Aspartame (NutraSweet, Equal): The Most Controversial Sweetener
Aspartame is the most widely used and most fiercely debated artificial sweetener in the world. Found in over 6,000 products globally — including diet sodas, sugar-free gum, tabletop sweeteners, cereals, yogurts, pharmaceuticals, and even children's vitamins — aspartame has been consumed by billions of people since its approval in 1981. In July 2023, the World Health Organization's International Agency for Research on Cancer (IARC) classified aspartame as "possibly carcinogenic to humans" (Group 2B), while the Joint FAO/WHO Expert Committee on Food Additives (JECFA) simultaneously maintained the acceptable daily intake (ADI) at 40 mg/kg body weight/day — a split decision that added new weight to decades of consumer concern.
Table of Contents
- Key Health Concerns at a Glance
- Chemical Composition
- Breakdown Products: Methanol-Formaldehyde-Formic Acid
- WHO/IARC Classification
- The Ramazzini Institute Cancer Studies
- Controversial FDA Approval History
- Headaches, Migraines, and Neurological Symptoms
- Seizure Risk
- Phenylketonuria (PKU): A Known Danger
- Presence in Over 6,000 Products
- EU Re-evaluation and Global Regulatory Status
- What the Evidence Tells Us
- Research Papers and References
- Connections
- Featured Videos
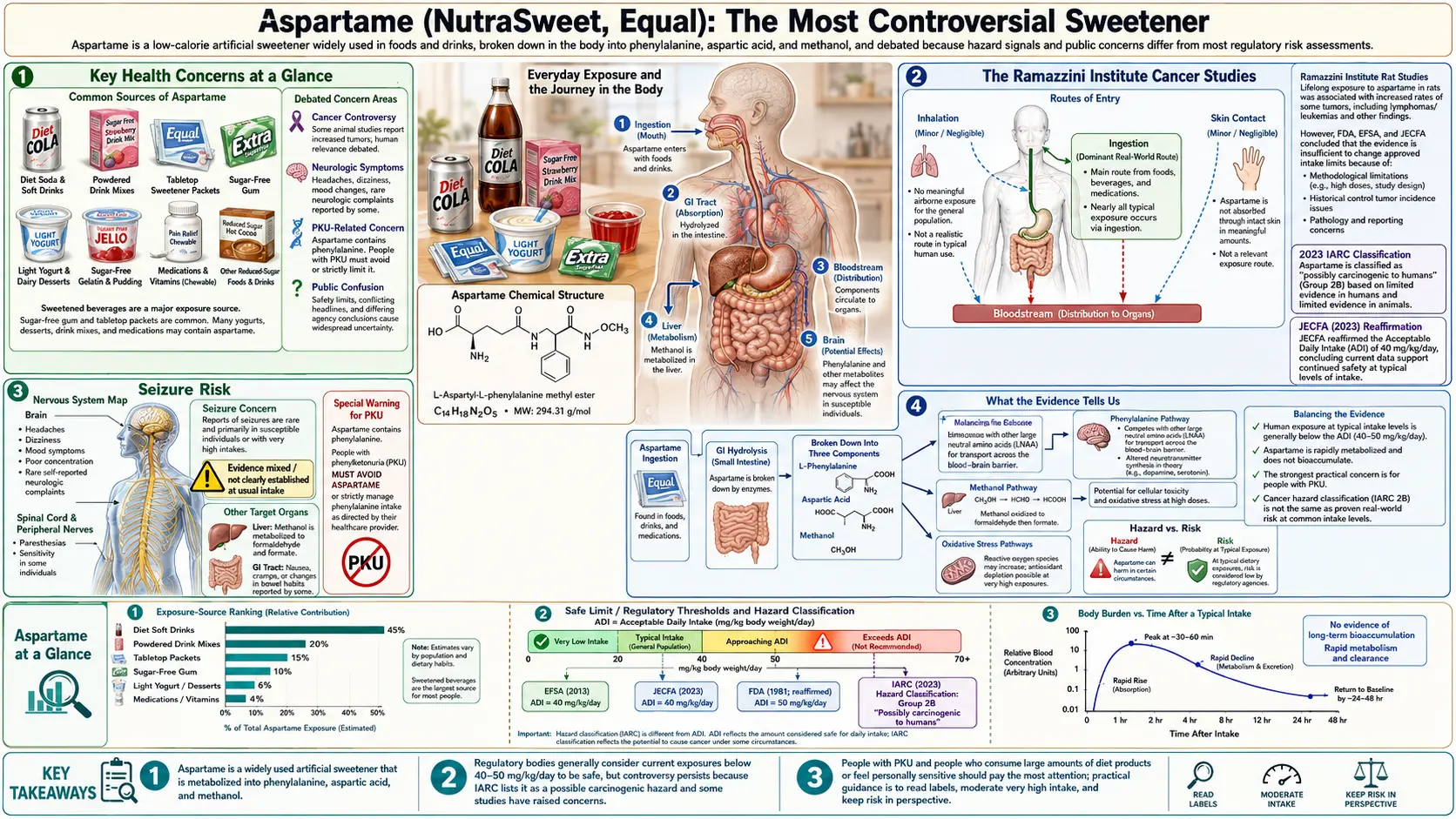
Key Health Concerns at a Glance
Before examining the mechanisms and studies in detail, here are the primary evidence-based concerns surrounding aspartame consumption, each explored further below and supported by the Research Papers section.
- IARC Group 2B carcinogen (2023) – Classified as "possibly carcinogenic to humans" based on limited evidence of hepatocellular carcinoma in humans and carcinogenic effects in animal studies.
- Methanol → formaldehyde conversion – The 10% methanol component is metabolized to formaldehyde (an IARC Group 1 carcinogen) and then to formic acid, potentially forming DNA and protein adducts with chronic exposure.
- Ramazzini Institute findings – Lifetime rodent bioassays reported statistically significant increases in lymphomas, leukemias, and other tumors at doses within the current ADI, including stronger effects when exposure began in utero.
- Migraine and headache trigger – Double-blind crossover studies (Van Den Eeden et al., 1994) confirmed aspartame provokes headache frequency and intensity in migraine-prone subjects.
- Seizure threshold concerns – Animal and case-report evidence suggests aspartame, combined with carbohydrates, may lower the seizure threshold in susceptible individuals.
- Strict danger in phenylketonuria (PKU) – Mandatory warning label required; heterozygous carriers (~1 in 50) may also have reduced metabolic capacity.
- Liver cancer signal in cohort studies – Prospective European cohort data suggested a positive association between artificially sweetened beverages and hepatocellular carcinoma.
- Controversial regulatory history – The 1981 FDA approval overturned a unanimous Public Board of Inquiry recommendation against approval, amid later-documented concerns about industry research integrity.
Chemical Composition: What Aspartame Actually Is
Aspartame is a methyl ester of a dipeptide, composed of two amino acids and a methanol component:
- Phenylalanine (50%) — An essential amino acid that is normally obtained from protein-rich foods. In isolation and at high concentrations, phenylalanine can cross the blood-brain barrier and affect neurotransmitter production, particularly serotonin and dopamine. It is especially dangerous for individuals with phenylketonuria (PKU), a genetic disorder affecting approximately 1 in 10,000-15,000 people.
- Aspartic acid (40%) — An excitatory amino acid that functions as a neurotransmitter in the brain. At elevated concentrations, aspartic acid can act as an excitotoxin, overstimulating neural receptors and potentially causing neuronal damage or death. Dr. Russell Blaylock, a neurosurgeon, has extensively documented the excitotoxic potential of aspartic acid from aspartame consumption.
- Methanol (10%) — A toxic alcohol that the body converts to formaldehyde and then to formic acid. While methanol occurs naturally in some foods (such as fruit juice), those sources also contain ethanol, which competitively inhibits methanol metabolism and provides protection. Aspartame delivers methanol without this protective co-factor.
