Carrageenan: The Seaweed-Derived Additive Linked to Gut Inflammation
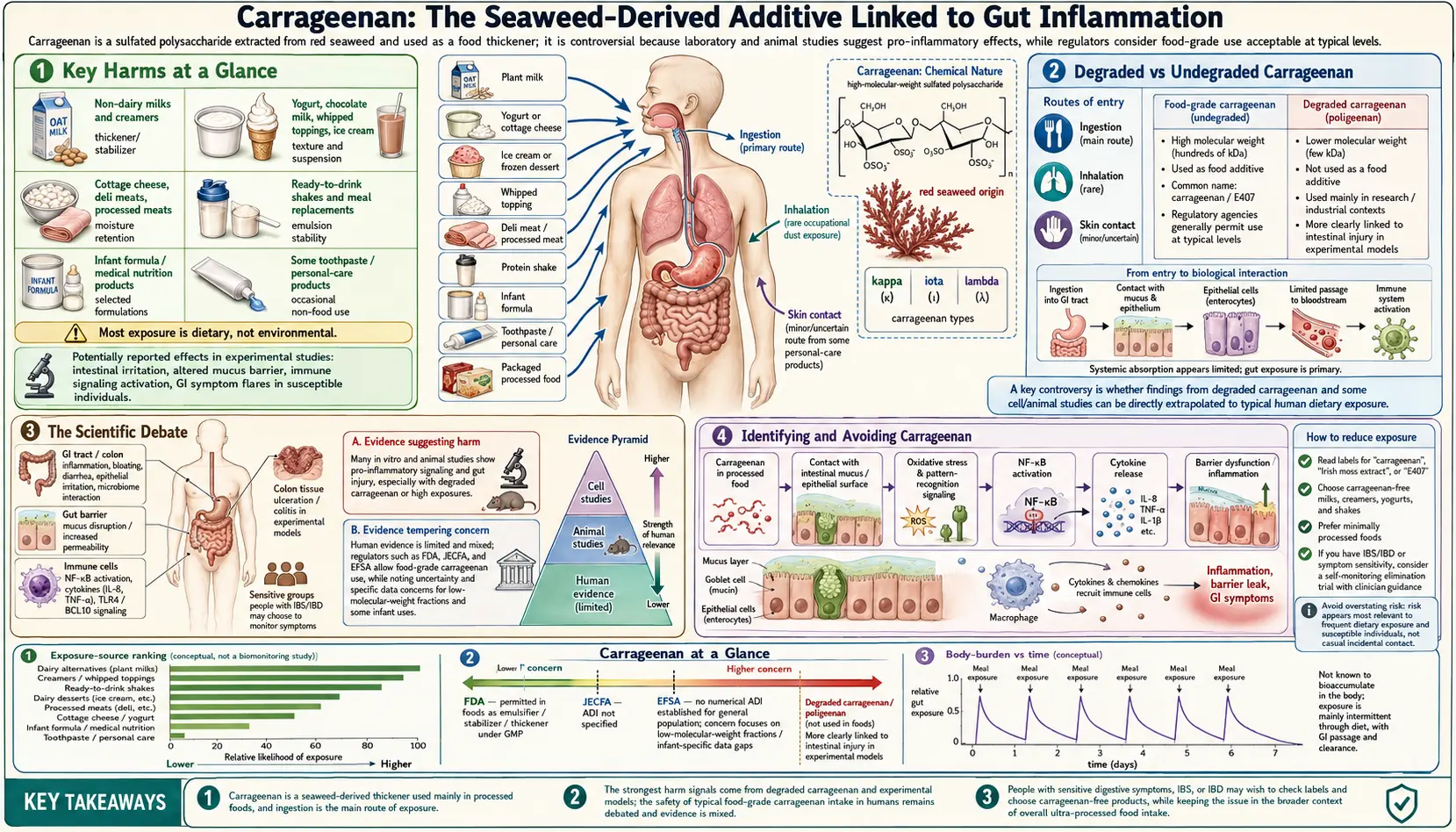
Table of Contents
- Key Harms at a Glance
- Overview
- Sources
- Degraded vs Undegraded Carrageenan
- Mechanism of Toxicity
- Health Effects
- The Scientific Debate
- Infant Formula Controversy
- Regulatory Status
- Identifying and Avoiding Carrageenan
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- Intestinal inflammation — induces colitis-like lesions in animal feeding studies.
- NF-kB / Bcl10 activation — drives chronic pro-inflammatory signaling in colonic epithelial cells.
- Tight-junction disruption — degrades intestinal barrier proteins (claudin, occludin, ZO-1).
- Glucose intolerance — impairs insulin signaling via inhibition of IGF1R.
- Tumor promotion — accelerates chemically-initiated colon cancer in animal models.
- In-vivo degradation concern — gastric acid may cleave food-grade carrageenan to pro-inflammatory fragments.
- Banned in EU infant formula (2018) — under precautionary reasoning for vulnerable infants.
Overview
Carrageenan, designated E407 in the European Union, is a family of linear sulfated polysaccharides extracted from red seaweed — primarily Chondrus crispus (Irish moss), Eucheuma cottonii, and Gigartina stellata. It has been used in food preparation in Ireland and other coastal communities for centuries, but large-scale industrial production and widespread use as a processed food additive began in the 1930s and accelerated dramatically from the 1950s onward.
There are three principal types of food-grade carrageenan, differing in their degree of sulfation and gel-forming properties:
- Kappa-carrageenan — forms strong, rigid gels in the presence of potassium ions; used in dairy products and meat processing
- Iota-carrageenan — forms soft, elastic gels with calcium ions; preferred in processed cheeses and some dairy applications
- Lambda-carrageenan — does not gel but acts as a thickener and stabilizer in cold applications; used in salad dressings and dairy beverages
Carrageenan serves multiple functional roles: it acts as a thickener, stabilizer, gelling agent, and emulsion stabilizer, preventing fat separation in dairy products, improving the texture of processed meats, and providing the characteristic smooth mouthfeel of plant-based milk alternatives. Its natural origin — extracted from seaweed using alkaline processing — has allowed it to be used in products marketed as natural or organic, a fact that has made the safety debate particularly contentious.
Sources
Carrageenan is pervasive in the modern food supply, appearing across a wide range of processed and seemingly wholesome products:
- Plant-based milks — almond milk, oat milk, coconut milk, soy milk, and rice milk frequently use carrageenan as a stabilizer to prevent separation; this is now a major source of exposure for consumers switching away from dairy
- Ice cream and frozen desserts — carrageenan provides smooth texture and prevents ice crystal formation during temperature fluctuations
- Yogurt and dairy beverages — chocolate milk, flavored yogurt drinks, and drinkable yogurts use carrageenan to maintain uniform texture
- Deli meats — turkey breast, ham, and other processed meats use carrageenan as a water-binding agent and to improve sliceability and yield
- Infant formula — carrageenan has been used in ready-to-feed infant formula as a stabilizer, though this use has become highly controversial (see Section 7)
- Toothpaste — carrageenan provides the smooth, cohesive texture of toothpaste and prevents separation during storage
- Pet food — widely used in canned and wet pet food as a gelling and thickening agent
- Cottage cheese and cream cheese — carrageenan provides creamy texture and prevents wheying off
- Nutritional shakes and meal replacements — protein shakes and liquid meal replacements often contain carrageenan to maintain suspension of nutrients
