Disseminated Intravascular Coagulation
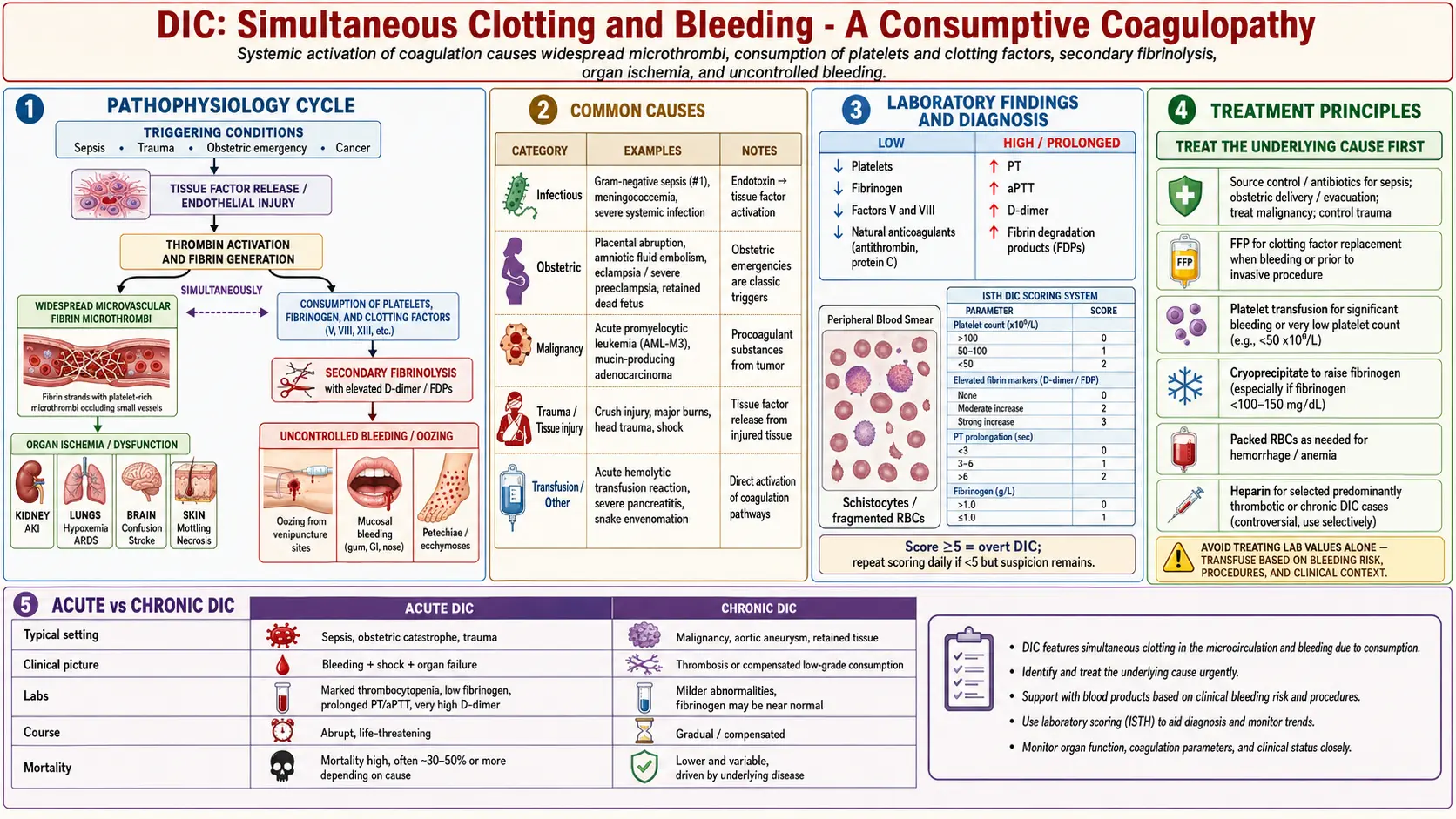
Table of Contents
- Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
1. Overview
Disseminated intravascular coagulation (DIC) is a life-threatening, acquired syndrome of systemic activation of both the coagulation and fibrinolytic systems, arising secondary to a defined underlying disorder. The ISTH defines DIC as "an acquired syndrome characterized by the intravascular activation of coagulation with loss of localization arising from different causes. It can originate from and cause damage to the microvasculature, which if sufficiently severe, can produce organ dysfunction." DIC is never a primary diagnosis — it always occurs as a complication of an identifiable precipitating condition.
The pathophysiologic hallmark is the paradox of simultaneous thrombosis and hemorrhage: uncontrolled systemic thrombin generation deposits fibrin in microvascular beds (causing ischemic organ damage), while consumption of coagulation factors and platelets, combined with secondary fibrinolysis, leads to clinical bleeding. DIC spans a spectrum from acute (decompensated) DIC with overt hemorrhage, to chronic (compensated) DIC with predominant thrombotic manifestations (as in solid tumor-associated DIC), to a subclinical form detectable only by laboratory abnormalities.
2. Epidemiology
DIC occurs in approximately 1% of hospitalized patients in the United States, with significantly higher rates in intensive care settings (up to 30% of patients with septic shock). The incidence varies markedly by underlying etiology and clinical setting:
- Sepsis is the most common trigger, accounting for ~30–50% of DIC cases in industrialized countries; DIC occurs in approximately 25–50% of patients with severe sepsis.
- Trauma and massive tissue injury contribute another 20–30% of cases.
- Obstetric DIC (abruptio placentae, amniotic fluid embolism, septic abortion, HELLP syndrome) represents a distinct and often fulminant subset.
- Malignancy-associated DIC, particularly in acute promyelocytic leukemia (APL), is characterized by a hyperfibrinolytic phenotype; chronic low-grade DIC occurs in up to 90% of patients with mucin-secreting adenocarcinomas.
Overall hospital mortality in overt DIC ranges from 40–80%, largely reflecting the severity of the underlying precipitating condition rather than DIC per se, though DIC independently worsens prognosis.
3. Pathophysiology
Coagulation Cascade Disruption
Under normal conditions, the coagulation response is precisely localized to sites of vessel injury through physical (endothelial integrity), biochemical (natural anticoagulants), and cellular mechanisms. In DIC, this spatial regulation is lost, and coagulation is systemically activated through several convergent mechanisms:
Tissue Factor-Driven Thrombin Generation
The central initiating event in most DIC subtypes is massive, uncontrolled expression of tissue factor (TF). TF is constitutively expressed on subendothelial cells and certain tissues but is normally concealed from the flowing bloodstream by intact endothelium. In sepsis, cytokines (IL-6, TNF-α, IL-1β) upregulate TF expression on monocytes and endothelial cells; in trauma, vascular disruption exposes subendothelial TF directly; in APL, malignant promyelocytes express TF and cancer procoagulant (a direct Factor X activator). TF forms a complex with Factor VIIa (extrinsic tenase: TF–VIIa), activating Factor X to Xa and Factor IX to IXa, initiating thrombin generation.
Once initiated, thrombin (Factor IIa) amplifies itself by:
- Activating Factors V, VIII, and XI (positive feedback loops of the coagulation cascade).
- Activating platelets via protease-activated receptors (PAR-1, PAR-4).
- Directly cleaving fibrinogen to fibrin monomer, which polymerizes into microvascular fibrin thrombi.
Natural Anticoagulant Depletion
Unchecked thrombin generation overwhelms and exhausts the three principal natural anticoagulant systems:
- Antithrombin (AT): Serine protease inhibitor that neutralizes thrombin and Factor Xa; markedly depleted in DIC through consumption and decreased synthesis; endotoxin and cytokines reduce hepatic AT synthesis.
- Protein C/Protein S system: Thrombin bound to thrombomodulin on endothelial cells activates protein C; activated protein C (APC) with its cofactor protein S inactivates Factors Va and VIIIa, inhibiting amplification. In DIC, endothelial thrombomodulin expression is downregulated by cytokines, impairing APC generation, and both protein C and S are consumed and reduced.
- Tissue Factor Pathway Inhibitor (TFPI): Inhibits the TF–VIIa–Xa complex; overwhelmed by sustained TF expression in DIC.
Fibrinolysis and its Dysregulation
Thrombin generation also drives endothelial release of tissue plasminogen activator (tPA), activating plasminogen to plasmin — the principal fibrinolytic enzyme. Normally, plasmin degrades fibrin clots, generating fibrin degradation products (FDP) and D-dimers (specific to cross-linked fibrin). In DIC, fibrinolysis is activated secondary to fibrin deposition but is then paradoxically suppressed by elevated plasminogen activator inhibitor-1 (PAI-1) (induced by endotoxin and cytokines), leading to inadequate clot clearance and persistence of microvascular thrombi. In some DIC subtypes (particularly APL, obstetric DIC, prostate cancer), the fibrinolytic response is exuberant (hyperfibrinolysis), exacerbating hemorrhage by prematurely dissolving clots and generating anticoagulant FDPs.
Consumptive Coagulopathy
The net result of sustained thrombin generation and fibrinolysis is consumption of fibrinogen, Factor V, Factor VIII, Factor XIII, and platelets faster than they can be replenished by synthesis, producing consumptive thrombocytopenia and hypofibrinogenemia — the laboratory signature of overt DIC. This coagulation factor depletion, combined with generation of FDPs that competitively inhibit fibrin polymerization, creates a profound hemorrhagic state superimposed on ongoing microvascular thrombosis.
4. Etiology and Risk Factors
- Sepsis and severe infection: Gram-negative bacteria (endotoxin/LPS), Gram-positive bacteria (exotoxins), fungi, viruses (dengue, HIV, influenza), malaria; most common etiology worldwide.
- Trauma: Polytrauma, traumatic brain injury (rich in TF), fat embolism, burns; "trauma-induced coagulopathy" is a related but distinct entity.
- Malignancy: Acute promyelocytic leukemia (APL; M3 AML — characteristic hyperfibrinolytic DIC); other hematologic malignancies; mucin-secreting adenocarcinomas (pancreatic, gastric, lung); prostate cancer; chronic DIC with thrombotic manifestations (Trousseau syndrome).
- Obstetric complications: Abruptio placentae (decidual TF release), amniotic fluid embolism (AF contains TF, phospholipids, and fetal material), HELLP syndrome, pre-eclampsia, retained dead fetus, septic abortion, uterine rupture.
- Liver disease: Reduced synthesis of coagulation factors, AT, and protein C; concurrent hyperfibrinolysis; hepatic ischemia in acute liver failure.
- Vascular disorders: Giant hemangiomas (Kasabach-Merritt syndrome; localized platelet and fibrinogen trapping), aortic aneurysm.
- Immunologic: ABO-incompatible blood transfusion, severe transplant rejection, anaphylaxis.
- Toxins and envenomation: Snake venom (Viperinae family; direct thrombin-like enzymes and phospholipases), certain drug toxicities.
5. Clinical Presentation
Acute (Overt) DIC
Acute DIC presents with a dramatic hemorrhagic syndrome in the context of a critically ill patient. Bleeding may be:
- Spontaneous mucocutaneous bleeding: Petechiae, purpura, ecchymoses; oozing from venipuncture sites, IV lines, surgical wounds, and mucous membranes (the most common early sign).
- Visceral hemorrhage: GI bleeding (hematemesis, melena), pulmonary hemorrhage (hemoptysis), genitourinary bleeding (hematuria), intracranial hemorrhage.
- Fulminant purpura (purpura fulminans): Extensive skin necrosis and gangrene from dermal vessel thrombosis; seen in sepsis-associated DIC (meningococcemia) and neonatal purpura fulminans (protein C deficiency).
