Myocarditis
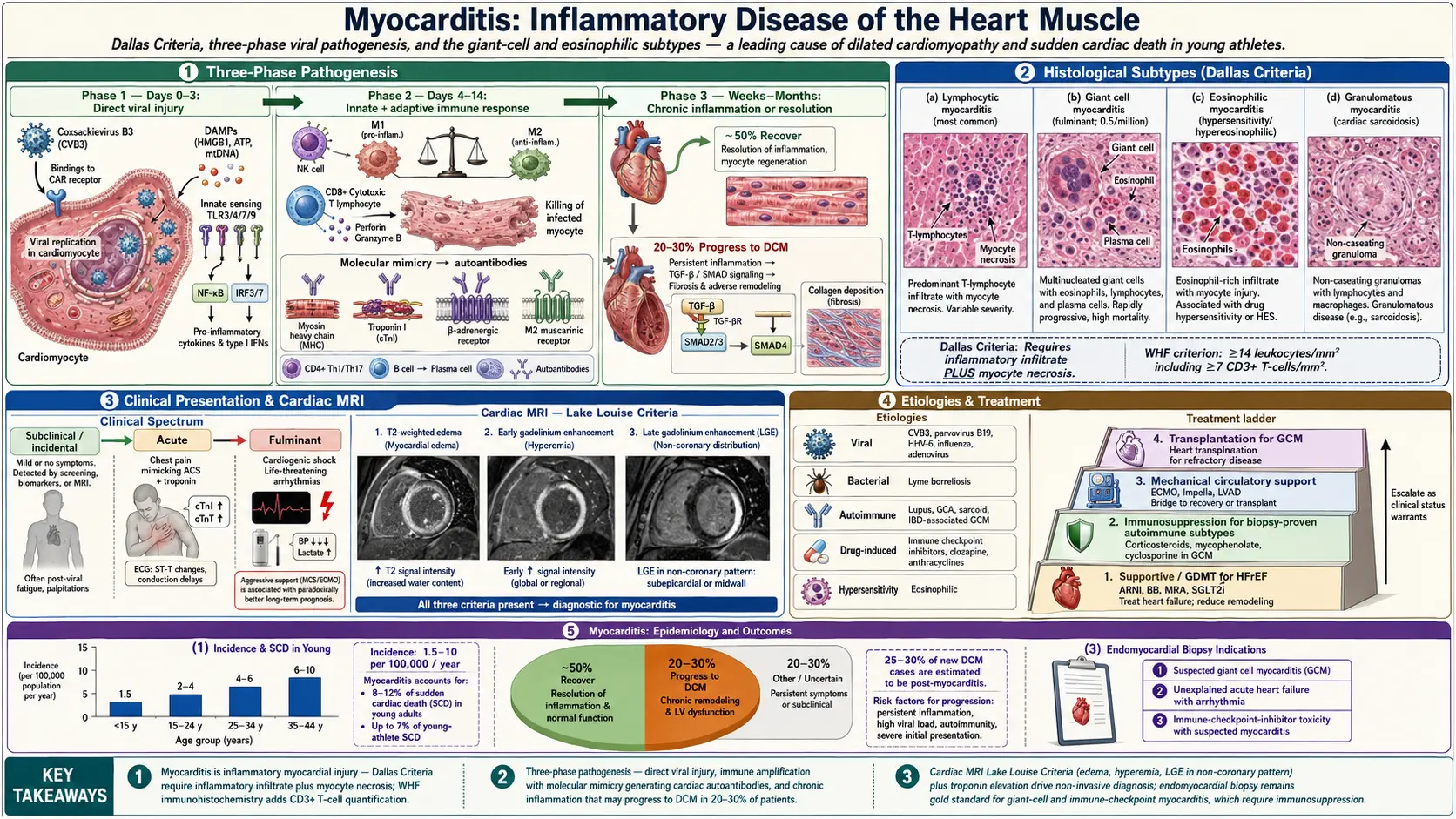
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Myocarditis is an inflammatory disease of the myocardium diagnosed by established histological, immunological, and immunohistochemical criteria. It is defined by the Dallas Criteria as an inflammatory infiltrate of the myocardium with associated myocyte necrosis or injury not characteristic of an ischemic etiology. The World Heart Federation (WHF) Task Force later introduced broader immunohistochemical criteria: ≥14 leukocytes/mm² including ≥7 CD3+ T-lymphocytes/mm² on endomyocardial biopsy (EMB).
Myocarditis encompasses a wide clinical spectrum — from subclinical disease detected incidentally to fulminant myocarditis with cardiogenic shock and life-threatening arrhythmias. It is a leading cause of dilated cardiomyopathy (25–30% of new-onset DCM cases) and sudden cardiac death in young adults and athletes.
Classification by clinical course:
- Fulminant myocarditis: Abrupt onset with severe hemodynamic compromise; often follows distinct viral prodrome; paradoxically better long-term prognosis with aggressive supportive care
- Acute myocarditis: Non-fulminant; <3 months; variable presentation
- Subacute/chronic myocarditis: >3 months; may progress to DCM
Classification by etiologic subtype (Dallas histopathologic criteria):
- Lymphocytic myocarditis (most common)
- Giant cell myocarditis (GCM)
- Eosinophilic myocarditis
- Granulomatous myocarditis (cardiac sarcoidosis)
2. Epidemiology
Myocarditis has a reported incidence of approximately 1.5–10 per 100,000 population per year, though true incidence is likely much higher given frequent subclinical presentation and underdiagnosis. Autopsy studies of sudden cardiac death in young adults identify myocarditis in 8–12% of cases. In series of young athletes with sudden cardiac death, myocarditis accounts for up to 7% of cases.
Myocarditis predominantly affects young adults (20–40 years), with a 3:1 male predominance — likely reflecting greater male susceptibility to viral-induced cardiac inflammation mediated by sex hormone effects on immune responses. The condition occurs at all ages, including infants and children (neonatal myocarditis is particularly severe).
Giant cell myocarditis, while rare (incidence 0.5 per million), is notable for its fulminant course and high mortality without cardiac transplantation.
3. Pathophysiology
The pathogenesis of viral myocarditis proceeds in three phases:
Phase 1: Direct Viral Injury (Days 0–3)
Cardiotropic viruses (e.g., Coxsackievirus B3 [CVB3]) enter cardiomyocytes via specific surface receptors — CVB3 binds the coxsackievirus-adenovirus receptor (CAR) — and replicate within cardiomyocytes. Direct cytopathic effects cause myocyte necrosis and apoptosis, releasing damage-associated molecular patterns (DAMPs: HMGB1, heat shock proteins, mitochondrial DNA). Pattern recognition receptors (Toll-like receptors TLR3, TLR4, TLR7, TLR9; RIG-I; MDA5) detect viral nucleic acids, activating NF-κB and IRF3/7 signaling pathways with consequent interferon-α/β production.
Phase 2: Innate and Adaptive Immune Response (Days 4–14)
Activated innate immune cells (natural killer cells, macrophages, dendritic cells) infiltrate the myocardium, amplifying inflammatory damage. Macrophages are categorized as pro-inflammatory M1 phenotype (iNOS, TNF-α production causing myocyte injury) and anti-inflammatory M2 phenotype (IL-10, TGF-β, myocardial repair). An imbalanced M1/M2 ratio perpetuates inflammation.
