsia risk 2.5–3-fold; shared pathophysiological mechanisms include insulin resistance, endothelial dysfunction, and angiogenic imbalance
- Cesarean delivery: Rate approximately 30–40% in GDM pregnancies versus 20–25% in normoglycemic controls; driven by macrosomia, labor induction failure, and clinical concern about shoulder dystocia
- Postpartum hemorrhage: Risk increased with macrosomic delivery and associated uterine overdistension and atony
- Type 2 diabetes: 50–70% lifetime risk; risk highest in women with early GDM diagnosis (before 24 weeks), severe hyperglycemia, need for insulin therapy, high BMI, and non-White ethnicity
- Recurrent GDM: 33–50% in subsequent pregnancies
- Cardiovascular disease: Women with prior GDM have increased long-term risk of hypertension, dyslipidemia, metabolic syndrome, and atherosclerotic cardiovascular disease independent of T2DM conversion
Fetal and Neonatal Complications
- Macrosomia: Birth weight above 4000g or above 90th percentile (LGA); occurs in approximately 20–25% of GDM pregnancies; driven by fetal hyperinsulinemia stimulating adipocyte proliferation and hepatic glycogen/fat deposition; central/abdominal fat excess is disproportionate to head size
- Shoulder dystocia and birth trauma: Brachial plexus injury (Erb's palsy), clavicle or humerus fracture, hypoxic-ischemic encephalopathy; macrosomia increases shoulder dystocia risk 4–12-fold
- Neonatal hypoglycemia: Hyperinsulinemia persists after delivery but maternal glucose supply abruptly ceases; neonatal glucose below 40–47 mg/dL (threshold varies by center) in up to 25% of infants of diabetic mothers; managed with early feeding and IV dextrose if severe
- Respiratory distress syndrome (RDS): Fetal hyperinsulinemia delays fetal lung maturation by inhibiting cortisol-mediated surfactant synthesis; risk is increased particularly with early delivery before 38–39 weeks
- Polycythemia and hyperbilirubinemia: Fetal hyperinsulinemia increases erythropoiesis; neonatal polycythemia leads to hyperviscosity and indirect hyperbilirubinemia requiring phototherapy
- Perinatal mortality: Stillbirth risk is increased approximately 2–3-fold with poorly controlled GDM; mechanism involves fetal hypoxia from macrosomic metabolic demands, cord accident, or uteroplacental insufficiency
- Long-term offspring consequences: GDM offspring have 3–8-fold increased risk of childhood and adolescent obesity, metabolic syndrome, and type 2 diabetes; proposed mechanisms include epigenetic programming of hypothalamic appetite regulation, pancreatic beta-cell development, and hepatic glucose metabolism by in utero hyperglycemic exposure
9. Prognosis
With appropriate diagnosis and management, the prognosis for GDM is generally excellent for both mother and infant. Women who achieve glycemic targets have neonatal outcomes and cesarean delivery rates approaching those of normoglycemic women. The ACHOIS (Australian Carbohydrate Intolerance Study in Pregnant Women) and US NICHD trials demonstrated significant reductions in macrosomia, shoulder dystocia, and serious perinatal morbidity through GDM treatment versus routine care.
Long-term maternal prognosis is shaped predominantly by T2DM conversion risk. Women who achieve and maintain normal weight postpartum and adopt regular physical activity and healthy diet have a 50–70% relative risk reduction for T2DM development versus those who do not implement lifestyle change. Metformin in the postpartum period reduces T2DM development by approximately 35% in women with prior GDM and concurrent pre-diabetes (DPP trial subgroup data).
Offspring long-term prognosis is an emerging concern — GDM constitutes a form of fetal metabolic programming that increases susceptibility to obesity and metabolic disease across the life course, potentially perpetuating intergenerational cycles of metabolic risk.
10. Prevention
GDM prevention is an area of active research and public health importance given rising global prevalence:
- Pre-conception weight management: Achieving healthy BMI before pregnancy is the single most impactful modifiable risk factor; 5–10% weight loss in overweight/obese women pre-conception substantially reduces GDM risk
- Physical activity: Regular aerobic and resistance exercise before and during pregnancy reduces GDM risk by approximately 30–38% in meta-analyses; at least 150 minutes/week moderate-intensity activity recommended throughout pregnancy