Chronic (Compensated) DIC
Chronic DIC, often associated with solid tumors, presents primarily with thrombotic manifestations:
- Migratory superficial thrombophlebitis (Trousseau syndrome): Recurrent thrombosis at unusual venous sites.
- Venous thromboembolism: DVT, PE, cerebral sinus thrombosis.
- Non-bacterial thrombotic endocarditis (NBTE; marantic endocarditis): Fibrin-platelet vegetations on cardiac valves; embolism risk.
- Microangiopathic hemolytic anemia (MAHA): Fragmentation of red cells on fibrin strands in microvessels.
Organ Dysfunction from Microvascular Thrombosis
- Acute kidney injury: Renal cortical microvascular fibrin deposition; oliguria, rising creatinine.
- Pulmonary involvement: Respiratory failure; pulmonary microvascular thrombosis contributing to ARDS.
- Hepatic dysfunction: Elevated transaminases and bilirubin; coagulopathy worsened by impaired coagulation factor synthesis.
- CNS: Altered mental status, seizures, focal deficits from microvascular thrombosis or hemorrhage.
- Adrenal insufficiency: Bilateral adrenal hemorrhage (Waterhouse-Friderichsen syndrome) in meningococcal DIC.
6. Diagnosis
ISTH Overt DIC Scoring System
The ISTH Overt DIC Score (2001) is the most widely used diagnostic tool, applicable only when an underlying DIC-associated disorder is present. Points are assigned based on:
- Platelet count: >100 × 10⁹/L = 0; 50–100 × 10⁹/L = 1; <50 × 10⁹/L = 2.
- Elevated fibrin markers (D-dimer or FDP): No increase = 0; moderate increase = 2; strong increase = 3.
- Prolonged PT: <3 seconds prolonged = 0; 3–6 seconds = 1; >6 seconds = 2.
- Fibrinogen level: ≥1.0 g/L = 0; <1.0 g/L = 1.
A score ≥5 is compatible with overt DIC; repeat scoring daily. A score <5 suggests non-overt DIC; repeat in 1–2 days.
Laboratory Tests
- Platelet count: Falling trend is more sensitive than an isolated value; severe thrombocytopenia (<50 × 10⁹/L) occurs in advanced DIC.
- PT and aPTT: Both prolonged in overt DIC due to factor consumption; PT more sensitive early (reflects extrinsic/common pathway).
- Fibrinogen: Markedly reduced (<1.0 g/L indicates severe DIC); fibrinogen is an acute phase reactant and may be paradoxically normal-to-elevated early in DIC, masking depletion.
- D-dimer: Elevated (often markedly, >4–8 mg/L FEU); reflects plasmin-mediated degradation of cross-linked fibrin; highly sensitive but not specific (elevated in VTE, surgery, inflammation).
- Fibrin degradation products (FDP): Elevated; include non-cross-linked fibrin and fibrinogen fragments; measured by latex agglutination.
- Antithrombin activity: Reduced; correlates with DIC severity and organ failure.
- Peripheral blood smear: Schistocytes (fragmented RBCs) indicate microangiopathic hemolytic process.
- Thrombin time (TT): Prolonged (fibrinogen depletion and FDP interference with fibrin polymerization).
- Factor V and VIII assays: Markedly reduced in overt DIC.
- Thromboelastography (TEG) / Rotational thromboelastometry (ROTEM): Point-of-care viscoelastic testing that characterizes the global hemostatic profile (clot formation time, clot strength, fibrinolysis); increasingly used in trauma and obstetric DIC to guide individualized component replacement.
7. Treatment
Principle: Treat the Underlying Cause
The most critical intervention in DIC is prompt identification and treatment of the precipitating disorder. Without effective treatment of the underlying cause (e.g., antibiotics and source control for sepsis, delivery of placenta in obstetric DIC, all-trans retinoic acid and arsenic trioxide for APL), DIC will not resolve regardless of supportive measures.
Supportive Hemostatic Therapy (For Active Bleeding or High-Risk Procedures)
- Fresh frozen plasma (FFP): 15–25 mL/kg; replaces all coagulation factors (including V, VIII, fibrinogen); administer when PT/aPTT >1.5× normal with active bleeding or before invasive procedures; does not worsen DIC in clinical practice.