Breakdown Products: The Methanol-Formaldehyde-Formic Acid Pathway
When aspartame is digested, the methanol component is released and metabolized through a well-characterized toxicological pathway:
- Step 1: Methanol release — Aspartame is hydrolyzed in the gastrointestinal tract, releasing free methanol. This process is accelerated by heat, which is why aspartame in hot beverages or products stored at high temperatures may release methanol before consumption.
- Step 2: Methanol to formaldehyde — The enzyme alcohol dehydrogenase converts methanol to formaldehyde, a known carcinogen (IARC Group 1). Formaldehyde is highly reactive and can form adducts with DNA, proteins, and other cellular components.
- Step 3: Formaldehyde to formic acid — Formaldehyde is further oxidized to formic acid, which inhibits mitochondrial cytochrome c oxidase, impairing cellular energy production. Formic acid accumulation is the primary cause of toxicity in methanol poisoning.
A study by Trocho et al. (1998) published in Life Sciences demonstrated that radiolabeled methanol from aspartame was converted to formaldehyde that accumulated in various tissues of laboratory animals, including the liver, kidneys, brain, and retina. The formaldehyde formed adducts with proteins and nucleic acids, suggesting potential for cumulative damage with chronic exposure.
WHO/IARC Classification: "Possibly Carcinogenic" (Group 2B)
In July 2023, after a comprehensive review of the available scientific literature, the IARC classified aspartame as "possibly carcinogenic to humans" (Group 2B). This classification was based on:
- Limited evidence in humans — Three observational cohort studies suggested positive associations between artificially sweetened beverage consumption and hepatocellular carcinoma (liver cancer), with the strongest evidence coming from a large European prospective study.
- Limited evidence in experimental animals — Multiple studies, most notably the Ramazzini Institute's lifetime bioassays, found increased incidence of malignant tumors in rodents exposed to aspartame.
- Mechanistic evidence — Evidence of oxidative stress, chronic inflammation, and genotoxicity associated with aspartame or its metabolites.
The food industry was quick to dismiss the classification, noting that coffee, aloe vera, and pickled vegetables are also in Group 2B. However, this comparison is misleading: the Group 2B classification specifically indicates that there is credible, though not yet conclusive, evidence of carcinogenicity. It is a warning, not a dismissal.
The Ramazzini Institute Cancer Studies
The European Ramazzini Foundation of Oncology and Environmental Sciences in Bologna, Italy, has conducted the most extensive long-term animal studies on aspartame ever performed. Unlike standard industry studies that typically terminate at 110 weeks (about two-thirds of a rat's natural lifespan), the Ramazzini studies allowed animals to live their full natural lifespan, more accurately modeling lifetime human exposure.
Their findings across multiple studies spanning 2005-2014 include:
- Increased lymphomas and leukemias — Statistically significant increases in malignant tumors of the lymphatic and hematopoietic systems in rats exposed to aspartame at doses equivalent to or below the current acceptable daily intake (ADI) for humans.
- Increased incidence in multiple tumor types — Elevated rates of liver and lung tumors in male mice, with a dose-response relationship.
- Fetal and prenatal exposure effects — When exposure began in utero (during pregnancy), cancer rates were even higher, suggesting increased vulnerability during development.
- Effects at "safe" doses — Importantly, increased cancer rates were observed at doses within the range of the ADI (40 mg/kg/day in the EU, 50 mg/kg/day in the US), calling into question the adequacy of current regulatory limits.
Controversial FDA Approval History
The approval of aspartame by the U.S. FDA is one of the most controversial episodes in regulatory history. Key events include:
- 1965 — James Schlatter at G.D. Searle & Company accidentally discovers aspartame while working on an anti-ulcer drug.