Adaptive immune response: Virus-specific CD8+ cytotoxic T lymphocytes (CTL) kill virus-infected cardiomyocytes. Crucially, molecular mimicry and bystander activation generate cross-reactive T cells and autoantibodies targeting cardiac proteins (myosin heavy chain, troponin I, beta-adrenergic receptors, L-type calcium channels, M2 muscarinic receptors). This autoimmune response persists after viral clearance, driving chronic inflammation.
Phase 3: Chronic Inflammation or Resolution (Weeks to Months)
In most patients (~50%), the immune response resolves and myocardial function recovers. In 20–30%, persistent autoimmune inflammation leads to progressive myocardial fibrosis (TGF-β/SMAD pathway), LV remodeling, and dilated cardiomyopathy. Viral RNA persistence (detected by PCR in 30–50% of chronic myocarditis biopsies) may also sustain chronic immune activation.
Giant Cell Myocarditis Pathophysiology
GCM is characterized by multinucleated giant cells (fused macrophages) with surrounding eosinophils, lymphocytes, and plasma cells. The pathophysiology involves T-cell-mediated autoimmune destruction of cardiomyocytes; 20% of GCM cases are associated with autoimmune diseases (inflammatory bowel disease, thymoma, thyroiditis). The inflammatory infiltrate is extensive and multifocal, rapidly destroying the ventricular myocardium.
4. Etiology and Risk Factors
Infectious Causes
- Viral (most common, 50–70% of biopsied cases):
- Enteroviruses: Coxsackievirus B1–B5 (historically most common in Western countries), echovirus, poliovirus
- Adenovirus types 2 and 5
- Parvovirus B19: Increasingly recognized; infects endothelial cells; positive in 30–50% of myocarditis biopsies
- Human herpesvirus 6 (HHV-6): Often co-detected with PVB19
- Epstein-Barr virus (EBV), cytomegalovirus (CMV), herpes simplex virus
- Influenza A/B; respiratory syncytial virus
- HIV: Direct cardiomyocyte invasion and opportunistic infections; myocarditis in 30–50% of HIV autopsy series
- Hepatitis C virus
- Bacterial: Borrelia burgdorferi (Lyme carditis; predominantly conduction system involvement), Corynebacterium diphtheriae (diphtheria myocarditis), Staphylococcus aureus, Clostridium perfringens, Treponema pallidum (syphilitic aortitis/myocarditis)
- Protozoan: Trypanosoma cruzi (Chagas disease — leading cause of myocarditis in Latin America affecting 8 million people; chronic chagasic cardiomyopathy is the primary cause of heart failure in endemic regions); Toxoplasma gondii
- Fungal: Aspergillus, Candida (immunocompromised hosts)
- Parasitic: Trichinella spiralis, Echinococcus
Immune-Mediated (Non-Infectious) Causes
- Hypersensitivity/eosinophilic myocarditis: Drug reactions (clozapine — most common; amphotericin B, dobutamine, tetanus toxoid); parasitic infections; idiopathic hypereosinophilic syndrome
- Giant cell myocarditis: Idiopathic; associated with systemic autoimmune diseases in 20%
- Granulomatous myocarditis: Cardiac sarcoidosis; giant cell arteritis
- Systemic autoimmune diseases: Systemic lupus erythematosus, polymyositis/dermatomyositis, systemic sclerosis, rheumatoid arthritis
- Checkpoint inhibitor-associated myocarditis: Anti-PD-1 (pembrolizumab, nivolumab), anti-PD-L1, anti-CTLA-4 (ipilimumab); rates 0.09–1.14%; fulminant course; mortality 25–50%
- Peripartum myocarditis
Toxic Causes
- Cocaine (catecholamine-mediated myocardial injury, coronary vasospasm)
- Alcohol (high-dose acute cardiac toxicity)
