Acute Respiratory Distress Syndrome (ARDS)
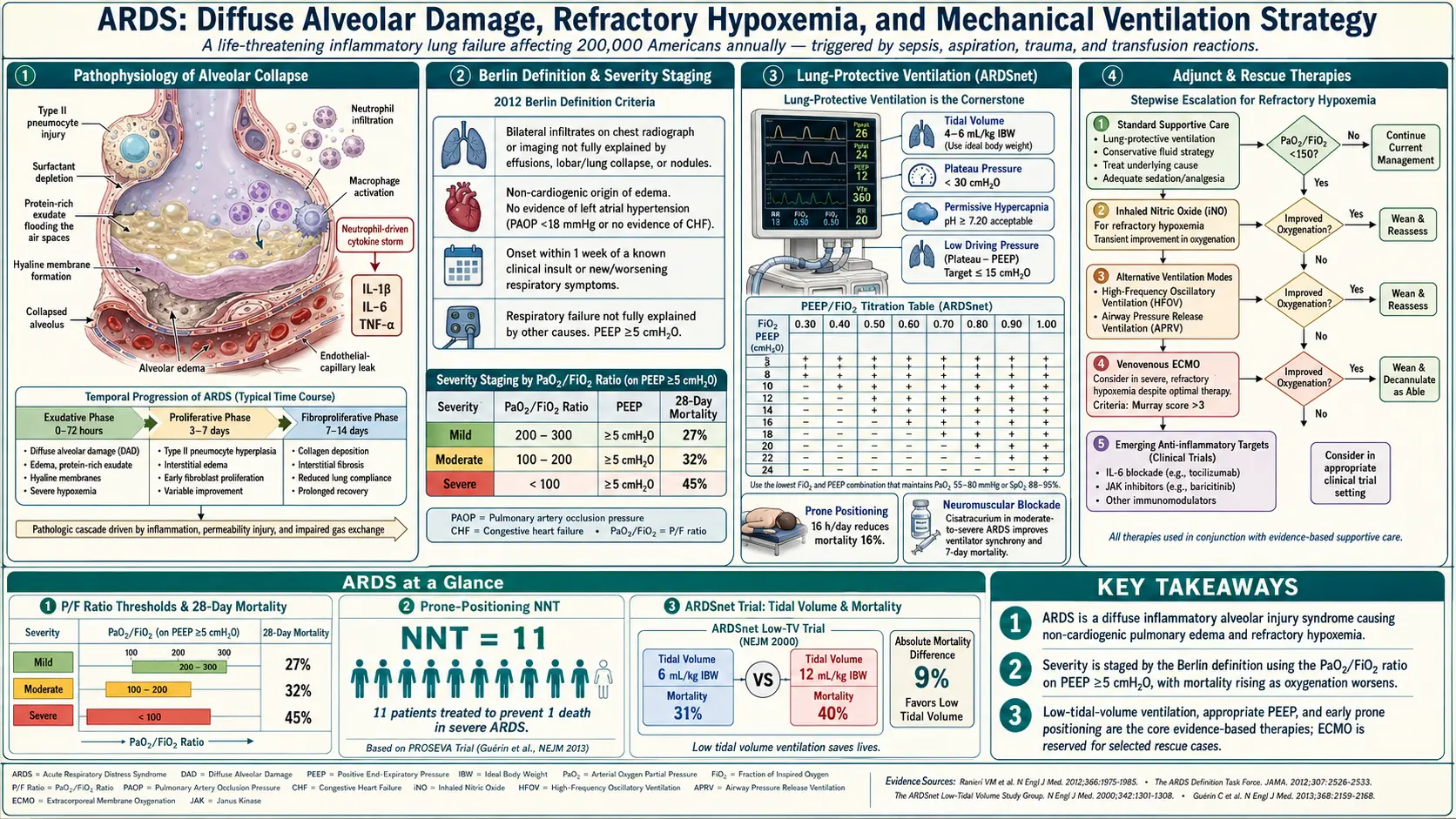
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- Research Papers
- Connections
- Featured Videos
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research
- References
1. Overview
Acute Respiratory Distress Syndrome (ARDS) is a life-threatening form of acute hypoxemic respiratory failure characterized by widespread alveolar damage, non-cardiogenic pulmonary edema, refractory hypoxemia, and reduced lung compliance. It represents the most severe manifestation of acute lung injury and carries substantial mortality, particularly in the intensive care unit (ICU) setting.
First described by Ashbaugh and colleagues in 1967, ARDS has undergone multiple definitional revisions over subsequent decades. The current standard — the Berlin Definition (2012) — classifies ARDS by severity based on the ratio of arterial oxygen partial pressure to the fraction of inspired oxygen (PaO2/FiO2), replacing the older American-European Consensus Conference (AECC) criteria. The Berlin Definition eliminated the term "acute lung injury" as a separate category and refined the timing, imaging, and oxygenation criteria to improve diagnostic reliability and prognostic stratification.
From a mechanistic standpoint, ARDS results from a dysregulated inflammatory cascade triggered by direct pulmonary insults or indirect systemic injuries. Massive cytokine release, neutrophil sequestration, alveolar epithelial and endothelial injury, and surfactant dysfunction culminate in alveolar flooding, collapse, and impaired gas exchange. The hallmark pathological lesion is diffuse alveolar damage (DAD), which progresses through distinct phases — exudative, proliferative, and fibrotic — each carrying distinct clinical and therapeutic implications.
Management has been transformed by the landmark ARDSNet ARMA trial demonstrating that low tidal volume ventilation (6 ml/kg ideal body weight) with plateau pressure limitation reduces mortality. Additional strategies including optimal PEEP titration, prone positioning, neuromuscular blockade, conservative fluid management, and in refractory cases, veno-venous extracorporeal membrane oxygenation (VV-ECMO), form the backbone of contemporary critical care.