- Dietary modification: Mediterranean diet, DASH diet, and low-glycemic index diet adherence before and during pregnancy reduce GDM risk; evidence strongest for reducing simple carbohydrate and sugar-sweetened beverage intake
- Metformin prophylaxis: The EMPOWaR and MiG TOFU trials and post-hoc DPP analyses suggest metformin initiated pre-conception or in early pregnancy may reduce GDM incidence in high-risk women (prior GDM, PCOS, obesity); not yet in routine guidelines but supported for women with PCOS on metformin who conceive
- Inositol supplementation: Myo-inositol and D-chiro-inositol (second messengers in insulin signaling) have shown GDM risk reduction in randomized trials among high-risk women; meta-analysis suggests approximately 50% reduction in GDM incidence with myo-inositol 4g/day; larger trials ongoing
- Vitamin D supplementation: Observational data suggest vitamin D deficiency is associated with GDM risk; randomized trials of supplementation have produced mixed results; maintaining vitamin D sufficiency is reasonable
- Intergenerational prevention: Breastfeeding of GDM offspring reduces infant obesity risk; healthy lifestyle in GDM offspring from childhood reduces their adult metabolic disease risk
11. Recent Research
- TOBOGM trial (2022): Landmark RCT (N=4061) comparing early GDM treatment (diagnosis and treatment of GDM at less than 20 weeks using IADPSG criteria) versus standard treatment at 24–28 weeks in high-risk women. Early treatment significantly reduced a composite of adverse perinatal outcomes (preeclampsia, gestational hypertension, birth trauma, NICU admission, neonatal respiratory distress, death) relative risk 0.82, without increasing small-for-gestational-age birth. Supports early universal OGTT in high-risk women.
- DECIDE trial (2023): Multicenter RCT comparing one-step (IADPSG) versus two-step (Carpenter-Coustan) GDM screening — primary results and health economic analyses are providing evidence on which screening strategy reduces adverse perinatal outcomes cost-effectively at population scale.
- Continuous glucose monitoring (CGM) in GDM: The CONCEPTT trial demonstrated CGM benefit in pre-gestational T1DM; BUMP trial (2021) of CGM in GDM showed CGM did not improve glycemic control or perinatal outcomes over SMBG in diet-controlled GDM, likely because CGM-identified excursions did not change management in the study protocol. Ongoing trials integrating CGM with real-time clinical decision support may demonstrate benefit in insulin-requiring GDM.
- GLP-1 receptor agonists in pregnancy: Currently contraindicated in pregnancy due to limited safety data, but preclinical and registry data are being accumulated; given the efficacy of GLP-1 agonists in obesity and T2DM, their potential role post-delivery for T2DM prevention in women with prior GDM is an active area.
- Microbiome and GDM: Gut and vaginal microbiome alterations have been identified in GDM patients; reduced Akkermansia muciniphila abundance and altered short-chain fatty acid (SCFA) production impair GLP-1 secretion and insulin sensitivity; probiotic RCTs (Lactobacillus and Bifidobacterium) show modest fasting glucose and HbA1c reductions in GDM.
- Epigenetic programming and offspring outcomes: DOHAD (Developmental Origins of Health and Disease) research demonstrates that in utero hyperglycemic exposure induces epigenetic modifications (DNA methylation, histone acetylation) in fetal pancreatic, adipose, and hepatic cells detectable in cord blood and persisting into childhood, offering potential biomarkers and intervention targets for intergenerational prevention.
- Pharmacogenomics of metformin in GDM: Variants in SLC22A1 (OCT1) and SLC22A2 (OCT2) transporter genes influence metformin placental transfer and efficacy; pharmacogenomic profiling may eventually guide personalized GDM pharmacotherapy selection.
12. References
- HAPO Study Cooperative Research Group; Metzger BE, Lowe LP, Dyer AR, et al. Hyperglycemia and adverse pregnancy outcomes. N Engl J Med. 2008;358(19):1991–2002.
- International Association of Diabetes and Pregnancy Study Groups Consensus Panel. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33(3):676–682.
- Carpenter MW, Coustan DR. Criteria for screening tests for gestational diabetes. Am J Obstet Gynecol. 1982;144(7):768–773.
