Potassium Sorbate (E202): The 'Safer' Preservative Under Scrutiny
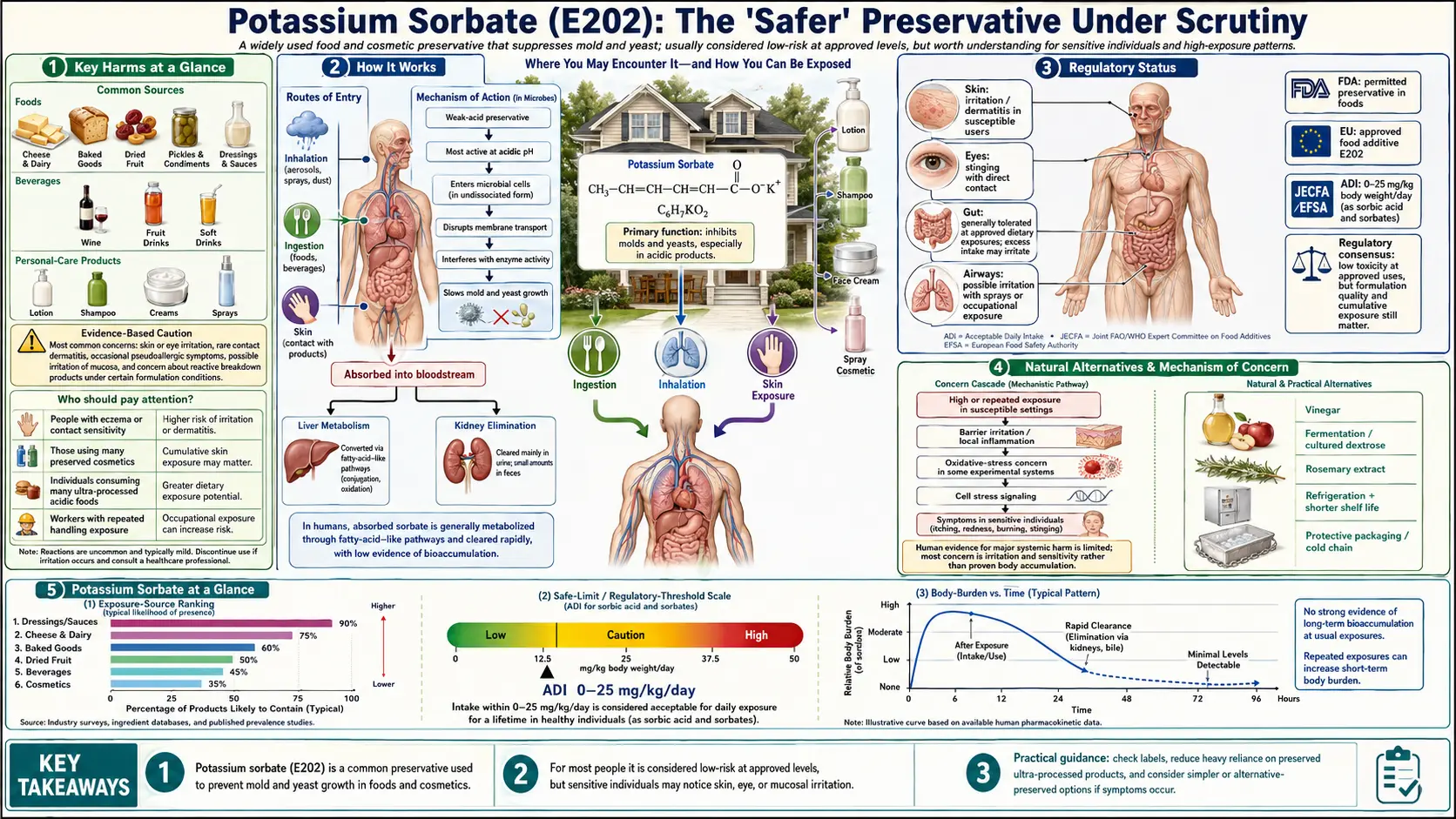
Table of Contents
- Key Harms at a Glance
- Overview
- Where It's Found (Exposure Routes)
- How It Works
- Health Effects
- Genotoxicity Research
- Regulatory Status & Safety Thresholds
- Combination Effects
- How to Avoid
- Natural Alternatives
- Research Papers
- Connections
- Featured Videos
Key Harms at a Glance
- Genotoxicity signal – Multiple in vitro studies show dose-dependent DNA damage, chromosomal aberrations, and micronucleus formation in human lymphocytes at 0.5–2 mM.
- Contact allergen – Recognized cause of allergic contact dermatitis in cosmetic and food-industry settings.
- Gut microbiome disruption – Inhibits beneficial Lactobacillus species at achievable intestinal concentrations.
- Cocktail exposure – Near-universal co-formulation with sodium benzoate and other preservatives; additive genotoxic effects reported.
- Migraine trigger – Reported by sensitive individuals; controlled trials are limited.
- ADI – JECFA/EFSA set at 25 mg/kg body weight/day as sorbic acid equivalents.
1. Overview
Potassium sorbate (E202) is the potassium salt of sorbic acid, a naturally occurring polyunsaturated fatty acid. It is one of the most widely used food preservatives globally, found in thousands of packaged food products, personal care formulations, and pharmaceutical preparations. Its popularity stems largely from a reputation as being "generally considered safer" than older preservatives such as sodium benzoate or sulfur dioxide — a characterization that is accurate in terms of acute toxicity but increasingly challenged by research into chronic and genotoxic effects.
The active compound, sorbic acid, was first isolated in 1859 by German chemist August Wilhelm von Hofmann from the unripe berries of the rowan tree (Sorbus aucuparia). Commercial production of potassium sorbate began in the 1950s, and today it is synthesized industrially rather than extracted from natural sources. Despite its botanical origins, all commercially used potassium sorbate is synthetic.
Potassium sorbate is water-soluble, odorless at low concentrations, and highly stable across a wide range of processing conditions, making it technically versatile. Its safety profile is often cited as favorable, but emerging research — particularly genotoxicity studies from the 2000s onward — has prompted regulators and researchers to take a more cautious look at long-term exposure effects.
2. Where It's Found
Potassium sorbate is used across an exceptionally broad range of product categories:
- Cheese and dairy products: Cottage cheese, shredded and sliced cheeses, processed cheese spreads, and sour cream commonly contain potassium sorbate to prevent surface mold growth.
- Wine: Used at concentrations up to 200 mg/L in wines to prevent refermentation after bottling; the EU permits up to 200 mg/L (Regulation EC No 606/2009).
- Baked goods: Cakes, muffins, tortillas, and bread products, particularly those with extended shelf lives.
- Dried fruits: Figs, dates, raisins, and prunes often receive potassium sorbate treatment to suppress mold during extended storage.
- Margarine and spreads: Prevents yeast and mold growth in water-in-oil emulsions.