2. Epidemiology
ARDS is a major global public health burden. Epidemiologic estimates vary substantially by case-finding methodology, diagnostic criteria applied, and clinical setting, but population-based studies from high-income countries provide consistent benchmarks.
- Incidence: Approximately 10–86 cases per 100,000 person-years in high-income settings. The LUNG SAFE study (2016), which surveyed ICUs across 50 countries, found ARDS accounted for 10.4% of all ICU admissions and 23.4% of mechanically ventilated patients.
- Age: ARDS can occur at any age, but incidence rises sharply with advancing age. Patients older than 75 years have incidence rates several-fold higher than younger adults.
- Sex: Male sex is associated with slightly higher incidence in some series; however, female sex may confer higher mortality in certain etiologies, including sepsis-related ARDS.
- Racial and socioeconomic disparities: Black patients in the United States experience higher ARDS incidence and mortality compared to White patients, driven in part by differential exposure to precipitating conditions (sepsis, pneumonia) and socioeconomic determinants of health.
- Underdiagnosis: The LUNG SAFE study revealed substantial underrecognition: only 60.2% of patients meeting Berlin criteria had ARDS documented as a clinical diagnosis by treating physicians, highlighting persistent diagnostic gaps.
- ICU resource utilization: ARDS consumes disproportionate ICU resources. The average ICU length of stay for ARDS survivors exceeds 12–14 days. Mechanical ventilation duration ranges from 5 to 14 days depending on severity.
- Seasonal variation: Incidence rises during winter respiratory-virus seasons, mirroring surges in influenza- and RSV-driven severe pneumonia that can progress to ARDS.
3. Pathophysiology
Diffuse Alveolar Damage (DAD)
The hallmark histopathological lesion of ARDS is diffuse alveolar damage (DAD), a stereotyped injury response seen regardless of the precipitating cause. DAD is characterized by injury to both the alveolar epithelium (primarily type I pneumocytes) and the pulmonary capillary endothelium, leading to loss of the alveolocapillary barrier. The result is flooding of alveolar spaces with protein-rich edema fluid, cellular debris, and inflammatory cells.
DAD progresses through three overlapping histological phases:
Exudative Phase (Days 1–7)
The exudative phase dominates the first week of illness and is characterized by:
- Alveolar flooding: Disruption of tight junctions between type I pneumocytes and endothelial cells allows plasma proteins and red blood cells to leak into alveolar spaces. Edema fluid has a protein-to-plasma ratio exceeding 0.7, distinguishing it from hydrostatic (cardiogenic) pulmonary edema.
- Hyaline membrane formation: Condensation of plasma proteins, fibrin, and cellular debris forms eosinophilic hyaline membranes lining the denuded alveolar walls — the pathognomonic light-microscopic feature of DAD.
- Neutrophil sequestration: Neutrophils accumulate in pulmonary capillaries, interstitium, and alveolar spaces. Activated neutrophils release proteases (elastase, MMP-8, MMP-9), reactive oxygen species, and neutrophil extracellular traps (NETs), amplifying epithelial and endothelial injury.
- Surfactant dysfunction: Type II pneumocyte injury reduces surfactant synthesis and secretion. Plasma proteins further inactivate surfactant, raising alveolar surface tension, promoting collapse (atelectasis), and increasing the work of breathing.
- Cytokine storm: Tumor necrosis factor-alpha (TNF-α), interleukin-1β (IL-1β), IL-6, IL-8, and other pro-inflammatory mediators are markedly elevated in bronchoalveolar lavage (BAL) fluid and plasma, driving systemic inflammation and multi-organ dysfunction.
Proliferative Phase (Days 7–21)
If the patient survives the exudative phase, the proliferative phase commences, characterized by attempts at lung repair:
- Type II pneumocyte hyperplasia: Type II cells proliferate and differentiate into type I cells to resurface denuded alveolar walls. This process is driven by growth factors including keratinocyte growth factor (KGF) and hepatocyte growth factor (HGF).
- Myofibroblast infiltration: Fibroblasts and myofibroblasts migrate into the alveolar space (intra-alveolar fibrosis) and interstitium, laying down collagen and fibronectin.
- Resolution of edema: Active sodium and water transport by type II cells and reconstitution of the alveolar-capillary barrier drive edema clearance. Impaired edema clearance correlates with worse outcomes.
- Clinical stabilization or improvement: Many patients begin to improve oxygenation and lung compliance during this phase, though weaning from mechanical ventilation may still be challenging.
Fibrotic Phase (Weeks 3–4 onward)
In a subset of patients — particularly those with prolonged severe ARDS — extensive fibroproliferation leads to the fibrotic phase:
- Architectural distortion: Replacement of normal alveolar architecture with dense fibrous tissue reduces lung compliance and impairs gas exchange. Honeycombing and cyst formation may occur.
- Pulmonary hypertension: Vascular remodeling with intimal thickening and medial hypertrophy contributes to pulmonary arterial hypertension, increasing right ventricular afterload.
- Prolonged ventilator dependence: Fibrotic ARDS is associated with markedly prolonged mechanical ventilation, difficulty weaning, and high in-hospital mortality. Survivors may develop long-term restrictive or mixed obstructive-restrictive pulmonary function abnormalities.
- Biomarkers of fibroproliferation: Elevated procollagen III propeptide (PCP-III) in BAL or plasma predicts worse outcomes and may identify patients who benefit from early corticosteroid therapy.
Mechanical Consequences: Baby Lung Concept
CT imaging of ARDS lungs reveals that consolidated and atelectatic regions are predominantly dependent (posterior in supine patients), while aerated regions are nondependent. The concept of the "baby lung," introduced by Gattinoni, refers to the relatively small volume of normally aerated lung available for ventilation — typically 200–500 mL. Ventilating this small compartment with standard tidal volumes (10–12 ml/kg) generates dangerously high regional pressures and stresses, causing ventilator-induced lung injury (VILI) through cyclic overdistension (volutrauma) and repetitive alveolar collapse and reopening (atelectrauma).