- 1973 — Searle submits a food additive petition to the FDA.
- 1975 — An FDA task force investigation finds "serious deficiencies" in Searle's research practices, including reporting errors, poor record-keeping, and questionable study design. The task force report describes "a pattern of conduct which compromises the scientific integrity of the studies."
- 1977 — FDA Chief Counsel Richard Merrill recommends a grand jury investigation of Searle for potentially misrepresenting the safety of aspartame. The investigation never moves forward.
- 1977-1980 — The FDA establishes a Public Board of Inquiry (PBOI) to review aspartame. In 1980, the PBOI unanimously recommends against approval, citing unresolved questions about brain tumor risk.
- 1981 — Newly appointed FDA Commissioner Arthur Hull Hayes Jr. overrules the PBOI and approves aspartame for dry foods. Hayes was appointed during the Reagan administration; Donald Rumsfeld, then CEO of Searle, was part of Reagan's transition team and is widely reported to have stated he would "call in his markers" to get aspartame approved.
- 1983 — Hayes approves aspartame for carbonated beverages, then shortly after leaves the FDA and takes a position as a consultant for Searle's public relations firm, Burson-Marsteller.
Headaches, Migraines, and Neurological Symptoms
Aspartame is one of the most commonly reported triggers for headaches and migraines. The FDA's own adverse event reporting system has received thousands of complaints about aspartame, with headaches being the most frequently reported symptom. Controlled studies support these reports:
- Migraine induction — A double-blind, randomized crossover study by Van Den Eeden et al. (1994) found that aspartame significantly increased both the frequency and intensity of headaches in migraine sufferers compared to placebo.
- Visual disturbances — Reported symptoms include blurred vision, bright flashes, tunnel vision, and eye pain, potentially related to methanol's known toxicity to the optic nerve and retina.
- Dizziness and balance problems — Multiple case reports and adverse event filings document dizziness, vertigo, and balance difficulties associated with aspartame consumption.
- Memory and cognitive effects — Animal studies have demonstrated that chronic aspartame exposure can impair memory and increase oxidative stress in the brain. A study in Nutritional Neuroscience found that rats fed aspartame showed significant impairment in spatial learning and memory.
- Depression and mood changes — A clinical study at Northeastern Ohio Universities College of Medicine found that individuals with a history of depression were significantly more sensitive to the behavioral effects of aspartame, leading the researchers to halt the study early due to the severity of adverse reactions.
Seizure Risk
The relationship between aspartame and seizures has been a concern since the earliest safety reviews. Aspartic acid, one of aspartame's components, is an excitatory neurotransmitter that can lower the seizure threshold. Animal studies by Wurtman (1985) at MIT demonstrated that aspartame in combination with carbohydrates could lower seizure thresholds in rats.
Both the U.S. Air Force magazine Flying Safety and the U.S. Navy magazine Navy Physiology published warnings about the potential dangers of aspartame for pilots, including concerns about seizures, vertigo, and visual disturbances at altitude. The Epilepsy Foundation has received numerous reports from individuals who believe aspartame triggered or worsened their seizures.
Phenylketonuria (PKU): A Known Danger
Aspartame is unequivocally dangerous for individuals with phenylketonuria (PKU), a genetic disorder in which the body cannot properly metabolize phenylalanine. In PKU patients, phenylalanine accumulates to toxic levels in the blood and brain, causing intellectual disability, seizures, and behavioral problems. This is why all aspartame-containing products carry a mandatory warning label: "PHENYLKETONURICS: CONTAINS PHENYLALANINE."
However, concerns extend beyond diagnosed PKU. Heterozygous carriers of the PKU gene (approximately 1 in 50 people) may also have reduced capacity to metabolize phenylalanine, potentially making them more susceptible to the neurological effects of aspartame consumption. These carriers typically do not know their status and receive no warning.