- Anthracycline chemotherapy
- Carbon monoxide poisoning
- Spider and scorpion venoms
Risk Factors
- Male sex (3:1 predominance in viral myocarditis)
- Young age (peak incidence 20–40 years)
- Immunosuppression (HIV, organ transplant, chemotherapy)
- Pregnancy and postpartum period
- Competitive athletics (may unmask subclinical myocarditis)
- Autoimmune predisposition
5. Clinical Presentation
Clinical Syndromes
Myocarditis manifests across four principal clinical syndromes:
- Acute chest pain syndrome (mimicking acute MI): Sharp, pleuritic chest pain; ST-segment elevation on ECG; elevated troponin; often in young patients following viral upper respiratory illness 1–3 weeks prior; coronary angiography reveals normal or non-obstructive coronary arteries
- New-onset heart failure/DCM: Exertional dyspnea, reduced exercise tolerance, orthopnea; acute or subacute LV dysfunction; may present as newly diagnosed cardiomyopathy
- Life-threatening arrhythmia or sudden cardiac death: Ventricular tachycardia, ventricular fibrillation, complete heart block (Lyme myocarditis); may be first presentation in previously healthy individuals
- Cardiogenic shock (fulminant myocarditis): Rapid hemodynamic deterioration within days of viral prodrome; biventricular failure; requires mechanical circulatory support
Prodromal Symptoms
The majority of patients (60–80%) report an antecedent viral illness 2–4 weeks before cardiac symptoms: fever, myalgia, upper respiratory infection (cough, rhinorrhea, sore throat), or gastrointestinal symptoms (nausea, vomiting, diarrhea).
Physical Examination
- Tachycardia (often disproportionate to fever)
- Fever
- New S3 or S4 gallop
- Soft mitral regurgitation murmur (from LV dilation)
- Pericardial friction rub (in myopericarditis)
- Elevated JVP, peripheral edema, pulmonary crackles (in heart failure)
- Hypotension, narrow pulse pressure, cool extremities (cardiogenic shock)
6. Diagnosis
Electrocardiography
ECG is frequently abnormal (50–80%) but non-specific:
- ST-segment elevation (diffuse, not territory-specific), ST depression, T-wave inversions
- New LBBB or RBBB
- AV block (first-, second-, third-degree) — hallmark of Lyme carditis
- Prolonged QTc interval
- Ventricular tachycardia/fibrillation
- Low voltage in pericardial effusion
Biomarkers
- High-sensitivity troponin I or T: Elevated in 50–80% of acute myocarditis; magnitude correlates with degree of myocyte necrosis; normal troponin does not exclude myocarditis
- BNP/NT-proBNP: Elevated with LV dysfunction; correlates with symptom severity and prognosis
- CK-MB: Less sensitive and specific than troponin
- ESR, CRP: Elevated in inflammatory phase; non-specific
- CBC: Leukocytosis with eosinophilia (eosinophilic/hypersensitivity myocarditis)
- Viral serologies: IgM titers for CVB, adenovirus, parvovirus B19, EBV, CMV, influenza; 4-fold rise in paired acute/convalescent sera; low sensitivity for current infection
- Serum autoantibodies: Anti-heart antibodies (anti-myosin, anti-alpha-myosin), anti-beta1-adrenergic receptor antibodies; research use; not routinely recommended
Echocardiography
Essential for: LV/RV function assessment; wall motion abnormalities (global or segmental); pericardial effusion; intracardiac thrombus; complications (MR from papillary muscle dysfunction). Echocardiographic findings are non-specific; a normal echo does not exclude myocarditis. Severe LV dysfunction (LVEF <35%), RV dysfunction, and large effusion predict worse prognosis.