4. Etiology and Risk Factors
ARDS etiologies are broadly categorized as direct (pulmonary) or indirect (extrapulmonary) lung injuries:
Direct Pulmonary Causes
- Pneumonia: The most common direct cause globally. Bacterial (Streptococcus pneumoniae, Staphylococcus aureus, gram-negative bacilli), viral (influenza A/H1N1, RSV, adenovirus), fungal (Pneumocystis jirovecii, Aspergillus), and atypical organisms implicated. Community-acquired pneumonia causes approximately 35–40% of ARDS cases.
- Aspiration of gastric contents: Low pH aspirate (Mendelson syndrome) causes chemical pneumonitis with rapid-onset ARDS. Aspiration is a major risk in patients with decreased consciousness, dysphagia, or during endotracheal intubation.
- Pulmonary contusion: Blunt chest trauma causes direct parenchymal injury. ARDS develops in 17–22% of patients with pulmonary contusion.
- Inhalation injury: Smoke, chlorine, ammonia, nitrogen dioxide, and other toxic gases cause direct airway and alveolar damage.
- Near-drowning: Both fresh and salt water aspiration can precipitate ARDS through osmotic and direct epithelial injury mechanisms.
Indirect Extrapulmonary Causes
- Sepsis: The leading overall cause of ARDS. Non-pulmonary sepsis (abdominal, urinary, soft tissue) induces lung injury via systemic cytokine release and neutrophil activation. Sepsis accounts for approximately 40% of all ARDS cases.
- Massive blood transfusion / TRALI: Transfusion-related acute lung injury (TRALI) is a leading cause of transfusion mortality. Mediated by donor antibodies against recipient HLA or neutrophil antigens, or by biologically active lipids accumulated in stored blood products.
- Pancreatitis: Severe acute pancreatitis triggers systemic inflammatory response with ARDS in 10–25% of cases, driven by phospholipase A2 and other pancreatic enzymes entering the systemic circulation.
- Major trauma and burns: Systemic inflammatory response following extensive tissue injury, hemorrhagic shock, or massive resuscitation predisposes to ARDS.
- Drug overdose: Heroin, cocaine, salicylates, amiodarone, and certain chemotherapy agents (bleomycin, cytarabine, gemcitabine) can precipitate ARDS through direct toxicity or hypersensitivity mechanisms.
- Fat embolism syndrome: Following long bone or pelvic fractures, systemic fat embolism can cause ARDS with concomitant neurologic and cutaneous manifestations.
Host Risk Factors
- Chronic alcohol use disorder (increases ARDS risk 2- to 4-fold by impairing alveolar macrophage function and glutathione antioxidant defense)
- Cigarette smoking
- Diabetes mellitus (paradoxically, may be protective in some analyses — possibly via reduced inflammatory signaling)
- Obesity (BMI >30 kg/m²) — associated with altered chest wall mechanics and increased aspiration risk
- Hypoalbuminemia and malnutrition
- Immunosuppression (organ transplant recipients, hematologic malignancies, HIV/AIDS)
- Genetic polymorphisms: Variants in genes encoding angiotensin-converting enzyme (ACE), surfactant protein B (SP-B), and pre-B cell colony-enhancing factor (PBEF/NAMPT) have been associated with ARDS susceptibility and outcomes.
5. Clinical Presentation
ARDS typically presents within 72 hours of the precipitating insult, though onset can be as rapid as hours after aspiration or as delayed as 5–7 days in sepsis-associated cases. The clinical presentation reflects progressive respiratory failure superimposed on the underlying condition.
Symptoms
- Dyspnea: Rapidly progressive breathlessness, often severe; patients may describe inability to take a satisfying breath.
- Tachypnea: Respiratory rates of 30–40 breaths per minute are characteristic. Accessory muscle use, intercostal and suprasternal retractions indicate increased work of breathing.
- Cough: Often non-productive; productive cough may suggest infectious precipitant.
- Hypoxemia symptoms: Cyanosis (central and peripheral), agitation, confusion, and altered consciousness reflect cerebral hypoxemia.
Signs
- Tachycardia and hypotension in severe cases, particularly when ARDS complicates septic shock.
- Pulmonary auscultation: Diffuse bilateral crackles (crepitations), more prominent in dependent zones. The absence of wheeze helps distinguish ARDS from bronchospastic disease, though coexistence is possible.
- Absence of signs of left heart failure: Distinguishing ARDS from cardiogenic pulmonary edema is critical. ARDS patients typically lack elevated jugular venous pressure, S3 gallop, or peripheral pitting edema (though these may be confounded by concurrent fluid resuscitation).
- Fever: Common, particularly in infectious etiologies; may complicate clinical assessment of treatment response.
Laboratory and Bedside Findings
- Arterial blood gas: Hypoxemia (PaO2 <60 mmHg on room air or FiO2 >0.4); initial respiratory alkalosis giving way to metabolic acidosis in severe cases or concurrent shock.
- PaO2/FiO2 ratio below 300 mmHg is required for diagnosis.
- Elevated inflammatory markers: WBC with left shift, CRP, procalcitonin, ferritin, LDH.
- Chest radiograph: Bilateral airspace opacities not fully explained by effusions, lobar collapse, or nodules.
6. Diagnosis
Berlin Definition (2012)
The Berlin Definition, developed by an expert panel under the auspices of the European Society of Intensive Care Medicine and endorsed by ATS and SCCM, replaced the AECC criteria (1994) and defines ARDS by four elements:
- Timing: Onset within 1 week of a known clinical insult or new or worsening respiratory symptoms.
- Chest imaging: Bilateral opacities on chest radiograph or CT not fully explained by effusions, lobar/lung collapse, or pulmonary nodules.
- Origin of edema: Respiratory failure not fully explained by cardiac failure or fluid overload. Objective assessment (echocardiography) required if no risk factor present.