- Fibrinogen concentrate or cryoprecipitate: Target fibrinogen >1.5 g/L (or >2.0 g/L in obstetric DIC). Each unit of cryoprecipitate contains ~250 mg fibrinogen, ~80–100 IU FVIII, ~30% of a plasma unit's VWF, and fibronectin. Fibrinogen concentrate (RiaSTAP, Haemocomplettan) provides a precise, pathogen-inactivated dose and is increasingly preferred in trauma and obstetric DIC.
- Platelet transfusion: Maintain platelets >50 × 10⁹/L in actively bleeding patients or >20 × 10⁹/L in stable patients; use ABO-compatible products.
- Prothrombin complex concentrate (PCC; 4-factor): Contains Factors II, VII, IX, X, protein C and S; used for urgent reversal when FFP is insufficient or volume is limiting.
- Vitamin K: Correct if nutritional deficiency contributing to factor depletion.
Anticoagulation
- Heparin: Use is controversial in overt DIC with bleeding; may be beneficial in chronic/thrombotic DIC (Trousseau syndrome, NBTE, purpura fulminans, or when a clotting-dominant phenotype predominates). Low-dose unfractionated heparin (5–10 units/kg/hour continuous infusion without bolus) or LMWH may interrupt the coagulation amplification loop. Contraindicated with active CNS bleeding or fulminant hemorrhagic DIC.
- Antithrombin concentrate: Replacement of depleted AT may benefit select patients; the KyberSept trial did not show mortality benefit in all septic DIC patients but suggested benefit in those not receiving heparin.
- Recombinant human thrombomodulin (rhTM; Recomodulin): Approved in Japan for DIC management; accelerates APC generation by binding thrombin; meta-analyses and Phase 3 trial data suggest reduced mortality in DIC, particularly sepsis-associated.
- Drotrecogin alfa (recombinant APC): Withdrawn from market in 2011 after PROWESS-SHOCK trial failed to show mortality benefit in severe sepsis.
Antifibrinolytic Therapy
- Tranexamic acid (TXA): Inhibits plasminogen activators; beneficial in trauma (CRASH-2 trial: early administration within 3 hours reduces mortality from hemorrhage) and postpartum hemorrhage (WOMAN trial). Use with extreme caution in DIC — contraindicated when fibrinolysis is not the dominant feature, as blocking fibrinolysis in the presence of ongoing microvascular thrombosis may worsen organ failure. Indicated in APL-associated DIC (hyperfibrinolytic phenotype) and obstetric DIC.
APL-Specific Management
APL (AML-M3) causes a uniquely severe, hyperfibrinolytic DIC driven by TF and cancer procoagulant expressed on malignant promyelocytes, and by annexin II-mediated plasminogen activation. All-trans retinoic acid (ATRA) and arsenic trioxide (ATO) rapidly differentiate promyelocytes, dramatically reducing procoagulant activity and resolving DIC within days; they are initiated immediately upon clinical suspicion, even before cytogenetics confirm APL.
8. Complications
- Multi-organ dysfunction syndrome (MODS): Renal failure, hepatic failure, pulmonary failure (ARDS), and CNS dysfunction driven by microvascular fibrin thrombosis; the primary cause of DIC-attributable mortality beyond the underlying disease.
- Purpura fulminans: Extensive skin and soft tissue necrosis from cutaneous vessel thrombosis; may require amputation in severe cases.
- Intracranial hemorrhage: Life-threatening; associated with thrombocytopenia, factor depletion, and FDP-mediated coagulopathy.
- Adrenal cortex necrosis (Waterhouse-Friderichsen syndrome): Bilateral adrenal hemorrhage causing acute adrenal insufficiency; classic complication of meningococcal sepsis with DIC.
- Post-DIC coagulopathy: Persistent coagulation factor and platelet deficits after resolution of underlying trigger; may require prolonged replacement.
- Thrombotic complications in chronic DIC: Venous and arterial thromboembolism, NBTE with cardioembolic stroke.
9. Prognosis