The breadth of these applications means that consumers eating a diet heavy in processed and convenience foods, or those specifically choosing plant-based dairy alternatives, may encounter carrageenan multiple times per day across different products.
Degraded vs Undegraded Carrageenan
Understanding carrageenan's safety profile requires distinguishing between two fundamentally different forms of the polymer based on molecular weight:
Undegraded carrageenan — also called food-grade carrageenan — has a high molecular weight, generally greater than 100 kDa (kilodaltons). This is the form approved for use in food under E407 designation. Its large size limits intestinal absorption, and it was historically believed to pass through the gut without systemic effects.
Poligeenan (degraded carrageenan) — produced by acid hydrolysis of carrageenan, yielding fragments with molecular weight generally less than 100 kDa, often in the 20–50 kDa range. Poligeenan is a well-established toxin and pro-inflammatory agent. It has been intentionally used for decades in laboratory research as a standard model compound to induce gut inflammation, colitis, and ulceration in experimental animals. It is also classified as a possible human carcinogen (IARC Group 2B). Poligeenan is not approved for food use.
The central toxicological controversy rests on two questions:
- Does food-grade carrageenan contain meaningful amounts of poligeenan as a contaminant from manufacturing?
- Does the acidic environment of the human stomach (pH 1–3) degrade food-grade carrageenan into lower-molecular-weight fragments with poligeenan-like properties during normal digestion?
Research by Dr. Joanne Tobacman and others has suggested that partial degradation does occur in vivo under physiological gastric conditions, particularly during prolonged gastric emptying. Industry-sponsored studies have contested this finding, arguing that the time and acid concentration required for significant degradation exceed normal physiological parameters. The debate remains scientifically unresolved, and its resolution has significant implications for the safety of food-grade carrageenan.
Mechanism of Toxicity
The proposed mechanisms by which carrageenan — particularly its lower-molecular-weight forms or degradation products — promotes intestinal inflammation involve several well-characterized molecular pathways:
- TLR4/NF-κB inflammatory pathway activation — carrageenan activates Toll-like receptor 4 (TLR4) on intestinal epithelial cells and macrophages, triggering the nuclear factor kappa-B (NF-κB) transcription factor cascade. NF-κB drives expression of pro-inflammatory cytokines including TNF-alpha, IL-1beta, and IL-6, establishing a chronic inflammatory state in the gut mucosa.
- Bcl10-mediated inflammation — research by Tobacman's group identified that carrageenan activates the Bcl10 signaling pathway, which intersects with innate immune signaling and amplifies inflammatory responses in intestinal epithelial cells, an effect distinct from its TLR4 action.
- Disruption of the intestinal epithelial barrier — carrageenan has been shown to reduce expression of tight junction proteins (claudin, occludin, ZO-1) that maintain the physical barrier between gut contents and the systemic circulation. Degradation of tight junctions increases intestinal permeability ("leaky gut"), allowing bacterial products and food antigens to access the lamina propria and trigger further immune activation.
- Reduction of beneficial gut bacteria — carrageenan consumption has been associated with reductions in Lactobacillus and Bifidobacterium populations, disrupting the protective microbiome that normally limits inflammatory activation in the gut.
- Increased intestinal permeability — the combination of barrier disruption and altered microbiome composition promotes bacterial translocation, where gut bacteria cross the intestinal wall and stimulate systemic immune responses.
These mechanisms collectively describe a plausible biological pathway from routine carrageenan consumption to chronic low-grade intestinal inflammation, which in turn may contribute to symptoms resembling irritable bowel syndrome and, in susceptible individuals, inflammatory bowel disease.