Presence in Over 6,000 Products
Aspartame's ubiquity in the food supply makes avoidance challenging. It is found in:
- Diet and zero-calorie beverages — Diet Coke, Coke Zero, Diet Pepsi (until 2015 reformulation, then re-added), Crystal Light, and thousands more.
- Tabletop sweeteners — NutraSweet, Equal, Sugar Twin.
- Sugar-free gum and candy — Nearly all major brands.
- Dairy products — Light yogurts, sugar-free ice cream, flavored milk.
- Baked goods and cereals — Many "light" or "reduced sugar" products.
- Pharmaceuticals — Chewable vitamins, cough drops, sugar-free cough syrups, and over 500 prescription medications.
- Condiments and sauces — Sugar-free ketchup, dressings, and syrups.
EU Re-evaluation and Global Regulatory Status
The European Food Safety Authority (EFSA) completed a full re-evaluation of aspartame in 2013, concluding that it was safe at current intake levels. However, this assessment has been criticized for relying heavily on industry-funded studies and for dismissing the Ramazzini Institute findings on methodological grounds that independent scientists have disputed.
Following the 2023 IARC classification, multiple countries and regulatory bodies have initiated fresh reviews of aspartame safety. Japan has maintained stricter regulations on aspartame than the United States and Europe, including lower acceptable daily intake limits and more stringent labeling requirements. Several countries in the Middle East and Asia have imposed their own restrictions or bans on aspartame in certain product categories.
What the Evidence Tells Us
The totality of evidence on aspartame paints a concerning picture. While the food and beverage industry continues to defend its safety, the independent scientific literature consistently points to potential harms across multiple organ systems. The WHO/IARC classification, the Ramazzini Institute findings, the epidemiological associations with cancer and metabolic disease, the documented neurological effects, and the troubled regulatory history all converge on a single conclusion: aspartame is not the harmless substance its manufacturers claim.
Consumers who wish to exercise precaution should read ingredient labels carefully, be aware that aspartame appears under multiple brand names, and consider that the long-term consequences of chronic, low-level exposure to this synthetic chemical remain incompletely understood. The fact that it is "approved" by regulatory agencies does not guarantee safety — it reflects the limitations of a regulatory system in which the burden of proof falls on those trying to demonstrate harm, rather than on those profiting from the product.
Research Papers and References
The following are landmark and frequently cited research papers underpinning the claims on this page. Links resolve to publisher DOIs, PubMed records, or the originating regulatory body.
IARC / WHO / JECFA Assessments (2023)
- Loomis D, Guha N, Hall AL, Straif K. Carcinogenicity of aspartame, methyleugenol, and isoeugenol. Lancet Oncol. 2023;24(8):848-850.
- WHO/IARC/JECFA — Aspartame hazard and risk assessment results released (14 July 2023).
- IARC Monographs on the Identification of Carcinogenic Hazards to Humans.
Cancer Epidemiology and Mechanistic Evidence
- Landrigan PJ, Straif K. Aspartame and cancer — new evidence for causation. Environmental Health. 2021;20:42.
- PubMed — Aspartame carcinogenicity search.
- PubMed — Soffritti / Ramazzini aspartame lifetime bioassay series.
Neurological Effects and Headache
- Van Den Eeden SK et al. Aspartame ingestion and headaches: a randomized crossover trial (1994) — PubMed.
- PubMed — Aspartame, excitotoxicity, and seizure threshold.
- PubMed — Aspartame, depression, and behavioral effects.
Regulatory and Authoritative Resources
- FDA — Aspartame and Other Sweeteners in Food.
- EFSA — Aspartame topic page.
- World Health Organization.
- PubMed — Aspartame safety review literature.
Connections
- Artificial Sweeteners
- Sucralose
- Acesulfame Potassium
- MSG
- Food Additives
- Food Dyes
- Banned Additives
- Detox Protocols
- Migraine
- Depression
- Phenylalanine
- Aspartic Acid
- Cancer
- Headache
- Oxidative Stress
- Diet Triggers and Elimination Protocol
Featured Videos