Prognosis in DIC is predominantly determined by the nature, severity, and treatability of the underlying precipitating disorder. Overall hospital mortality in overt DIC is 40–80%. In sepsis-associated DIC, the presence of overt DIC doubles mortality compared to sepsis without DIC. In obstetric DIC from amniotic fluid embolism, mortality historically reached 60–80% though improved resuscitation has lowered this. APL-associated DIC has been transformed by ATRA/ATO — early hemorrhagic death (once the leading cause of APL death) now occurs in <10% of patients at specialized centers. Chronic, tumor-associated DIC (Trousseau syndrome) carries the prognosis of the underlying malignancy.
Laboratory parameters associated with increased mortality include extremely low fibrinogen (<0.5 g/L), refractory thrombocytopenia, high ISTH DIC score (>7), and elevated antithrombin depletion. Sequential ISTH scoring allows dynamic reassessment of treatment response.
10. Prevention
- Early and aggressive treatment of precipitating conditions: Rapid antimicrobial therapy and source control in sepsis; prompt delivery in obstetric emergencies; immediate ATRA initiation in suspected APL.
- Prevention of sepsis: Infection prevention bundles in ICU settings (hand hygiene, central line bundles, ventilator bundles) reduce sepsis incidence and thus DIC risk.
- Trauma resuscitation protocols: Damage control resuscitation (balanced ratio transfusion of PRBCs:FFP:platelets at 1:1:1) limits trauma-induced coagulopathy and prevents progression to DIC; early use of TXA within 3 hours of injury.
- Obstetric protocols: Active management of the third stage of labor reduces postpartum hemorrhage; early recognition of abruptio placentae and amniotic fluid embolism; WOMAN trial demonstrated TXA benefit in postpartum hemorrhage.
- Prophylactic anticoagulation: In cancer patients at high VTE and Trousseau syndrome risk; low-molecular-weight heparin preferred.
- Antithrombin supplementation: Maintaining AT levels >70% during at-risk states (e.g., post-cardiac surgery, liver failure) may reduce DIC risk, though evidence for routine use is limited.
11. Recent Research and Advances
The field of DIC management has seen renewed interest in recombinant human thrombomodulin (rhTM). The Phase 3 ART-123 trial (2019) in patients with sepsis-associated coagulopathy did not achieve statistical significance for 28-day mortality in the primary analysis but showed trends toward benefit and significant reduction in DIC score, supporting ongoing refinement of patient selection. Japanese DIC treatment guidelines continue to recommend rhTM as standard therapy for sepsis-associated DIC.
Viscoelastic hemostatic assay (VHA)-guided resuscitation (TEG, ROTEM) in trauma and obstetric hemorrhage has gained substantial evidence, with multiple RCTs showing reduced blood product usage and improved hemostasis when resuscitation is guided by real-time clot dynamics rather than conventional laboratory tests. The ITACTIC trial compared VHA-guided vs. conventional resuscitation in trauma patients.
The endotheliopathy of trauma — with shedding of the endothelial glycocalyx, release of TF, and auto-heparinization from heparanase-mediated HS shedding — is an active research target. Protein C pathway restoration and novel anticoagulant strategies targeting Factor XIa (upstream in the amplification loop) are in early clinical development for DIC prevention without increasing hemorrhagic risk. Fibrinogen-first resuscitation protocols (driven by ROTEM FIBTEM A5 thresholds) have been adopted at many trauma and obstetric centers following the RETIC and FIB24 trials.
Research Papers
The following PubMed topic searches return current peer-reviewed literature relevant to this condition. Each link opens a live PubMed query.
- DIC pathophysiology
- DIC sepsis
- ISTH DIC score
- DIC obstetric
- DIC malignancy
- DIC treatment
- DIC fresh frozen plasma
- DIC heparin
- DIC D-dimer fibrinogen
- DIC trauma
- Acute promyelocytic leukemia DIC
- DIC mortality
Connections
- Thrombocytopenia
- Sepsis
- Hemophilia
- Von Willebrand Disease
- Deep Vein Thrombosis
- Leukemia
- Cancer
- Stroke
- Anemia
- Vitamin K
- Complete Blood Count
- Acute Kidney Injury
- Liver Disease
- Hemochromatosis
- Sickle Cell Disease
- Preeclampsia
- Polycythemia Vera
- Vitamin K and Blood Clotting
Featured Videos