Health Effects
The health effects attributed to carrageenan span a spectrum from mild gastrointestinal discomfort to more serious systemic conditions in animal models:
- Intestinal inflammation and colitis-like effects — the most consistently demonstrated effect in animal studies; carrageenan-fed animals develop mucosal ulceration, crypt distortion, and inflammatory cell infiltration resembling human inflammatory bowel disease
- Bloating and IBS symptoms — many individuals with irritable bowel syndrome or inflammatory bowel disease report symptom improvement after eliminating carrageenan from their diet; clinical evidence for this is primarily anecdotal and observational rather than from controlled trials
- Glucose intolerance and metabolic effects — animal studies have shown that carrageenan consumption impairs insulin signaling and promotes glucose intolerance, potentially through inflammation-mediated disruption of insulin receptor pathways in the liver and muscle
- Tumor promotion — in animals with chemically initiated colon cancer, carrageenan feeding has been shown to accelerate tumor development; this tumor-promoting rather than initiating role is consistent with its pro-inflammatory mechanism
- Immune suppression — paradoxically, some studies have found carrageenan suppresses certain aspects of adaptive immunity, potentially reflecting immune exhaustion in response to chronic inflammatory stimulation
It is important to note that human clinical evidence directly linking food-grade carrageenan to these health outcomes remains limited. The strongest evidence is from animal studies, in vitro cell culture work, and patient-reported symptom improvement on elimination diets. Well-powered randomized controlled human trials are largely absent from the literature.
The Scientific Debate
Few food additives have generated as prolonged and contentious a scientific debate as carrageenan. The controversy spans regulatory petitions, academic disputes, industry responses, and conflicting expert committee conclusions:
The Cornucopia Institute, a food advocacy organization, submitted a formal petition to the FDA in 2008 requesting removal of carrageenan from organic food products, citing extensive animal and in vitro evidence of intestinal harm. The petition was denied, but it generated renewed scientific scrutiny.
Dr. Joanne Tobacman of the University of Illinois at Chicago has conducted the most sustained independent research program on carrageenan toxicity, publishing numerous peer-reviewed studies demonstrating NF-κB activation, Bcl10 pathway involvement, tight junction disruption, and glucose intolerance from carrageenan exposure in cell and animal models. Her work has been the primary scientific basis for the calls to restrict carrageenan in food.
Industry-sponsored counter-studies have consistently concluded that food-grade carrageenan is safe at current exposure levels, arguing that the doses used in animal studies are supraphysiological, that human gastric conditions do not degrade carrageenan to poligeenan, and that epidemiological evidence does not support a link between carrageenan consumption and inflammatory bowel disease in human populations.
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) and EFSA have re-evaluated carrageenan multiple times and maintained its approved status, while acknowledging that questions remain about potential degradation in vivo and effects in vulnerable populations.
The National Organic Standards Board (NOSB) voted in 2016 to recommend removing carrageenan from the National Organic Program — an unprecedented step by the oversight body that certifies organic food. However, the USDA overrode the NOSB recommendation and retained carrageenan's organic certification, a decision that drew criticism from organic food advocates and illustrated the limits of scientific advisory bodies in the face of commercial interests.