- Canned fruits and vegetables: Used in pickled products and some acidified canned goods.
- Personal care products: A very common preservative in shampoos, conditioners, lotions, creams, and cosmetics, where it typically appears in the 0.1–0.3% concentration range.
- Pharmaceutical products: Used as a preservative in liquid oral medications, topical preparations, and ophthalmic solutions.
- Herbal and dietary supplements: Liquid and gel-cap supplements frequently contain potassium sorbate as a shelf-life extender.
The ubiquity of potassium sorbate means that daily cumulative exposure, when food and personal care products are considered together, can be substantially higher than regulatory estimates based on food intake alone.
3. How It Works
Potassium sorbate functions primarily as an antifungal agent, inhibiting the growth of molds and yeasts. It is significantly less effective against bacteria, which limits its use in products where bacterial contamination is the primary concern.
The mechanism of action involves multiple pathways:
- Cell membrane disruption: Sorbic acid interferes with the transport proteins embedded in microbial cell membranes, impairing nutrient uptake and waste excretion.
- Enzyme inhibition: It inhibits dehydrogenase enzymes involved in the citric acid cycle, disrupting cellular energy metabolism.
- Spore germination inhibition: At relevant concentrations, potassium sorbate suppresses the germination of fungal spores rather than killing established cells outright.
Efficacy is strongly pH-dependent. Potassium sorbate performs optimally at pH levels below 6.5; as pH rises toward neutral and alkaline conditions, the proportion of sorbate anion increases while free sorbic acid decreases, substantially reducing antimicrobial activity. This pH sensitivity means it is most effective in acidic foods such as cheese, wine, fermented products, and fruit-based items.
4. Health Effects
The conventional toxicological profile of potassium sorbate appears reassuring. The LD50 in rats is approximately 4,920 mg/kg body weight (oral), placing it in the same low-toxicity range as table salt. Acute toxicity at dietary concentrations is not a recognized concern.
However, several areas of chronic and subclinical health impact have drawn increasing research attention:
- Genotoxicity: Multiple peer-reviewed studies (detailed in Section 5) have documented DNA damage in human cells at concentrations within or close to dietary exposure ranges. This is the most significant unresolved safety concern.
- Skin sensitization and allergic contact dermatitis: Potassium sorbate is a recognized contact allergen, particularly in cosmetic and personal care applications. Patch-test positive reactions have been documented in atopic individuals, and occupational sensitization has been reported in food industry workers (Soschin & Hashim, 2016).
- Migraine triggering: Anecdotal and some clinical reports suggest potassium sorbate may trigger migraines in sensitive individuals, though controlled mechanistic studies are limited.
- Gut microbiome effects: Research has shown that potassium sorbate inhibits Lactobacillus species — key beneficial bacteria of the human gut — at concentrations achievable in the intestinal environment following dietary consumption. A 2019 study by Feng et al. demonstrated significant growth inhibition of Lactobacillus rhamnosus and Lactobacillus acidophilus at concentrations of 1–5 mM, raising questions about the net effect of regular dietary exposure on microbiome composition.
