Pulmonary Embolism
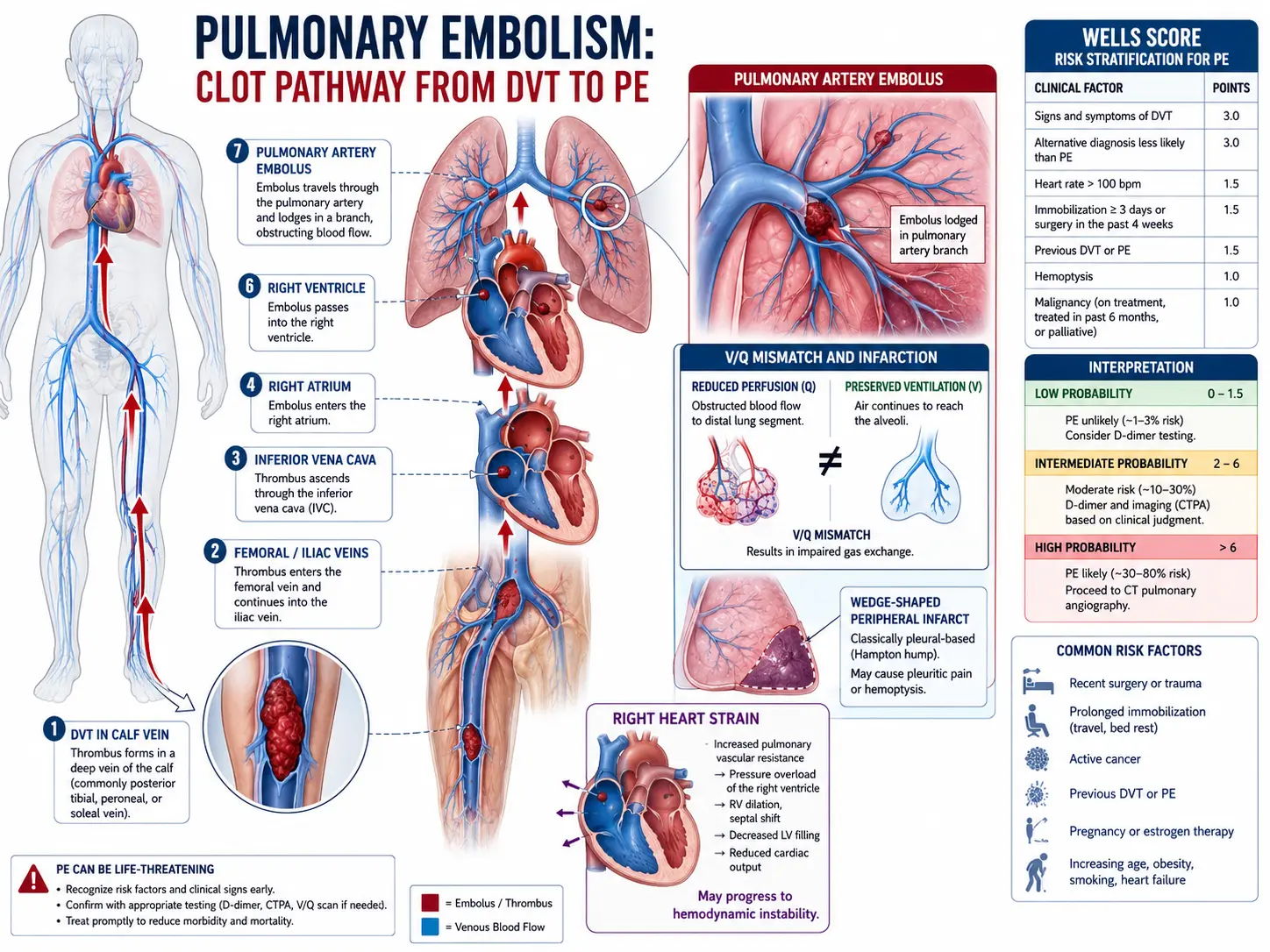
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- Research Papers
- Connections
- Featured Videos
1. Overview
Pulmonary embolism (PE) is a potentially life-threatening condition caused by the occlusion of one or more pulmonary arteries, most commonly by thrombi originating in the deep veins of the lower extremities or pelvis. PE and deep vein thrombosis (DVT) are two manifestations of the same disease spectrum, collectively termed venous thromboembolism (VTE). The clinical severity ranges from asymptomatic subsegmental PE to massive PE with hemodynamic collapse and cardiac arrest. Prompt recognition and risk stratification are essential for appropriate management and to reduce mortality.
2. Epidemiology
PE is the third most common cause of cardiovascular mortality after myocardial infarction and stroke. It accounts for approximately 100,000–180,000 deaths annually in the United States. The annual incidence is estimated at 60–70 cases per 100,000 persons, with an increasing trend attributable to aging populations, greater use of CT imaging, and rising prevalence of obesity and cancer. DVT accompanies PE in up to 70% of confirmed cases. PE is responsible for 5–10% of all in-hospital deaths. Incidence doubles with each decade of life after age 40, and men have a slightly higher age-adjusted risk than women, though oral contraceptive use and pregnancy substantially elevate risk in premenopausal women.
3. Pathophysiology
The central pathophysiological consequence of PE is ventilation-perfusion (V/Q) mismatch. When a thrombus lodges in a pulmonary artery, the affected lung segment continues to be ventilated but receives no blood flow, creating dead space ventilation. Simultaneously, hypoxic vasoconstriction redistributes blood to non-obstructed zones, which may become overperfused relative to their ventilation, causing intrapulmonary shunting.
The hemodynamic impact depends on the extent of vascular obstruction and the underlying cardiopulmonary reserve. Obstruction of >30–50% of the pulmonary vascular bed in a previously healthy patient typically triggers acute right ventricular (RV) pressure overload. The RV dilates, interventricular septal bowing toward the left ventricle (the "D-sign") reduces left ventricular preload and output, potentially precipitating cardiogenic shock. Neurohumoral mediators — serotonin, histamine, thromboxane A2 — cause additional pulmonary vasoconstriction beyond mechanical obstruction. Elevated RV wall tension and reduced coronary perfusion pressure create a positive feedback loop of RV ischemia, further impairing RV function.
Hypoxemia results from multiple mechanisms: V/Q mismatch, intracardiac right-to-left shunting through a patent foramen ovale (present in ~25% of adults), low mixed venous oxygen saturation from reduced cardiac output, and surfactant depletion causing atelectasis in areas of prolonged ischemia.
4. Etiology and Risk Factors
The pathogenesis of venous thromboembolism is classically described by Virchow's Triad:
- Venous stasis — Prolonged immobility (hospitalization, long-haul travel), paralysis, heart failure, obesity.
- Endothelial injury — Trauma, surgery (especially orthopedic), central venous catheters, infection, inflammation.
- Hypercoagulability — Inherited (Factor V Leiden mutation, Prothrombin G20210A mutation, Protein C/S deficiency, Antithrombin III deficiency) or acquired (antiphospholipid antibody syndrome, malignancy, pregnancy, oral contraceptives, heparin-induced thrombocytopenia, myeloproliferative neoplasms).
Major risk factors include:
- Prior VTE (increases risk 3-fold)
- Active malignancy (risk increased 4–7 fold; especially mucin-secreting adenocarcinomas)
- Major surgery or trauma within 3 months
- Hospitalization >3 days
- Pregnancy and the postpartum period (up to 6 weeks)
- Hormone replacement therapy or combined oral contraceptives
- Long-distance travel (>8 hours)
- Age >40 years
- Obesity (BMI >30 kg/m²)
5. Clinical Presentation
The clinical presentation of PE is notoriously variable. Symptoms range from none (incidental finding) to sudden cardiac death. The classic triad of dyspnea, chest pain, and hemoptysis occurs in fewer than 20% of patients.
Common symptoms:
- Dyspnea (most common; ~80% of symptomatic PE)
- Pleuritic chest pain (40–50%)
- Cough (20–30%)
- Hemoptysis (10–15%)
- Syncope or pre-syncope (massive PE)
- Leg pain, swelling (suggestive of concurrent DVT)
Signs:
- Tachycardia (most common sign; >100 bpm)
- Tachypnea (>20 breaths/min)
- Low-grade fever
- Hypoxemia (O2 saturation <95%)
- Hypotension or shock (massive PE)
- Loud P2, right-sided S3/S4 gallop, elevated JVP (RV strain)
The Wells Score for PE stratifies pre-test probability:
- Clinical signs/symptoms of DVT: 3 points
- PE more likely than alternative diagnosis: 3 points
- Heart rate >100 bpm: 1.5 points
- Immobilization or surgery in past 4 weeks: 1.5 points
- Previous DVT/PE: 1.5 points
- Hemoptysis: 1 point
- Malignancy: 1 point
Score ≤4: PE unlikely; >4: PE likely. The Revised Geneva Score and PERC Rule offer additional validated pre-test probability assessments.
6. Diagnosis
D-dimer: A highly sensitive (>95%) but nonspecific biomarker of fibrin degradation. Age-adjusted D-dimer threshold (age × 10 ng/mL in patients >50 years) improves specificity without sacrificing sensitivity. A negative D-dimer effectively excludes PE in low-to-moderate probability patients. Elevated in many conditions (infection, malignancy, pregnancy, post-surgery).
CT Pulmonary Angiography (CTPA): The diagnostic gold standard. Sensitivity 83–100%, specificity 89–98%. Provides direct visualization of thrombus, allows assessment of RV size (RV/LV ratio >0.9 indicates RV strain), and identifies alternative diagnoses. Preferred in patients with high pre-test probability or positive D-dimer.
Ventilation-Perfusion (V/Q) Scintigraphy: Preferred when CTPA is contraindicated (contrast allergy, severe renal impairment, pregnancy). A high-probability scan combined with high clinical probability confirms PE with >95% positive predictive value.