The Nerve Doctors - Neuropathy Pain Treatment — Aspartame Is Destroying Your Nerves! - The Nerve Doctors

CBC News: The National — How much aspartame is too much?

Renaissance Periodization — The WHO Is Wrong! Aspartame In Diet Soda Might Cause Cancer

Kiana Docherty — The Disturbing History of Aspartame

Fact Quickie — Is Aspartame Actually Bad for You?

Thomas DeLauer — Splenda & Aspartame: The End Is Near– Dr. Nick Norwitz Explains

How To Cook That — The truth about ASPARTAME!

Dr. Layne Norton — Aspartame May Cause Cancer Says the WHO | Educational Video | Biolayne

Dario Bressanini — l'ASPARTAME nella COCA ZERO è cancerogeno?

JJ Medicine — Absorption and Metabolism of Sugar Substitutes (Artificial Sweeteners) | Aspartame, Sucralose, Etc.

Nutrition Library — How Aspartame Affects Testosterone Levels In Men (New Research)

Dr. Layne Norton Podcast — Study Deep Dive: Does Aspartame Increase Insulin? | Episode 20

NutritionFacts.org — Aspartame and the Brain

NewsNation — Should everyone stop consuming aspartame? | Morning in America

Lisa J — My Scary Story about Aspartame Poisoning | Feel Good Friday

Today I Found Out — Is Aspartame Bad for You?