5. Genotoxicity Research
The genotoxic potential of potassium sorbate has been the subject of sustained scientific debate since the mid-2000s. The body of evidence is not conclusive, but it is substantial enough to warrant serious consideration.
Mamur et al. (2010) — Published in Food and Chemical Toxicology, this landmark study examined the genotoxic effects of potassium sorbate in cultured human peripheral lymphocytes. At concentrations of 0.5, 1.0, and 2.0 mM — which the authors argued were within the range achievable in human blood following normal dietary intake — potassium sorbate induced statistically significant increases in chromosomal aberrations, sister chromatid exchanges (SCE), and micronucleus formation. The effect was dose-dependent. The authors concluded that potassium sorbate exhibits mutagenic activity in human lymphocytes at food-relevant concentrations. (DOI: 10.1016/j.fct.2010.06.032)
Mpountoukas et al. (2008) — An earlier study published in Food and Chemical Toxicology similarly found dose-dependent DNA damage in human lymphocytes exposed to potassium sorbate at 0.5–2 mM concentrations, using the comet assay and SCE analysis. The findings corroborated the Mamur results and reinforced concerns about in vivo relevance. (DOI: 10.1016/j.fct.2008.09.062)
Debate over in vitro relevance: Critics of these studies note that the concentrations used in cell culture experiments are difficult to translate directly to in vivo systemic exposure. Potassium sorbate is metabolized in the human body similarly to other fatty acids — oxidized via beta-oxidation — and systemic blood levels from dietary exposure may not routinely reach the 0.5–2 mM range used in vitro. EFSA's 2015 re-evaluation of sorbic acid and potassium sorbate acknowledged the genotoxicity findings but concluded that in vivo studies did not confirm genotoxicity at relevant exposure levels, and maintained the acceptable daily intake (ADI) of 25 mg/kg body weight/day.
The critical unresolved question is whether the in vitro genotoxicity signals translate to meaningful human health risk at realistic long-term dietary exposures, particularly in children who may consume proportionally higher amounts relative to body weight.
6. Regulatory Status
Potassium sorbate holds approval across all major regulatory jurisdictions:
- United States (FDA): Generally Recognized As Safe (GRAS) status. Permitted in numerous food categories with specific use limitations under 21 CFR.
- European Union: Approved as E202 under Regulation (EC) No 1333/2008. EFSA completed a full re-evaluation in 2015, maintaining approval with an ADI of 25 mg/kg body weight/day (expressed as sorbic acid equivalents).
- Organic foods: Notably, potassium sorbate is one of a very small number of synthetic preservatives permitted in USDA certified organic products (listed in the National Organic Program's National List of Allowed and Prohibited Substances). It is permitted in organic wine production at the same limit as conventional wine.
- Codex Alimentarius: The Joint FAO/WHO Expert Committee on Food Additives (JECFA) has evaluated sorbic acid and its salts, establishing an ADI of 0–25 mg/kg body weight/day, consistent with EFSA.