Echocardiography: Valuable in hemodynamically unstable patients where CTPA may not be feasible. RV dilation, septal flattening, tricuspid regurgitation, and the "60/60 sign" (PA acceleration time <60ms with tricuspid gradient <60 mmHg) suggest massive PE. Transesophageal echo may directly visualize central clot.
Cardiac biomarkers:
- Troponin I/T: Elevated in RV myocardial injury; prognostic value for short-term mortality.
- BNP/NT-proBNP: Reflects RV pressure overload; elevated levels associated with worse outcomes.
Lower extremity compression ultrasonography: When positive for proximal DVT in a patient with clinical suspicion, may preclude the need for CTPA and confirm the need for anticoagulation.
Pulmonary angiography: Invasive gold standard, now rarely used for diagnosis; reserved for catheter-directed interventions.
7. Treatment
Management is guided by risk stratification into massive (high-risk), submassive (intermediate-risk), and low-risk PE categories.
Anticoagulation
The cornerstone of PE treatment. Prevents thrombus propagation and allows endogenous fibrinolysis. Initiated empirically when clinical suspicion is high, before confirmatory imaging where appropriate.
- Unfractionated heparin (UFH): Preferred in massive PE (rapid reversibility, enables transition to thrombolytics). Weight-based IV bolus (80 units/kg) followed by continuous infusion titrated to aPTT 60–100 seconds.
- Low-molecular-weight heparin (LMWH): Enoxaparin 1 mg/kg SC every 12 hours. Preferred over UFH for non-massive PE due to predictable pharmacokinetics and lower HIT risk. Avoid in severe renal impairment (CrCl <30 mL/min).
- Direct oral anticoagulants (DOACs): Rivaroxaban (15 mg BID ×21 days, then 20 mg daily) or apixaban (10 mg BID ×7 days, then 5 mg BID) are first-line for hemodynamically stable PE. Non-inferior to VKA with reduced bleeding risk. Dabigatran and edoxaban require parenteral bridging.
- Vitamin K antagonists (warfarin): Still used when DOACs are contraindicated (antiphospholipid syndrome, severe renal failure, certain drug interactions). Target INR 2.0–3.0.
Systemic Thrombolysis
Indicated for massive PE with hemodynamic instability (SBP <90 mmHg, cardiac arrest, or obstructive shock). Alteplase 100 mg IV over 2 hours is the standard regimen. Contraindicated in patients with recent surgery (<3 weeks), active bleeding, prior hemorrhagic stroke, or intracranial neoplasm. Reduces short-term mortality and hemodynamic deterioration in massive PE at the cost of major bleeding (~9.2%) and intracranial hemorrhage (~1.5%).
For submassive PE, the PEITHO trial showed thrombolysis prevented hemodynamic decompensation but increased intracranial hemorrhage. Thrombolysis in submassive PE is reserved for patients with significant RV dysfunction, elevated biomarkers, and low bleeding risk.
Catheter-Directed Therapy
Catheter-directed thrombolysis (CDT) or ultrasound-assisted thrombolysis (EKOS system) delivers lower-dose alteplase directly into the clot, potentially reducing systemic bleeding. Surgical embolectomy is reserved for massive PE refractory to thrombolysis or when thrombolytics are absolutely contraindicated. Catheter-directed mechanical thrombectomy (e.g., FlowTriever, Penumbra) offers reperfusion without thrombolytics.