- Oxygenation: Classified by PaO2/FiO2 ratio measured on a minimum PEEP/CPAP of 5 cmH2O.
Severity Classification by PaO2/FiO2 Ratio
- Mild ARDS: PaO2/FiO2 200–300 mmHg (with PEEP or CPAP ≥5 cmH2O)
- Moderate ARDS: PaO2/FiO2 100–200 mmHg (with PEEP ≥5 cmH2O)
- Severe ARDS: PaO2/FiO2 <100 mmHg (with PEEP ≥5 cmH2O)
Note: PaO2/FiO2 ratios should ideally be measured after stabilization on the ventilator setting (typically 30 minutes to 2 hours after initiation or change in settings). SpO2/FiO2 (S/F ratio) can serve as a noninvasive surrogate when arterial blood gas is unavailable; an S/F ratio of ≤315 corresponds approximately to PaO2/FiO2 ≤300.
Differential Diagnosis
The following conditions must be excluded or distinguished from ARDS:
- Cardiogenic pulmonary edema: Elevated left atrial pressure (PCWP >18 mmHg) causes hydrostatic edema. Echocardiography, BNP/NT-proBNP, and response to diuresis assist differentiation. Bilateral effusions and cardiomegaly on CXR are more common in cardiogenic edema.
- Acute interstitial pneumonitis (AIP) / Hamman-Rich syndrome: Rapidly progressive idiopathic ARDS — diagnosis of exclusion; histopathology may be required.
- Diffuse alveolar hemorrhage (DAH): Consider in vasculitis, anticoagulated patients, or bone marrow transplant recipients; BAL shows progressively bloodier aliquots.
- Eosinophilic pneumonia (acute): May mimic ARDS; BAL eosinophilia (>25%) is diagnostic and responds dramatically to corticosteroids.
- Cryptogenic organizing pneumonia (COP): Subacute course; bilateral consolidation on CT; responds to corticosteroids.
Ancillary Investigations
- High-resolution CT chest: Reveals characteristic dependent consolidation, ground-glass opacification, and sparing of nondependent lung. CT can quantify lung recruitability, guide PEEP titration, and identify complications (pneumothorax, pleural effusion).
- Bronchoalveolar lavage (BAL): Not required for diagnosis but useful in identifying infectious etiology, DAH, or eosinophilic pneumonia. BAL protein-to-plasma ratio >0.7 confirms increased permeability edema.
- Echocardiography: Transthoracic or transesophageal echo evaluates left ventricular function, estimates pulmonary artery pressures, and quantifies right ventricular dilation — important since ARDS-associated pulmonary hypertension and cor pulmonale predict poor outcome.
- Biomarkers: Plasma Receptor for Advanced Glycation End-products (RAGE), Club cell protein 16 (CC16), angiopoietin-2, and IL-6/IL-8 are under investigation as diagnostic and prognostic biomarkers but are not standard of care.
7. Treatment
Lung-Protective Mechanical Ventilation — ARDSNet Protocol
The cornerstone of ARDS management is lung-protective ventilation (LPV), established by the ARDSNet ARMA trial (NEJM, 2000). This landmark multicenter RCT randomized 861 patients to tidal volume (Vt) of 6 ml/kg ideal body weight (IBW) versus 12 ml/kg IBW and demonstrated a 22% relative reduction in 28-day mortality (31% vs. 39.8%, p=0.007).
Ideal Body Weight Calculation (ARDSNet):
- Males: IBW (kg) = 50 + 2.3 × [height (inches) − 60]
- Females: IBW (kg) = 45.5 + 2.3 × [height (inches) − 60]
ARDSNet ARMA Ventilator Protocol Key Parameters:
- Mode: Volume-controlled assist-control (AC/VC) ventilation
- Tidal volume: Start at 6 ml/kg IBW; may reduce to 4 ml/kg IBW if plateau pressure (Pplat) target not achieved
- Plateau pressure target: ≤30 cmH2O (measured during a 0.5-second inspiratory pause)
- Driving pressure: Driving pressure = Pplat − PEEP; target <15 cmH2O (retrospective analysis by Amato et al. 2015 identified driving pressure as the strongest predictor of outcome)
- Respiratory rate: Up to 35 breaths/minute to maintain minute ventilation; titrate to pH goal
- I:E ratio: Typically 1:1 to 1:3; avoid auto-PEEP
- FiO2 and PEEP titration: Coordinated per FiO2/PEEP table (see below)
- Oxygenation target: SpO2 88–95% or PaO2 55–80 mmHg
- pH management: Target pH 7.30–7.45; permissive hypercapnia (PaCO2 up to 60–80 mmHg) acceptable if pH ≥7.20. Sodium bicarbonate infusion if pH <7.15
PEEP Titration
PEEP (positive end-expiratory pressure) prevents end-expiratory alveolar collapse (atelectrauma), recruits collapsed alveoli, and improves oxygenation. Optimal PEEP minimizes atelectrauma without causing overdistension of aerated units.
ARDSNet Lower PEEP / Higher FiO2 Table:
- FiO2 0.30 → PEEP 5 cmH2O
- FiO2 0.40 → PEEP 5–8 cmH2O
- FiO2 0.50 → PEEP 8–10 cmH2O
- FiO2 0.60 → PEEP 10 cmH2O
- FiO2 0.70 → PEEP 10–12 cmH2O
- FiO2 0.80 → PEEP 14 cmH2O
- FiO2 0.90 → PEEP 14–18 cmH2O
- FiO2 1.00 → PEEP 18–24 cmH2O
ARDSNet Higher PEEP / Lower FiO2 Table (ALVEOLI study):
- FiO2 0.30 → PEEP 5–14 cmH2O
- FiO2 0.40 → PEEP 14–18 cmH2O
- FiO2 0.50 → PEEP 16–20 cmH2O
- FiO2 0.60 → PEEP 20–22 cmH2O
- FiO2 0.80–1.0 → PEEP 22–24 cmH2O
The ALVEOLI, LOV, and EXPRESS trials found no mortality benefit of higher versus lower PEEP strategies when analyzed by intention to treat, though subgroup analyses suggest benefit in moderate-to-severe ARDS. Recruitment maneuvers (e.g., 40 cmH2O sustained inflation for 40 seconds, or PEEP titration via decremental PEEP trial after full recruitment) may improve oxygenation acutely but the ART trial demonstrated potential harm (increased 28-day mortality) with aggressive recruitment maneuver strategies in unselected patients.