- Labeling: In the EU, it must be declared as "potassium sorbate" or "E202" in ingredient lists. In the US, it must be listed by name.
7. Combination Effects
Potassium sorbate is rarely used in isolation in commercial food products. Its combination with other preservatives raises "cocktail effect" concerns that are not adequately captured by single-substance safety assessments:
- Potassium sorbate + sodium benzoate: This combination is extremely common in carbonated and non-carbonated beverages, salad dressings, and condiments. While each compound individually has an established safety profile, the combination has been less thoroughly studied. The Mamur group examined the combination in a 2011 follow-up study and found additive genotoxic effects in human lymphocytes.
- Potassium sorbate + nitrites in cured meats: Sorbate is sometimes used alongside sodium nitrite in processed meat products. Under conditions of high heat (cooking) or acidic stomach pH, interactions between sorbate and nitrite can theoretically produce mutagenic compounds, though the clinical significance of this pathway remains debated.
- Cumulative dietary exposure: Given potassium sorbate's presence in both food and personal care products, total daily exposure estimates based on food alone may substantially undercount actual systemic exposure, particularly for individuals using multiple cosmetic products daily.
8a. How to Avoid
- Read ingredient labels for “potassium sorbate,” “sorbic acid,” or “E202.”
- Choose USDA Organic wines without added sorbate, or fermented-dry wines that do not require post-bottling stabilization.
- Prefer fresh cheeses sold in wax-rind or brine formats rather than shrink-wrapped shredded/sliced cheese.
- Replace cosmetic/personal-care products with formulations preserved by phenethyl alcohol, ethylhexylglycerin, or fermentation-derived acids.
- Buy dried fruit that is sulfite- and sorbate-free (read labels carefully; “no preservatives added” is a regulated claim).
- Limit processed baked goods with long shelf lives; fresh-baked alternatives typically omit sorbate.
8. Natural Alternatives
Several naturally derived and fermentation-based preservation strategies can substitute for potassium sorbate in specific applications:
- Natamycin (E235): A natural antifungal antibiotic from Streptomyces natalensis, highly effective against molds on cheese surfaces and cured meats. Unlike potassium sorbate, natamycin does not penetrate food matrices, limiting its use to surface applications.
- Fermentation-based preservation: Lactic acid fermentation lowers pH and produces natural antimicrobial metabolites (lactic acid, acetic acid, bacteriocins), providing broad-spectrum preservation without synthetic additives.
- Rosemary extract: Rich in carnosic acid and rosmarinic acid, rosemary extract provides antioxidant and mild antimicrobial activity, approved as a natural food antioxidant (E392) in the EU. Most effective against oxidative spoilage rather than microbial growth.
- Vinegar and organic acids: In acidic products, natural acetic acid (vinegar) or citric acid can substitute part of the antimicrobial function attributed to potassium sorbate.
9. Connections
- Preservatives
- Natamycin