Supportive Care
- Supplemental oxygen to maintain SpO2 >94%
- Cautious fluid resuscitation (avoid RV volume overload)
- Vasopressors (norepinephrine preferred) for hemodynamic support
- Veno-arterial ECMO as bridge to definitive therapy in refractory massive PE
- IVC filter placement only when anticoagulation is absolutely contraindicated or has failed
Duration of Anticoagulation
- Provoked PE (transient risk factor): 3 months
- Unprovoked PE: minimum 3 months; extended indefinitely in low bleeding-risk patients after shared decision-making
- Cancer-associated VTE: LMWH or DOACs (edoxaban, rivaroxaban) for duration of malignancy or at least 6 months
- Recurrent unprovoked VTE: indefinite anticoagulation
8. Complications
- Chronic thromboembolic pulmonary hypertension (CTEPH): Occurs in ~4% of PE survivors; persistent obstruction leads to progressive pulmonary hypertension. Treated with surgical pulmonary endarterectomy, balloon pulmonary angioplasty, or riociguat.
- Post-PE syndrome: Functional limitation, dyspnea, and reduced quality of life persisting beyond acute phase.
- Right heart failure: Acute cor pulmonale with massive PE.
- Paradoxical embolism: Arterial embolization via patent foramen ovale; causes stroke, limb ischemia.
- Pulmonary infarction: Uncommon (~10%) due to dual pulmonary blood supply; may cause hemoptysis and pleuritis.
- Recurrent VTE: Risk is ~5–10% per year without anticoagulation after unprovoked first PE.
- Bleeding complications: Major hemorrhage from anticoagulation or thrombolytic therapy.
9. Prognosis
In-hospital mortality for all-comers with PE is approximately 7–11%, rising to 25–65% for massive PE with cardiogenic shock. The Pulmonary Embolism Severity Index (PESI) is the most validated prognostic tool:
- Class I–II (low risk): 30-day mortality <3.6%. Eligible for outpatient or early discharge management.
- Class III–V (high risk): 30-day mortality 7–24.5%. Requires hospitalization and closer monitoring.
The simplified PESI (sPESI) uses six binary variables; a score of 0 identifies low-risk patients with <1% 30-day mortality. Intermediate-risk (submassive) PE carries 3–15% short-term mortality; elevated troponin and BNP, combined with RV dysfunction on imaging or echo, identifies the intermediate-high subgroup at greatest risk of deterioration.
Long-term mortality is predominantly driven by underlying comorbidities (cancer, cardiopulmonary disease) rather than PE itself in provoked events.
10. Prevention
- Pharmacological thromboprophylaxis: UFH 5,000 units SC every 8–12 hours, LMWH, or fondaparinux in hospitalized medical and surgical patients at moderate-high VTE risk.
- Mechanical prophylaxis: Graduated compression stockings, intermittent pneumatic compression devices — particularly when pharmacological agents are contraindicated.
- Early ambulation: Post-operative mobilization within 24 hours significantly reduces VTE incidence.