Cardiac Magnetic Resonance Imaging (CMR)
CMR is the preferred non-invasive diagnostic modality for suspected myocarditis (class I recommendation, ESC 2013 position statement). The Lake Louise Criteria (updated 2018) define CMR criteria for myocardial inflammation:
T1-based markers:
- Elevated native T1 (reflects edema, hyperemia, and necrosis)
- Elevated extracellular volume fraction (ECV >27–30%)
- Early gadolinium enhancement (EGE) — hyperemia/capillary leak
T2-based markers:
- Elevated native T2 (reflects myocardial edema)
- T2 ratio (T2-STIR): Myocardial T2 signal vs. skeletal muscle
Late gadolinium enhancement (LGE): Myocardial fibrosis/necrosis — typically non-ischemic pattern: epicardial or mid-wall enhancement in the lateral wall (subepicardial LGE in lateral wall or inferior/inferolateral wall is the most common LGE pattern in viral myocarditis); distinguishes myocarditis from ischemic MI (subendocardial to transmural in coronary territory distribution).
Diagnosis of myocarditis is supported when ≥1 T1-based and ≥1 T2-based criteria are met, OR when LGE is present with one T1 or T2-based criterion.
Endomyocardial Biopsy (EMB)
EMB remains the gold standard for histopathologic and immunohistochemical diagnosis but is underutilized due to sampling error (<50% sensitivity for focal myocarditis), procedure complications, and the diagnostic utility of CMR. Indications for EMB:
- New-onset heart failure <2 weeks with hemodynamic compromise (to diagnose giant cell myocarditis or eosinophilic myocarditis — both requiring specific treatment)
- New-onset heart failure 2 weeks to 3 months with new ventricular arrhythmia or Mobitz II or complete heart block unresponsive to treatment
- Heart failure 3 months to 12 months with dilated CM with arrhythmia or failure to respond to GDMT
- When specific diagnosis would change management (GCM, cardiac sarcoidosis, eosinophilic myocarditis, Chagas disease)
Histopathologic findings: Dallas criteria (inflammatory infiltrate + myocyte necrosis); immunohistochemistry (CD3+, CD68+ macrophage quantification); viral PCR (parvovirus B19, HHV-6, adenovirus, enteroviruses) from biopsy tissue; electron microscopy for storage diseases.
Differential Diagnosis
- Acute coronary syndrome (ACS): Differentiated by coronary angiography; LGE distribution
- Takotsubo (stress) cardiomyopathy: Apical ballooning pattern; emotional or physical stress trigger; predominantly postmenopausal women; normal coronary arteries
- Acute pericarditis: Predominantly pericardial inflammation; ECG diffuse saddle-shaped ST elevation; pleuritic chest pain relieved by sitting forward; minimal troponin elevation; epicardial LGE (pericardium-centric)
- Dilated cardiomyopathy from other causes: Genetic, ischemic, alcohol; absence of active inflammation on CMR
7. Treatment
Acute Phase: Supportive Care
- Activity restriction: All patients with acute myocarditis should avoid competitive sports and strenuous exercise for ≥3–6 months (or until CMR normalization and symptom resolution); exercise increases viral replication and immune-mediated injury in animal models and is associated with higher mortality
- Standard heart failure therapy (GDMT): ACE inhibitors/ARBs (or ARNI), beta-blockers, MRAs, and SGLT2 inhibitors for LV dysfunction (EF <50%); loop diuretics for volume overload
- Antiarrhythmic therapy: Beta-blockers for ventricular arrhythmias; amiodarone for refractory VT; temporary pacemaker for high-degree AV block; ICD for persistent VT/VF after recovery assessment