- Sodium Nitrite
- Sodium Benzoate
- BHA and BHT
- Nisin
- US vs EU Food Safety
- Sulfites
- Acesulfame Potassium
- Food Additives
- Potassium
- Gut Microbiome
- Migraine
- Banned Additives
- Food Dyes Banned Worldwide
- Allergies
- Rosemary
10. Research Papers
- Mamur S, Yüzbaşıoğlu D, Ünal F, Yılmaz S. Does potassium sorbate induce genotoxic or mutagenic effects in lymphocytes? Toxicology in Vitro. 2010;24(3):790–794.
- Mamur S, Yüzbaşıoğlu D, Ünal F, Aksoy H, Yılmaz S. Genotoxicity of food preservative sodium sorbate in lymphocytes in vitro. Cytotechnology. 2012;64(5):553–562.
- Mpountoukas P, Vantarakis A, Sivridis E, Lialiaris T. Cytogenetic study in cultured human lymphocytes treated with three commonly used preservatives. Food and Chemical Toxicology. 2008;46(8):2390–2393.
- EFSA Panel on Food Additives and Nutrient Sources. Re-evaluation of sorbic acid (E 200), potassium sorbate (E 202) and calcium sorbate (E 203) as food additives. EFSA Journal. 2015;13(6):4144.
- Feng T, Wang J, Shao S, Zhao Y. Inhibitory effects of common food preservatives on the growth of Lactobacillus. Food Control. 2019;105:182–188.
- Soschin D, Hashim PW. Sorbic acid and potassium sorbate as contact allergens in cosmetics. Dermatitis. 2016;27(1):3–7.
- Davidson PM, Critzer FJ, Taylor TM. Naturally occurring antimicrobials for minimally processed foods. Annual Review of Food Science and Technology. 2013;4:163–190.
- Commission Regulation (EC) No 606/2009 laying down certain detailed rules for implementing Council Regulation (EC) No 479/2008 as regards the categories of grapevine products, the oenological practices and the applicable restrictions. Official Journal of the European Union. 2009.
- JECFA. Sorbic acid. In: WHO Food Additives Series 5. Joint FAO/WHO Expert Committee on Food Additives; 1974. WHO Geneva.
- Brul S, Coote P. Preservative agents in foods: mode of action and microbial resistance mechanisms. International Journal of Food Microbiology. 1999;50(1–2):1–17.
- Neltner TG, Alger HM, O'Reilly JT, Krimsky S, Bero LA, Maffini MV. Conflicts of interest in approvals of additives to food determined to be generally recognized as safe. JAMA Internal Medicine. 2013;173(22):2032–2036.
- Carocho M, Barreiro MF, Morales P, Ferreira IC. Adding molecules to food, pros and cons: a review on synthetic and natural food additives. Comprehensive Reviews in Food Science and Food Safety. 2014;13(4):377–399.
- Mihail RC. Oral food challenges with sorbate and benzoate preservatives: a clinical study. Annals of Allergy, Asthma & Immunology. 1992;68(5):423–428.
- National Organic Program. National List of Allowed and Prohibited Substances. 7 CFR Part 205. USDA Agricultural Marketing Service. 2023.
Featured Videos

Potassium Sorbate: A Safe and Versatile Way to Extend Shelf Life. WTF - Ep. 319

Allergies: My Experience with Solving my own Allergy to Potassium Sorbate

Sorbate and Sulfite in Wine and Mead Making

Can You Taste Sorbate & Metabisulfite? (1 Year Later)

4 Common Myths About Potassium | Dispelling Myths About Potassium Related Kidney Diet

Is potassium good for high blood pressure? (Benefits of potassium!)

4 Terrible Common Preservatives

CHEMICAL IN PROCESSED FOOD AND OUR HEALTH.. chemical poisons in packed food affecting health..

Are preservatives safe to use?