Infant Formula Controversy
Perhaps the most consequential application of carrageenan from a public health standpoint is its use in ready-to-feed infant formula. Carrageenan has been used as a stabilizer in liquid infant formula to prevent fat separation and maintain a homogeneous suspension of nutrients.
The concern is straightforward: infants have an immature gastrointestinal system with underdeveloped tight junction protein expression, reduced mucus layer thickness, incomplete microbiome establishment, and heightened immunological sensitivity. All of these factors could render the infant gut substantially more susceptible to carrageenan-induced inflammatory effects than the adult gut.
The European Union took decisive action in 2018, banning carrageenan from all infant formula products (both liquid and powder form) under EU Commission Directive 2016/127. The EU's scientific reasoning explicitly cited precautionary concerns given the vulnerable population and the unresolved questions about in vivo degradation and inflammatory potential.
The United States has not followed suit. As of 2026, carrageenan remains permitted in infant formula sold in the US market. The FDA's position is that existing evidence does not demonstrate harm at the levels present in formula. Consumer advocacy groups continue to press for a US ban consistent with the EU standard, arguing that the precautionary principle should apply when vulnerable infants are the exposed population and when biological mechanisms of harm have been plausibly demonstrated.
Regulatory Status
Carrageenan occupies a complex regulatory landscape that varies significantly by jurisdiction and application:
- United States (FDA) — carrageenan holds GRAS status and is approved for use in a wide range of food applications. There is no established ADI. The FDA has declined petitions to restrict its use in organic foods or infant formula.
- European Union — approved as E407 for general food use with an ADI of "not specified" (meaning JECFA has not identified a need for a quantitative limit). However, banned from infant formula since 2018 under precautionary reasoning.
- Codex Alimentarius (JECFA) — ADI of "not specified", meaning the committee found no numerical limit necessary based on available evidence, though this designation also reflects uncertainty rather than a clean safety bill of health.
- Organic certification — carrageenan is permitted in USDA-certified organic products despite the NOSB's 2016 recommendation for removal. This inconsistency between the advisory body's recommendation and USDA policy continues to generate controversy in the organic food community.
- EU infant formula — prohibited. This represents the most significant regulatory restriction on carrageenan globally and reflects growing official concern about its use in products consumed by vulnerable populations.
Identifying and Avoiding Carrageenan
For individuals who wish to reduce or eliminate carrageenan from their diet — particularly those with inflammatory bowel conditions, IBS, or infants — the following guidance applies:
- Read labels carefully — carrageenan will be listed on ingredient labels as "carrageenan," "E407," or occasionally "Irish moss extract." There are no hidden names or synonyms that obscure its presence (unlike BHT from packaging migration).
- Plant-based milks — seek brands specifically labeled "carrageenan-free." Many brands have reformulated in response to consumer demand. Homemade nut milks contain no carrageenan.
- Infant formula — check ingredient labels; liquid/ready-to-feed formulas are more likely to contain carrageenan than powder formulas.
- Deli meats and processed meats — choose minimally processed whole cuts over pre-sliced deli products.
Effective alternatives to carrageenan for food manufacturers include:
- Locust bean gum (carob bean gum) — a natural galactomannan that provides thickening and gelling without the inflammatory concerns associated with carrageenan; widely used in dairy and meat applications