- Extended outpatient prophylaxis: After major orthopedic surgery (total hip/knee replacement, hip fracture), extended LMWH or rivaroxaban/apixaban for 28–35 days is recommended.
- Risk stratification tools: Caprini Risk Assessment Model (surgical patients), Padua Prediction Score (medical patients) guide prophylaxis decisions.
- Patient education: Hydration and leg exercises during prolonged air travel; compression stockings for high-risk travelers.
- Avoid unnecessary immobility and prompt treatment of underlying hypercoagulable states.
11. Recent Research and Advances
Pulmonary Embolism Response Teams (PERTs): Multidisciplinary teams involving pulmonology, hematology, cardiology, vascular surgery, and interventional radiology have been shown to improve decision-making and outcomes in intermediate- and high-risk PE. PERT utilization is associated with increased use of advanced therapies and reduced mortality.
Ultrasound-Accelerated Thrombolysis (EKOS): The SEATTLE II and OPTALYSE PE trials demonstrated that ultrasound-assisted CDT achieves RV/LV ratio normalization with lower doses of alteplase compared to systemic thrombolysis, potentially reducing bleeding risk. Ongoing PE-TRACT trial is comparing CDT to anticoagulation alone in submassive PE.
Mechanical Thrombectomy: The FlowTriever FLARE and FLASH studies demonstrated safety and efficacy of large-bore mechanical thrombectomy for intermediate- and high-risk PE without thrombolytics. The Penumbra INDIGO system has shown similar promise.
Factor XI Inhibitors: Abelacimab, osocimab, and milvexian target Factor XIa, offering anticoagulation with potentially lower bleeding risk than current agents. Phase III trials in VTE are underway.
Artificial Intelligence in PE Diagnosis: AI algorithms applied to CTPA images can automatically detect PE, quantify thrombus burden, and assess RV strain with accuracy approaching expert radiologists, potentially accelerating diagnosis in emergency settings.
12. References
- Konstantinides SV, et al. 2019 ESC Guidelines for the diagnosis and management of acute pulmonary embolism. Eur Heart J. 2020;41(4):543–603.
- Stevens SM, et al. Antithrombotic Therapy for VTE Disease: Second Update of the CHEST Guideline. Chest. 2021;160(6):e545–e608.
- Goldhaber SZ, Bounameaux H. Pulmonary embolism and deep vein thrombosis. Lancet. 2012;379(9828):1835–1846.
- Meyer G, et al. (PEITHO Investigators). Fibrinolysis for patients with intermediate-risk pulmonary embolism. N Engl J Med. 2014;370(15):1402–1411.
- Aujesky D, et al. Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med. 2005;172(8):1041–1046.
- Tapson VF, et al. (SEATTLE II Investigators). A multicenter, randomized trial of ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute intermediate-risk pulmonary embolism. JACC Cardiovasc Interv. 2015;8(10):1382–1392.
- Piazza G, et al. (OPTALYSE PE). Optimum Duration and Dose of Ultrasound-Facilitated Thrombolysis for Intermediate-Risk Pulmonary Embolism. JACC Cardiovasc Interv. 2018;11(14):1401–1410.
- Sista AK, et al. Thirty-day outcomes, 30-day readmissions, and safety of the FlowTriever thrombectomy system for intermediate-risk pulmonary embolism. J Vasc Interv Radiol. 2021;32(10):1395–1403.