Alternative PEEP titration methods include:
- Esophageal pressure-guided PEEP (EPVent trial): Uses transpulmonary pressure (Ptp = Paw − Pes) to individualize PEEP; targets Ptp 0–10 cmH2O at end-expiration. EPVent-2 trial showed no mortality benefit over empirical strategy in unselected ARDS.
- Electrical impedance tomography (EIT): Real-time regional ventilation monitoring to identify PEEP at which overdistension and collapse are balanced. Increasingly available in tertiary centers.
- Stress index: Shape of the pressure-time curve during constant-flow ventilation indicates dynamic overdistension (upward concavity) or recruitment (downward concavity).
Prone Positioning
Prone positioning redistributes lung stress more homogeneously, recruits dependent atelectatic regions, reduces ventral overdistension, and improves cardiac preload in certain physiologic conditions. The landmark PROSEVA trial (Guerin et al., NEJM 2013) demonstrated dramatic survival benefit of early prone positioning in severe ARDS:
- Inclusion: PaO2/FiO2 <150 mmHg on FiO2 ≥0.60, PEEP ≥5 cmH2O, Vt ~6 ml/kg IBW, stabilized for 12–24 hours
- Protocol: Prone for ≥16 consecutive hours per session; continued until PaO2/FiO2 >150 on FiO2 ≤0.60 and PEEP ≤10 cmH2O for ≥4 hours in supine position
- Result: 28-day mortality 16% (prone) vs. 32.8% (supine), p<0.001; 90-day mortality 23.6% vs. 41%, p<0.001
Current guidelines (SSC, ATS) strongly recommend prone positioning for moderate-to-severe ARDS (PaO2/FiO2 <150 mmHg). Contraindications include spinal instability, open abdomen, facial/pelvic fractures, elevated intracranial pressure, and hemodynamic instability refractory to vasopressors.
Neuromuscular Blockade (NMB)
The ACURASYS trial (Papazian et al., NEJM 2010) reported improved 90-day survival with 48-hour cisatracurium infusion in severe ARDS (PaO2/FiO2 <150 mmHg). The proposed mechanisms included reduced patient-ventilator asynchrony, improved lung protection through elimination of spontaneous effort-induced lung injury (P-SILI), and anti-inflammatory effects of cisatracurium specifically.
However, the larger ROSE trial (NEJM 2019) — performed in a context of lighter sedation protocols — found no mortality benefit of routine early NMB versus a strategy of light sedation without NMB (42.5% vs. 42.8% in-hospital mortality). Current guidance recommends NMB be considered in severe ARDS when:
- Persistent hypoxemia refractory to other strategies
- Severe ventilator dyssynchrony despite adequate sedation
- High transpulmonary driving pressure during assisted breathing (P-SILI)
- As a bridge to prone positioning in unstable patients
Cisatracurium is preferred for its Hofmann elimination (not dependent on renal or hepatic clearance) and its potential direct anti-inflammatory properties. Critical illness myopathy and neuropathy risk must be weighed against potential benefit; ICU-acquired weakness is more prevalent with prolonged NMB use.
Conservative Fluid Strategy
The FACTT trial (ARDSNet, NEJM 2006) compared liberal versus conservative fluid management in ARDS. Conservative strategy (targeting CVP 4–6 mmHg, PAOP 8–12 mmHg with diuresis or fluid restriction) resulted in:
- Improved oxygenation index and lung injury score
- More ventilator-free days (14.6 vs. 12.1 days, p<0.001)
- More ICU-free days (13.4 vs. 11.2 days, p<0.001)
- No difference in 60-day mortality (25.5% vs. 28.4%, p=0.30) — although trend toward benefit
- No increase in renal failure requiring dialysis
Conservative fluid management is now standard practice, using diuretics (furosemide) or continuous renal replacement therapy (CRRT) to achieve negative or neutral fluid balance, provided hemodynamic stability is maintained. Serum albumin supplementation (combined with furosemide) may improve diuresis in hypoalbuminemic patients.
Corticosteroids
The role of corticosteroids in ARDS remains nuanced and context-dependent:
- Early ARDS (<14 days): Methylprednisolone 1 mg/kg/day for 14 days then tapered may reduce duration of mechanical ventilation and improve oxygenation. The Meduri/LaSRS trial and subsequent meta-analyses support benefit; the ARDSNet LaSRS trial found no 60-day mortality benefit but significantly improved ventilator-free days and shock-free days when started within 14 days.
- Unresolving ARDS (>7 days): Persistent fibroproliferation may benefit from corticosteroids if PCP-III or other fibroproliferative markers are elevated.
- Initiation after day 14: May be associated with harm; the LaSRS trial found a trend toward higher mortality when steroids were started after 14 days of ARDS onset.
Extracorporeal Membrane Oxygenation (ECMO)
Veno-venous ECMO (VV-ECMO) provides extracorporeal gas exchange in patients with refractory severe ARDS unresponsive to optimized conventional management. VV-ECMO allows "ultraprotective ventilation" — further reducing Vt (3–4 ml/kg IBW) and Pplat (<25 cmH2O) to minimize VILI while the ECMO circuit handles CO2 removal and oxygenation.
Key evidence:
- CESAR trial (Peek et al., Lancet 2009): Transfer to ECMO center improved 6-month survival without severe disability (63% vs. 47%, p=0.03), though the comparator arm had variable lung-protective ventilation adherence.