- Anticoagulation: Therapeutic anticoagulation (LMWH, UFH, warfarin) for intracardiac thrombus; anticoagulation for AF when EF <35%; thromboprophylaxis for immobile, severely ill patients
Fulminant Myocarditis: Mechanical Circulatory Support (MCS)
- Intra-aortic balloon pump (IABP): First-line MCS option; reduces afterload and myocardial work
- Impella (microaxial flow pump): Superior hemodynamic support; CP (3.5 L/min), 5.0 (5.0 L/min), ECP for biventricular failure
- Veno-arterial extracorporeal membrane oxygenation (VA-ECMO): Most powerful MCS; biventricular and respiratory failure; bridge to recovery (ECMO removal in 1–2 weeks if hemodynamics improve) or bridge to durable LVAD/transplantation
- Temporary biventricular assist devices (BiVAD): Impella RP for concurrent RV failure
- Principle of "bridge to recovery": In fulminant myocarditis, aggressive early MCS allows the inflamed myocardium time to recover — 70–80% of fulminant myocarditis patients who survive the acute phase recover LV function
Etiology-Specific Therapy
- Giant cell myocarditis: Immunosuppression is the only therapy that modifies the uniformly fatal natural history: prednisone 1 mg/kg/day + cyclosporine 2–3 mg/kg/day (based on GIANT cell myocarditis registry experience); azathioprine as maintenance; early cardiac transplantation for refractory GCM (15–25% recurrence in transplanted heart)
- Eosinophilic/hypersensitivity myocarditis: Discontinue offending drug; high-dose systemic corticosteroids (prednisolone 1–1.5 mg/kg/day); rapid clinical and echocardiographic response typical if causative agent identified promptly
- Cardiac sarcoidosis: Corticosteroids (prednisone 0.5–1 mg/kg/day, tapering over months) reduce inflammation; steroid-sparing agents (methotrexate, azathioprine, hydroxychloroquine) for maintenance; ICD for ventricular arrhythmias and complete heart block
- Checkpoint inhibitor myocarditis: Permanently discontinue causative immune checkpoint inhibitor; high-dose corticosteroids (methylprednisolone 1 g IV/day × 3–5 days, then prednisone 1–2 mg/kg/day); refractory cases: mycophenolate mofetil, anti-thymocyte globulin, infliximab (avoid in HF), abatacept (CTLA-4 fusion protein — emerging evidence for anti-PD-1 myocarditis); intensive cardiac monitoring; hemodynamic support
- Lyme myocarditis: Doxycycline 100 mg twice daily × 14–21 days (early Lyme); IV ceftriaxone 2 g/day × 14–21 days (high-degree AV block); temporary pacemaker for complete heart block (often resolves with antibiotic therapy); permanent pacemaker rarely required
- Chagas disease myocarditis (acute phase): Benznidazole 5–7 mg/kg/day × 60 days (first-line); nifurtimox 8–10 mg/kg/day × 60–90 days (alternative); antiparasitic therapy in acute phase reduces chronic cardiomyopathy risk
- Lymphocytic viral myocarditis: No evidence for antiviral therapy; immunosuppression NOT recommended in the absence of autoimmune or granulomatous histopathology (TIMIC and ESETCID trials); supportive GDMT is standard
Role of IVIG and Immunosuppression in Lymphocytic Myocarditis
IVIG (2 g/kg over 24 hours) has been advocated based on small studies in pediatric myocarditis; current adult evidence is insufficient to recommend routine IVIG in viral lymphocytic myocarditis. Corticosteroids are not recommended for acute viral myocarditis (may increase viral replication and worsen outcomes). The ESETCID trial found no benefit of specific immunosuppression in viral PCR-positive myocarditis.