- American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 230: Gestational Diabetes Mellitus. Obstet Gynecol. 2021;137(2):e114–e127.
- American Diabetes Association. 15. Management of Diabetes in Pregnancy: Standards of Medical Care in Diabetes — 2024. Diabetes Care. 2024;47(Suppl 1):S282–S294.
- Rowan JA, Hague WM, Gao W, Battin MR, Moore MP; MiG Trial Investigators. Metformin versus insulin for the treatment of gestational diabetes. N Engl J Med. 2008;358(19):2003–2015.
- Crowther CA, Hiller JE, Moss JR, McPhee AJ, Jeffries WS, Robinson JS; ACHOIS Trial Group. Effect of treatment of gestational diabetes mellitus on pregnancy outcomes. N Engl J Med. 2005;352(24):2477–2486.
- Landon MB, Spong CY, Thom E, et al. A multicenter, randomized trial of treatment for mild gestational diabetes. N Engl J Med. 2009;361(14):1339–1348.
- Simmons D, Immanuel J, Hague WM, et al. Treatment of gestational diabetes mellitus diagnosed early in pregnancy. N Engl J Med. 2023;388(23):2132–2144.
- Farrar D, Simmonds M, Bryant M, et al. Hyperglycaemia and risk of adverse perinatal outcomes: systematic review and meta-analysis. BMJ. 2016;354:i4694.
- Kim C, Newton KM, Knopp RH. Gestational diabetes and the incidence of type 2 diabetes: a systematic review. Diabetes Care. 2002;25(10):1862–1868.
- Bellamy L, Casas JP, Hingorani AD, Williams D. Type 2 diabetes mellitus after gestational diabetes: a systematic review and meta-analysis. Lancet. 2009;373(9677):1773–1779.
- Dabelea D, Mayer-Davis EJ, Lamichhane AP, et al. Association of intrauterine exposure to maternal diabetes and obesity with type 2 diabetes in youth: the SEARCH Case-Control Study. Diabetes Care. 2008;31(7):1422–1426.
- Catalano PM, McIntyre HD, Cruickshank JK, et al. The hyperglycemia and adverse pregnancy outcome study: associations of GDM and obesity with pregnancy outcomes. Diabetes Care. 2012;35(4):780–786.
- Xu T, Zhang D, Dong H, Wang J. Effects of exercise intervention on gestational diabetes mellitus: a meta-analysis of randomized controlled trials. J Diabetes Complications. 2020;34(7):107597.
- Bain E, Crane M, Tieu J, Han S, Crowther CA, Middleton P. Diet and exercise interventions for preventing gestational diabetes mellitus. Cochrane Database Syst Rev. 2015;4:CD010443.
- Moyer VA; U.S. Preventive Services Task Force. Screening for gestational diabetes mellitus: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2014;160(6):414–420.
- Metzger BE, Gabbe SG, Persson B, et al. International association of diabetes and pregnancy study groups recommendations on the diagnosis and classification of hyperglycemia in pregnancy. Diabetes Care. 2010;33(3):676–682.
- Blumer I, Hadar E, Hadden DR, et al. Diabetes and pregnancy: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2013;98(11):4227–4249.
Research Papers
The following PubMed topic searches aggregate the current peer-reviewed literature. Each link opens a live PubMed query — results update as new studies are indexed.
- PubMed — gestational diabetes mellitus
- PubMed — oral glucose tolerance test pregnancy
- PubMed — gestational diabetes screening
- PubMed — metformin gestational diabetes
- PubMed — insulin gestational diabetes
- PubMed — macrosomia gestational diabetes
- PubMed — gestational diabetes postpartum
- PubMed — IADPSG criteria
- PubMed — gestational diabetes diet
- PubMed — gestational diabetes exercise
- PubMed — future type 2 diabetes gestational
- PubMed — HAPO study
Connections
- Preeclampsia
- Insulin Resistance
- Diabetes
- Obesity
- Polycystic Ovary Syndrome
- Metabolic Syndrome
- Vitamin D3
- Hashimoto's Thyroiditis
- Magnesium
- Pregnancy and Hashimoto's
- Chromium
- Continuous Glucose Monitor
- Inositol
- Fasting
- Hypertension
- Cardiovascular Disease
- GLP-1 Receptor Agonists
- Infertility
Featured Videos

Gestational Diabetes Mellitus (Pregnancy) Nursing Care, Symptoms for Maternity Nursing

Gestational Diabetes : Myths & Facts | Signs, Symptoms, Diagnosis & Treatment

Diabetes: Pathophysiology, Signs/Symptoms, Labs, Treatment & more - Medical-Surgical | @LevelUpRN
Gestational Diabetes (Pregnancy Diabetes) | Causes, Risk Factors, Symptoms, Consequences, Treatment

Gestational Diabetes: Everything You Need to Know

Gestational Diabetes Mellitus (GDM)

Complications: Gestational Diabetes Mellitus (GDM) - Maternity Nursing | @LevelUpRN

Gestational Diabetes During Pregnancy Symptoms and Signs

Diabetes mellitus (type 1, type 2) & diabetic ketoacidosis (DKA)