- EOLIA trial (Combes et al., NEJM 2018): No significant difference in 60-day mortality by intention-to-treat (35% ECMO vs. 46% control, p=0.07) — the trial was stopped early for futility. However, a Bayesian analysis yielded 0.97 posterior probability of benefit with ECMO, and a crossover rescue ECMO rate of 28% in the control arm confounded results.
Current practice: VV-ECMO is offered at ECMO-capable centers for severe ARDS (PaO2/FiO2 <80 mmHg on FiO2 1.0 or pH <7.25 with PaCO2 >60 mmHg) failing prone positioning and optimized conventional ventilation. Absolute contraindications include irreversible underlying disease incompatible with recovery, unresolved major bleeding, prolonged high-pressure ventilation (>7 days at Pplat >30 cmH2O), severe immunosuppression, and significant pre-existing end-organ dysfunction.
High-Flow Nasal Cannula (HFNC) and Non-Invasive Ventilation (NIV)
In mild ARDS or early hypoxemic respiratory failure, HFNC (flows up to 60 L/min, FiO2 up to 1.0) reduces intubation rates and 90-day mortality compared to conventional oxygen in selected patients (FLORALI trial). The ROX index (SpO2/FiO2 ratio divided by respiratory rate) >4.88 at 12 hours predicts successful HFNC and avoidance of intubation.
NIV (BiPAP) may be used in mild-to-moderate ARDS but failure rates are high in moderate-to-severe disease, and delayed intubation in failing NIV is associated with worse outcomes. Awake prone positioning during HFNC or NIV may improve oxygenation and reduce intubation rates in selected patients with hypoxemic respiratory failure, though mortality benefit remains uncertain.
Treatment of Underlying Cause
Identifying and aggressively treating the precipitating cause is essential and parallels ventilatory management:
- Broad-spectrum antibiotics for bacterial pneumonia or sepsis, guided by culture results and local antibiograms; de-escalation per susceptibilities
- Antifungal therapy for invasive fungal infections in immunocompromised hosts
- Antivirals (oseltamivir or baloxavir for influenza; acyclovir for HSV/VZV pneumonitis)
- Source control for abdominal sepsis (surgical drainage, percutaneous drainage)
- Specific therapies for non-infectious causes (e.g., IVIG for TRALI, corticosteroids for drug-induced lung injury)
8. Complications
Ventilator-Induced Lung Injury (VILI)
VILI encompasses multiple mechanisms of mechanical ventilation-related lung injury occurring even in the absence of ARDS:
- Volutrauma: Overdistension of aerated alveoli from excessive tidal volume or pressure, causing alveolar rupture, capillary stress failure, and release of cytokines (biotrauma).
- Atelectrauma: Repetitive cycling of alveolar collapse and reopening with each breath generates shear forces at the transition zone between aerated and collapsed regions, amplifying DAD.
- Barotrauma: Air leaks resulting from high airway pressures — pneumothorax, pneumomediastinum, subcutaneous emphysema, pneumopericardium, and systemic air embolism.
- Biotrauma: Mechanical stress activates pulmonary NF-κB and other signaling pathways, releasing cytokines into the systemic circulation and contributing to multi-organ dysfunction.
Barotrauma and Air Leak Syndromes
Pneumothorax occurs in 5–10% of mechanically ventilated ARDS patients and is immediately life-threatening when tension physiology develops. Clinical hallmarks of tension pneumothorax include sudden hemodynamic deterioration, absent breath sounds unilaterally, tracheal deviation, and elevated peak airway pressures. Immediate needle decompression followed by chest tube thoracostomy is required. Pneumothorax should be distinguished from massive consolidation on portable CXR, requiring CT confirmation when hemodynamic stability permits.
Ventilator-Associated Pneumonia (VAP)
VAP occurs in 10–25% of intubated ARDS patients, typically after 48 hours of mechanical ventilation. Common pathogens include Pseudomonas aeruginosa, Acinetobacter baumannii, and methicillin-resistant Staphylococcus aureus (MRSA). VAP prolongs mechanical ventilation by a median of 5 days and increases ICU mortality. Prevention bundles include head-of-bed elevation (30–45°), oral decontamination with chlorhexidine, sedation minimization, and daily spontaneous awakening and breathing trials.
Pulmonary Hypertension and Acute Cor Pulmonale (ACP)
ACP complicates 20–25% of moderate-to-severe ARDS. Hypoxic vasoconstriction, hypercapnia-induced vasoconstriction, microvascular thrombosis, and mechanical compression by PEEP increase pulmonary vascular resistance. Right ventricular (RV) dilation, interventricular septal shift (D-sign on echo), and reduced RV ejection fraction characterize ACP. ACP is associated with significantly increased ICU mortality (OR ~2.0). Management includes optimization of PEEP to minimize pulmonary vascular resistance, avoidance of excessive hypercapnia, inhaled pulmonary vasodilators (nitric oxide, prostacyclin), and prone positioning (which unloads the RV).
Multi-Organ Dysfunction Syndrome (MODS)
ARDS-associated systemic inflammation and hypoxia contribute to dysfunction of kidneys (acute kidney injury requiring RRT in 30–55%), liver (cholestatic hepatopathy), brain (ICU-associated delirium and cognitive dysfunction), gut (ileus, mucosal barrier failure), and adrenal glands (relative adrenal insufficiency). MODS is the most common cause of death in ARDS, accounting for over 80% of in-hospital mortality.
Post-Intensive Care Syndrome (PICS) and Long-Term Sequelae
ARDS survivors face substantial long-term morbidity beyond discharge:
- Pulmonary function: Most survivors show near-normal spirometry and lung volumes at 6–12 months, but DLCO (diffusing capacity) often remains mildly reduced. Exertional dyspnea and reduced exercise tolerance persist in 20–40%.
- Cognitive impairment: Up to 70% of ARDS survivors demonstrate cognitive deficits (memory, attention, executive function) at 1 year.