8. Complications
- Dilated cardiomyopathy: 20–30% of acute myocarditis cases progress to chronic DCM; most common long-term complication; may require heart transplantation
- Ventricular arrhythmias: Ventricular tachycardia, ventricular fibrillation; major cause of sudden cardiac death; particularly dangerous with GCM and eosinophilic myocarditis
- Complete heart block: Sinus node dysfunction or AV nodal/infra-Hisian block; may require temporary or permanent pacemaker; classic in Lyme myocarditis
- Cardiogenic shock: Fulminant myocarditis with hemodynamic collapse; requires MCS; mortality 50% without aggressive support
- Intracardiac thrombus and systemic embolism: LV thrombus formation in dilated, poorly contracting ventricle; anticoagulation required
- Sudden cardiac death: Due to ventricular fibrillation; can be the first manifestation; accounts for myocarditis-related mortality in young athletes
- Persistent myocardial inflammation: Chronic myocarditis; progressive LV dysfunction despite GDMT
- Pericardial effusion/tamponade: In myopericarditis; rarely causes hemodynamic compromise
- Heart failure with reduced EF: End-stage consequence of progressive LV remodeling
9. Prognosis
Prognosis in myocarditis varies widely by etiologic subtype and clinical presentation:
- Viral lymphocytic myocarditis: Complete recovery in 50–70% with supportive care; 10–15% progress to DCM; 5% die in the acute phase; late sudden cardiac death possible even after apparent recovery
- Fulminant myocarditis: Paradoxically excellent long-term prognosis in survivors — 93% 10-year transplant-free survival vs. 45% in acute non-fulminant myocarditis (McCarthy et al., Mayo Clinic series); the inflammatory response is more vigorous but the substrate is ultimately more recoverable
- Giant cell myocarditis: Without transplantation, median survival 5–6 months from symptom onset; with immunosuppression and transplantation, 5-year survival 70%; recurrence in transplanted heart possible
- Cardiac sarcoidosis: 5-year mortality approximately 50% in unrecognized disease; corticosteroids reduce progression but do not prevent arrhythmia-related mortality; ICD is critical
- Checkpoint inhibitor myocarditis: Overall mortality 25–50%; complete heart block, ventricular arrhythmias, and cardiogenic shock predict death
- Prognostic factors: LV function at presentation (EF <30% predicts poor outcome), RV dysfunction, hemodynamic instability, ventricular arrhythmias, GCM/eosinophilic histology, LGE extent on CMR, biopsy positive for virus
- Return-to-sports after myocarditis: CMR imaging, ambulatory monitoring, exercise testing recommended before return to competitive athletics; at least 3–6 months restriction
10. Prevention
Primary Prevention
- Prompt treatment of viral upper respiratory infections and GI illness
- Infection control measures (hand hygiene, respiratory precautions)
- Harm reduction and treatment of drug use disorders
- Pre-participation cardiac screening in competitive athletes (ECG-based screening in Europe, with some adoption in the US)
Secondary Prevention (Recurrence Prevention)
- Activity restriction for ≥3–6 months until full CMR normalization and clinical recovery
- Optimization of GDMT in patients with residual LV dysfunction
- Cardiology surveillance: ECG, troponin, BNP, and CMR at 3–6 months and 12 months post-diagnosis
- ICD consideration if persistent LV dysfunction (EF ≤35%) after 3 months of GDMT