- Gellan gum — a microbially produced polysaccharide with excellent gelling properties used in dairy beverages and plant-based milks
- Guar gum — a highly effective cold-water-soluble thickener derived from guar beans, used in ice cream, sauces, and dairy products
- Pectin — fruit-derived gelling agent appropriate for dairy and non-dairy applications requiring a cleaner label
Research Papers and References
Key research papers and authoritative sources. Links resolve to DOI, PubMed, or the issuing agency.
- Tobacman JK. Review of harmful gastrointestinal effects of carrageenan in animal experiments. Environ Health Perspect. 2001;109(10):983-994.
- Bhattacharyya S, Borthakur A, Pant N, et al. Carrageenan inhibits insulin signaling through GRB10-mediated inhibition of IGF1R and insulin receptor. J Biol Chem. 2012;287(38):32259-32272.
- Bhattacharyya S, Dudeja PK, Tobacman JK. Carrageenan-induced NFkappaB activation depends on distinct pathways mediated by reactive oxygen species and Hsp27 or by Bcl10. Biochim Biophys Acta. 2008;1780(7-8):973-982.
- Borthakur A, Bhattacharyya S, Anbazhagan AN, et al. Prolongation of carrageenan-induced inflammation in human colonic epithelial cells by activation of an NFκB-BCL10 loop. Biochim Biophys Acta. 2012;1822(8):1300-1307.
- Martino JV, Van Limbergen J, Cahill LE. The role of carrageenan and carboxymethylcellulose in the development of intestinal inflammation. Front Pediatr. 2017;5:96.
- EFSA Panel on Food Additives and Nutrient Sources. Re-evaluation of carrageenan (E 407) and processed Eucheuma seaweed (E 407a) as food additives. EFSA J. 2018;16(4):5238.
- McKim JM Jr. Food additive carrageenan: Part I: A critical review of carrageenan in vitro studies, potential pitfalls, and implications for human health and safety. Crit Rev Toxicol. 2014;44(3):211-243.
- Cohen SM, Ito N. A critical review of the toxicological effects of carrageenan and processed eucheuma seaweed on the gastrointestinal tract. Crit Rev Toxicol. 2002;32(5):413-444.
- Weiner ML. Food additive carrageenan: Part II: A critical review of carrageenan in vivo safety studies. Crit Rev Toxicol. 2014;44(3):244-269.
- Bhattacharyya S, Liu H, Zhang Z, et al. Carrageenan-induced colonic inflammation is reduced in Bcl10 null mice and increased in IL-10-deficient mice. Mediators Inflamm. 2010;2010:785986.
- Tobacman JK, Wallace RB, Zimmerman MB. Consumption of carrageenan and other water-soluble polymers used as food additives and incidence of mammary carcinoma. Med Hypotheses. 2001;56(5):589-598.
- European Commission. Commission Delegated Regulation (EU) 2016/127 supplementing Regulation (EU) No 609/2013 as regards the specific compositional and information requirements for infant formula and follow-on formula. Off J Eur Union. 2016.
- Cornucopia Institute. Carrageenan: How a 'Natural' Food Additive is Making Us Sick. Cornucopia Institute Report. 2013.
- Bhattacharyya S, Tobacman JK. Molecular signature of kappa-, iota-, and lambda-carrageenans and effect of these carrageenans on the expression of MUC5AC and inflammatory cytokines in human bronchial epithelial cells. J Nutr Biochem. 2015;26(10):1083-1092.
- Necas J, Bartosikova L. Carrageenan: a review. Vet Med (Praha). 2013;58(4):187-205.
- Liu H, Bhattacharyya S, Bhattacharyya R, et al. Carrageenan stimulation of cytokine production in macrophages and intestinal epithelial cells. J Carbohydr Chem. 2012;31(6):476-488.
- Bhatnagar S, Bhattacharyya S, Tobacman JK. Poligeenan and inflammatory bowel disease. Indian J Pediatr. 2014;81(8):777-782.
- PubMed — carrageenan intestinal inflammation search
Connections
- Food Additives
- Polysorbate 80
- Titanium Dioxide
- BHT
- Banned Additives
- Irritable Bowel Syndrome
- Crohns Disease
- Ulcerative Colitis
- Inflammatory Bowel Disease
- Gut Healing
- Gut Brain Axis
- Leaky Gut
- Bloating
- Milk
- Yogurt
- Cancer
- Ultra Processed Foods
- Diet for IBD
Featured Videos
Health X Universe — Carrageenan Causes Stomach Inflammation 2022