- Psychiatric morbidity: PTSD in 20–40%, depression in 25–30%, anxiety in 40–50%.
- Neuromuscular weakness: ICU-acquired weakness (ICUAW) affects 25–50% of patients with prolonged ARDS; recovery may be incomplete over years.
- Quality of life: SF-36 physical and mental component scores remain significantly below population norms at 5 years (Herridge et al., NEJM 2011).
9. Prognosis
ARDS mortality has declined significantly from the 50–70% reported in early series (1970s–1990s) to approximately 30–45% in the modern era, attributable largely to lung-protective ventilation strategies, prone positioning, and improved sepsis management.
Mortality by Severity (Berlin Definition)
- Mild ARDS (PaO2/FiO2 200–300): Hospital mortality approximately 27%
- Moderate ARDS (PaO2/FiO2 100–200): Hospital mortality approximately 32%
- Severe ARDS (PaO2/FiO2 <100): Hospital mortality approximately 45%
These figures from the Berlin Definition validation cohort (ARDS Definition Task Force, JAMA 2012) reflect outcomes in the pre-PROSEVA era; contemporary severe ARDS mortality with optimal care including prone positioning may be lower in specialized centers.
Predictors of Mortality
- Driving pressure >15 cmH2O (strongest modifiable predictor in retrospective analyses)
- PaO2/FiO2 <100 mmHg at 24 hours
- Development of acute cor pulmonale
- Absence of improvement in oxygenation within 12–24 hours of prone positioning
- Underlying etiology (immunocompromised states, malignancy carry highest mortality)
- Age and pre-existing comorbidities (APACHE II, SOFA scores)
- Non-pulmonary organ failures (renal failure, shock requiring vasopressors)
- Fibroproliferative markers (elevated PCP-III, elevated plasma fibronectin)
Causes of Death
The majority of ARDS deaths are attributable to multi-organ dysfunction rather than refractory hypoxemia. In the ARDSNet ARMA trial, only 16% of deaths were from respiratory failure alone; the remainder resulted from sepsis, cardiovascular collapse, and multiple organ failure. This underscores the importance of source control, hemodynamic optimization, and prevention of secondary complications.
10. Prevention
Primary prevention targets reduction of ARDS incidence in at-risk populations:
- Lung-protective ventilation in the OR: Multiple RCTs and meta-analyses demonstrate that intraoperative use of low tidal volumes (6–8 ml/kg IBW) with PEEP in patients without ARDS reduces postoperative pulmonary complications and ARDS incidence. The IMPROVE trial showed perioperative LPV reduced major pulmonary complications and ICU admissions.
- Restrictive transfusion strategies: Limiting packed red blood cell transfusions to Hb <7–8 g/dL reduces TRALI risk. Use of fresh frozen plasma and platelets from male-only donors or HLA-matched donors reduces TRALI from antibody-mediated mechanisms. Leukoreduction of blood products may also reduce TRALI risk.
- Aspiration prevention: Appropriate pre-procedural fasting, rapid sequence intubation in high-risk patients, early management of dysphagia, head-of-bed elevation, and minimization of sedation to protect airway reflexes reduce aspiration-associated ARDS.
- Early goal-directed therapy and sepsis bundles: Timely antibiotics (within 1 hour of recognition), source control, and initial resuscitation targeting hemodynamic endpoints may reduce the severity of sepsis-related ARDS by attenuating the systemic inflammatory cascade.
- Alcohol cessation: Chronic alcohol use is a major modifiable risk factor. Counseling and alcohol cessation programs have potential to reduce ARDS susceptibility, particularly in perioperative and ICU settings.
- Protective ventilation during non-invasive respiratory support: Excessive spontaneous effort during HFNC or NIV may cause patient self-inflicted lung injury (P-SILI). Monitoring respiratory drive (via esophageal pressure, respiratory rate, and dyspnea scores) and timely intubation when effort is excessive may prevent progression.
11. Recent Research
The field of ARDS research has advanced substantially, with several areas of active investigation:
ARDS Phenotyping — Precision Medicine Approaches
Latent class analysis has identified two reproducible ARDS phenotypes with distinct biological signatures and treatment responses:
- Hyperinflammatory phenotype (Phenotype 2): Higher plasma IL-6, IL-8, TNF-R1, and lower protein C; more vasopressor-dependent; higher mortality (~40–50%). Paradoxically, may respond favorably to higher PEEP, liberal fluid management, and simvastatin.
- Hypoinflammatory phenotype (Phenotype 1): Relatively lower inflammatory markers; lower mortality (~20%). May be harmed by strategies that benefit Phenotype 2.
This phenotypic heterogeneity may explain the failure of numerous ARDS pharmacotherapy trials (statins, activated protein C, beta-agonists, omega-3 fatty acids) in unselected populations. Adaptive platform trial designs (REMAP-CAP) that incorporate phenotypic stratification represent the next frontier in ARDS therapeutics.
Driving Pressure as a Ventilator Target
The 2015 retrospective analysis by Amato et al. (NEJM) demonstrated that driving pressure (Pplat − PEEP) was more strongly associated with survival than either Pplat or PEEP alone, across multiple RCT datasets. Driving pressure ≤15 cmH2O appears to be the key mechanical threshold. Prospective trials specifically targeting driving pressure reduction as the primary ventilator management goal are ongoing.
Ultra-Protective Ventilation with Extracorporeal CO2 Removal (ECCO2R)
Low-flow ECCO2R devices enable tidal volume reduction below 6 ml/kg IBW (to 3–4 ml/kg) without hypercapnia by removing CO2 extracorporeally. The REST trial (McNamee et al., JAMA 2021) found no mortality benefit of very low Vt (3 ml/kg) ventilation with ECCO2R versus standard LPV and signal toward harm, including more bleeding events in the ECCO2R arm. Ultra-protective ventilation via ECCO2R requires further investigation with refined patient selection.