- Avoidance of cardiotoxic substances (alcohol, cocaine, excessive NSAIDs)
- Avoidance of competitive sports until cardiac assessment clears return to sport
- Drug rechallenge avoidance if drug-induced hypersensitivity myocarditis identified
11. Recent Research and Advances
- Updated Lake Louise Criteria (2018): Revised CMR criteria incorporating native T1 and T2 mapping, ECV quantification, and LGE; increased sensitivity for subacute myocarditis and reduced need for gadolinium; now the standard diagnostic framework.
- Checkpoint inhibitor myocarditis (2018–present): Growing recognition with expanding immunotherapy use; combination checkpoint blockade (anti-PD-1 + anti-CTLA-4) carries the highest myocarditis risk (~1%); abatacept emerged as a novel treatment strategy based on mechanistic rationale and early case series (ORCHESTRA trial ongoing).
- Cardiac MRI for sport clearance: 2022 ACC/AHA scientific statement on return-to-play after myocarditis endorses CMR-guided approach; LGE persistence after clinical recovery is associated with arrhythmia risk; emerging evidence that athletes with healed myocarditis and absent LGE can safely return to sports.
- Anti-fibrotic therapies: Pirfenidone and nintedanib (approved for pulmonary fibrosis) being investigated for prevention of post-myocarditis DCM in preclinical and early clinical studies; anti-TGF-β strategies under development.
- Wearable cardiac monitors: Real-world validation of prolonged ambulatory ECG monitoring (30-day patch monitors, implantable loop recorders) to detect subclinical ventricular arrhythmias in myocarditis recovery phase; informing return-to-sport and ICD decisions.
- Precision medicine approaches: Immunohistochemical biomarker profiling (EMB) combined with viral PCR and autoantibody panels enables etiology-specific therapy; ongoing MYOVAC and BIOPSY trials aim to validate CMR vs. EMB for diagnosis and to define optimal immunosuppression regimens.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- Myocarditis epidemiology
- Viral myocarditis
- Myocarditis cardiac MRI
- Myocarditis endomyocardial biopsy
- Giant cell myocarditis
- Eosinophilic myocarditis
- Myocarditis treatment
- Myocarditis dilated cardiomyopathy
- Myocarditis troponin
- Myocarditis diagnosis guidelines
- Myocarditis mortality
- Autoimmune myocarditis
Connections
- Cardiomyopathy
- Heart Failure
- Arrhythmia
- Endocarditis
- Valvular Heart Disease
- Complete Blood Count
- Omega-3 Fatty Acids
- Magnesium
- Sarcoidosis
- Chest Pain
- Edema
- Influenza
- Hepatitis C
- Lupus
- Atrial Fibrillation
- Cardiovascular Disease
- Vaccine Injury Recovery
- Lyme Carditis and Cardiac Complications
Featured Videos

Myocarditis - causes, symptoms, diagnosis, treatment, pathology

Myocarditis - causes, pathophysiology, investigation and treatment

Causes of myocarditis | Circulatory System and Disease | NCLEX-RN | Khan Academy

Myocarditis from COVID - Heart Inflammation

mRNA COVID Shots Cause Myocarditis

What is myocarditis and pericarditis? | Circulatory System and Disease | NCLEX-RN | Khan Academy

Myocarditis: Causes, symptoms, diagnosis, treatment and prognosis | Kenhub

Myocarditis

When to Suspect Myocarditis - Dr Sam Mohiddin (HD reupload)

ICI Myocarditis

Myocarditis Causes and Inflammation Support

Myocarditis Presentation and Recent Advances: Hayes-Rosenbaum

Myocarditis: What’s new for 2025
Myocarditis

Myocarditis - A Mother's Story

Endocarditis, Pericarditis, and Myocarditis - CRASH! Medical Review Series