Osmosis from Elsevier — Disseminated intravascular coagulation (DIC): Clinical Nursing Care

NursingSOS — Disseminated Intravascular Coagulation (DIC)

Medicosis Perfectionalis — Disseminated Intravascular Coagulation (DIC) - Mnemonic - Hematology

CanadaQBank — Disseminated Intravascular Coagulation

Dr.G Bhanu Prakash Animated Medical Videos — 12. Disseminated Intravascular Coagulation (DIC) | USMLE Step 1 Pathology

sqadia.com — Disseminated Intravascular Coagulation (DIC) | DIC

Oncology Nursing Society — Episode 228: Oncologic Emergencies 101: Disseminated Intravascular Coagulation

Learning in 10 — Disseminated Intravascular Coagulation

Rabiul Haque — DIC : Disseminated Intravascular Coagulation (HD)

Level Up RN — Polycythemia & Coagulation Disorders (Hemophilia, ITP, HIT, DIC) - Medical-Surgical | @LevelUpRN
e health Africa — Disseminated intravascular coagulation - causes, symptoms, diagnosis, treatment, pathology

NURSINGcom w/Jon Haws, RN — Straight forward way to write a nursing care plan for Disseminated Intravascular Coagulation..DIC
Medilife — Disseminated Intravascular Coagulation (DIC): Causes, Symptoms, and Treatment Explainer.

dr afzal critical care medicine — Disseminated Intravascular Coagulation(Urdu Version)

Archer Review NCLEX, USMLE, TEAS7, and FNP — EP 27 - Disseminated Intravascular Coagulation (DIC)

AETCM Emergency Medicine — Case Discussion.Disseminated intravascular coagulation #DIC #Coagulopathy #aetcm