Eat Well Crohn's Colitis — Food Additives to Watch Out For with IBD
Kick It Naturally — Dealing with Chronic Canker Sores
Violin MD — SHOCKING cause of Deadly Meat Allergy: Medical Mystery Solved!
VisitJoy — 10 Foods to Eat with Ulcerative Colitis | VisitJoy

Dr. Richard Hagmeyer D.C, CFMP — 5 Food Additives You Should Avoid If You have IBS/SIBO

Active Health Care — 10 Ultra-Processed Foods DESTROYING Your Immune System with Sjögren's Syndrome

The Nerve Doctors - Neuropathy Pain Treatment — Sea Moss: Nature’s Remedy for Nerves - The Nerve Doctors

Just In Health — Food Additives That Cause Gut Inflammation

Dr. Arland Hill — Alpha gal Syndrome: It’s Not a Meat Allergy, It’s an Immune System Warning

OFF GRID with DOUG & STACY — HAVING DIGESTIVE PROBLEMS? THIS COULD BE WHY!!

Fitness Camp — KeraGenis Reviews - ⚠️WARNING⚠️ Does KeraGenis Work or Scam?

American Society for Microbiology — Nasal Sprays and Respiratory Infections - Microbial Minutes

Dr Dray — Calamine Lotion Hacks Nobody Told You About

Embrace Health — C diff Diet: Safe Foods & Trigger Foods to Avoid

Mark Hyman, MD — A Doctor Ranks The Top 5 Cancer-Causing Foods