Awake Prone Positioning (APP)
Multiple RCTs have investigated APP in non-intubated patients with acute hypoxemic respiratory failure receiving HFNC or NIV. Pooled meta-analyses suggest that APP reduces intubation rates in selected patients, though an unambiguous mortality benefit has not been established. Duration of 8–16 hours per day of APP appears necessary for meaningful benefit, and tolerance remains a major barrier in the awake, non-sedated patient.
Mesenchymal Stromal Cell (MSC) Therapy
MSCs exert immunomodulatory, anti-inflammatory, and reparative effects in preclinical ARDS models. Phase I/II trials (START-1, START-2) demonstrated safety and potential signals of efficacy. The ongoing Phase IIb START trial (NCT03818854) is evaluating MSC efficacy in ARDS with mortality as a secondary endpoint.
Keratinocyte Growth Factor (KGF) / Palifermin
KGF stimulates type II pneumocyte proliferation and enhances alveolar fluid clearance in preclinical models. The Phase II KARE trial found that KGF did not improve outcomes and was potentially harmful in ARDS, highlighting the challenge of translating preclinical biology into effective clinical interventions.
Biomarker-Guided Treatment
The SMART-ARDS adaptive platform trial and other biomarker-enrichment approaches are testing whether phenotyping at the bedside using readily available plasma markers (IL-8, TNFR1, protein C, PAI-1) can guide treatment allocation — matching patients to therapies they are most likely to benefit from, rather than applying uniform treatments across a heterogeneous syndrome.
12. References
- ARDS Definition Task Force; Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307(23):2526–2533.
- Acute Respiratory Distress Syndrome Network; Brower RG, Matthay MA, Morris A, et al. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342(18):1301–1308.
- Guérin C, Reignier J, Richard JC, et al; PROSEVA Study Group. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368(23):2159–2168.
- Papazian L, Forel JM, Gacouin A, et al; ACURASYS Study Investigators. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363(12):1107–1116.
- National Heart, Lung, and Blood Institute PETAL Clinical Trials Network; Moss M, Huang DT, Brower RG, et al. Early neuromuscular blockade in the acute respiratory distress syndrome. N Engl J Med. 2019;380(21):1997–2008.
- Wiedemann HP, Wheeler AP, Bernard GR, et al; National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354(24):2564–2575.
- Combes A, Hajage D, Capellier G, et al; EOLIA Trial Group, REVA, and ECMONet. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378(21):1965–1975.
- Amato MBP, Meade MO, Slutsky AS, et al. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372(8):747–755.
- Bellani G, Laffey JG, Pham T, et al; LUNG SAFE Investigators; ESICM Trials Group. Epidemiology, patterns of care, and mortality for patients with acute respiratory distress syndrome in intensive care units in 50 countries. JAMA. 2016;315(8):788–800.
- Herridge MS, Tansey CM, Matté A, et al; Canadian Critical Care Trials Group. Functional disability 5 years after acute respiratory distress syndrome. N Engl J Med. 2011;364(14):1293–1304.
- Calfee CS, Delucchi K, Parsons PE, et al; NHLBI ARDS Network. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med. 2014;2(8):611–620.
- Peek GJ, Mugford M, Tiruvoipati R, et al; CESAR trial collaboration. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374(9698):1351–1363.
- Frat JP, Thille AW, Mercat A, et al; FLORALI Study Group; REVA Network. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015;372(23):2185–2196.
- McNamee JJ, Gillies MA, Barrett NA, et al; REST Investigators. Effect of lower tidal volume ventilation facilitated by extracorporeal carbon dioxide removal vs standard care ventilation on 90-day mortality in patients with acute hypoxemic respiratory failure: the REST randomized clinical trial. JAMA. 2021;326(11):1013–1023.
- Guo Z, Sun H, Li Z. Advances in research of adult ARDS clinical subphenotypes. J Clin Med. 2022;11(14):4188.
- Sweeney RM, McAuley DF. Acute respiratory distress syndrome. Lancet. 2016;388(10058):2416–2430.
Research Papers
The following PubMed topic searches surface the current peer-reviewed literature on ARDS. Each link opens a live PubMed query; results update as new papers are indexed.
- PubMed search: acute respiratory distress syndrome
- PubMed search: ARDS Berlin definition
- PubMed search: ARDS lung protective ventilation
- PubMed search: ARDS prone positioning
- PubMed search: ARDS ECMO
- PubMed search: ARDS driving pressure
- PubMed search: ARDS neuromuscular blockade
- PubMed search: ARDS corticosteroids dexamethasone
- PubMed search: ARDS fluid management conservative
- PubMed search: ARDS sepsis
- PubMed search: ARDS pneumonia
- PubMed search: ARDS mortality
Connections
- Pneumonia
- Influenza
- Heart Failure
- Pulmonary Embolism
- Interstitial Lung Disease
- Vitamin D3
- Vitamin C
- Sepsis
- Edema
- Pulmonary Hypertension
- Inflammatory Markers
- Acute Kidney Injury
- Pancreatitis
- Hypertension
- NAC and Glutathione
- Omega-3 Fatty Acids
- Pleural Effusion
- Diabetes
Featured Videos

ARDS (Acute Respiratory Distress Syndrome) Nursing - Pathophysiology, Treatment

Acute Respiratory Distress Syndrome (ARDS) | Clinical Medicine

What is ARDS? Acute Respiratory Distress Syndrome

ARDS | Made Simple for Nursing Students and NCLEX Prep

ARDS and ARF - Medical-Surgical - Respiratory System | @LevelUpRN

Acute respiratory distress syndrome ( ARDS ) Etiology, Clinical features, Diagnosis, and Treatment
Acute Respiratory Distress Syndrome (ARDS)

Acute Respirtory Distress Syndrome (ARDS) Treatment, Pathophysiology, Medicine Lecture USMLE NeetPg