- Jaff MR, et al. Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation. 2011;123(16):1788–1830.
- Wells PS, et al. Derivation of a simple clinical model to categorize patients' probability of pulmonary embolism. Thromb Haemost. 1998;80(1):42–48.
- Torbicki A, et al. Right heart thrombi in Europe: incidence, size, treatment and outcome data from the Management Strategies and Prognosis of Pulmonary Embolism Registry. Eur Heart J. 2003;24(13):1153–1163.
- Pengo V, et al. Incidence of chronic thromboembolic pulmonary hypertension after pulmonary embolism. N Engl J Med. 2004;350(22):2257–2264.
- Carrier M, et al. Systematic review: the Trousseau syndrome revisited. Ann Intern Med. 2010;152(6):366–374.
- Righini M, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism: the ADJUST-PE study. JAMA. 2014;311(11):1117–1124.
- Tritschler T, et al. Venous Thromboembolism: Advances in Diagnosis and Treatment. JAMA. 2018;320(15):1583–1594.
Research Papers
The following PubMed topic searches surface the current peer-reviewed literature on Pulmonary Embolism. Each link opens a live PubMed query; results update as new papers are indexed.
- PubMed search: pulmonary embolism
- PubMed search: pulmonary embolism Wells score
- PubMed search: pulmonary embolism CT angiography
- PubMed search: pulmonary embolism anticoagulation
- PubMed search: pulmonary embolism thrombolysis
- PubMed search: pulmonary embolism DOAC apixaban rivaroxaban
- PubMed search: massive pulmonary embolism
- PubMed search: pulmonary embolism PESI score
- PubMed search: pulmonary embolism deep vein thrombosis
- PubMed search: pulmonary embolism cancer
- PubMed search: chronic thromboembolic pulmonary hypertension
- PubMed search: pulmonary embolism pregnancy
Connections
- Pulmonary Hypertension
- Deep Vein Thrombosis
- Cancer
- Chest Pain
- Heart Failure
- Vitamin K
- Acute Respiratory Distress Syndrome
- Shortness of Breath
- Atrial Fibrillation
- Stroke
- Obesity
- Thrombocytopenia
- Coagulation Panel
- Pneumonia
- Interstitial Lung Disease
- Vitamin D3
- Omega-3 Fatty Acids
- Edema
Featured Videos
Pulmonary Embolism PART I (Overview)
Pulmonary Embolism
Pulmonary Embolism PART II

Pulmonary Embolism, An approach to Management -- BAVLS

Pulmonary Embolism (PE) - Medical-Surgical - Respiratory System | @LevelUpRN

Pulmonary Embolism Explained Clearly - Risk factors, Pathophysiology, DVT, Treatment

Pulmonary Embolism (PE) | Med-Surg Nursing | Nursing School | Pathology | Signs & Symptoms

Pulmonary Embolism Remastered - Pathophysiology, Symptoms, Diagnosis, DVT
Pulmonary Embolism, Animation

Imaging of Pulmonary Embolism with Sally Ayesa
DVT and Pulmonary Embolism (Updated 2022) - CRASH! Medical Review Series

Pulmonary Embolism (PE) - Causes, Symptoms, Signs, Diagnosis & Treatment - Respiratory System

Pulmonary Embolism / Thromboembolism Updates Explained Clearly!

Case Study #8: Pulmonary Embolism

Pulmonary Embolism | The High Risk EM Course

How Long for a Pulmonary Embolism to Become Fatal?