Interstitial Cystitis
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation
- Diagnosis
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Research Papers
- Connections
- Featured Videos
1. Overview
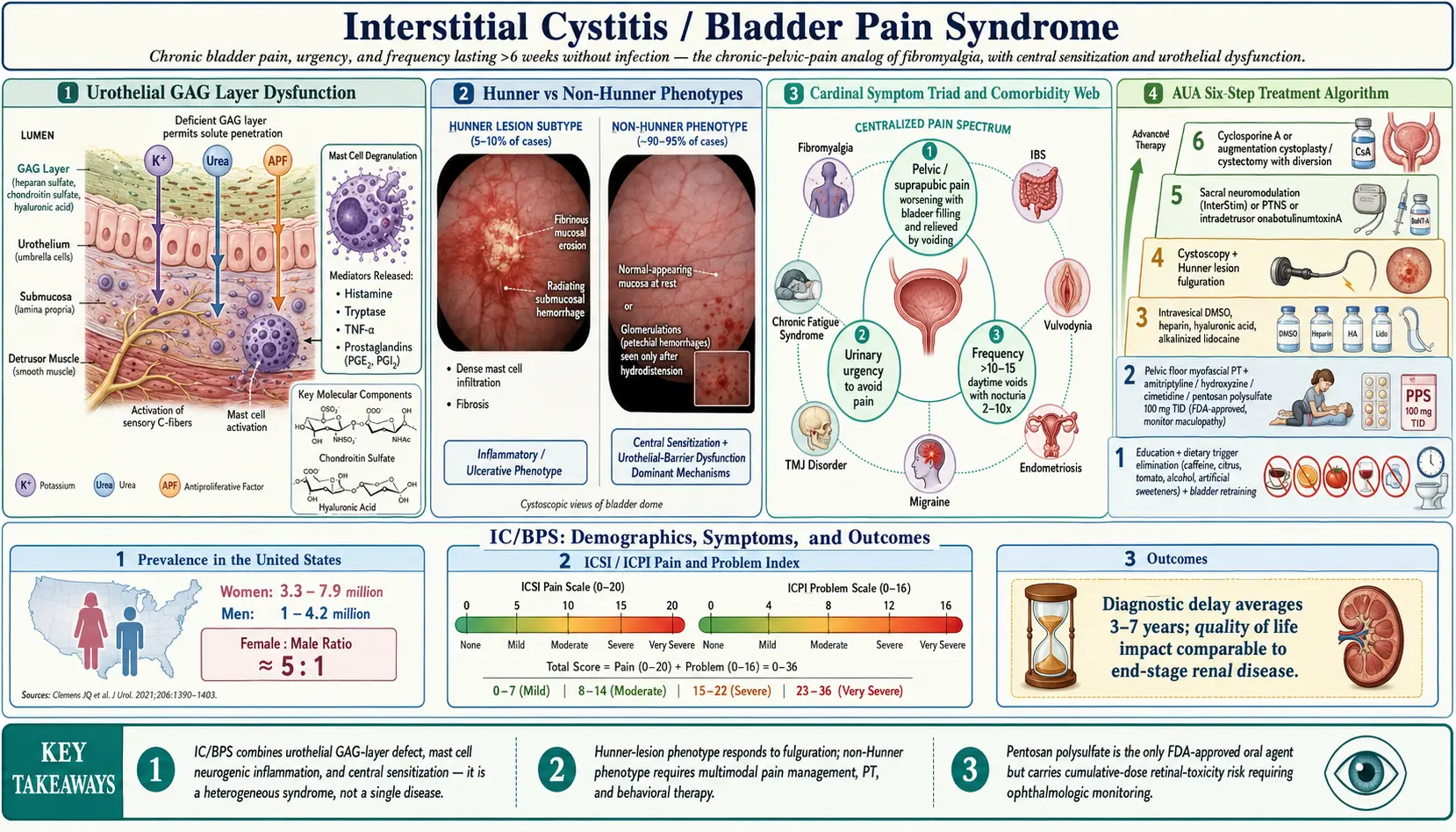
Interstitial Cystitis/Bladder Pain Syndrome (IC/BPS) is a chronic, debilitating condition of the urinary bladder characterized by pelvic pain, pressure, or discomfort perceived to be related to the bladder, accompanied by lower urinary tract symptoms lasting more than six weeks in the absence of infection or other identifiable causes. The American Urological Association (AUA) and Society of Urodynamics, Female Pelvic Medicine and Urogenital Reconstruction (SUFU) define it as an unpleasant sensation (pain, pressure, discomfort) perceived to be related to the bladder and associated with lower urinary tract symptoms of more than six weeks duration.
IC/BPS was formerly considered an exclusively inflammatory condition of the bladder wall, but is now conceptualized as a heterogeneous syndrome likely encompassing multiple phenotypes with distinct pathophysiologic mechanisms. The condition profoundly affects quality of life, with a negative impact on sexual function, sleep, work productivity, and mental health comparable to end-stage renal disease.
2. Epidemiology
Prevalence estimates for IC/BPS vary widely depending on diagnostic criteria employed. The RAND Interstitial Cystitis Epidemiology (RICE) study estimated that approximately 3.3–7.9 million women and 1.0–4.2 million men in the United States fulfill IC/BPS symptom criteria, yielding a female-to-male prevalence ratio of approximately 5:1. Age-specific prevalence peaks in the fourth to sixth decades of life.
Diagnosis is frequently delayed by 3–7 years from symptom onset, partly because IC/BPS overlaps clinically with other chronic pelvic pain conditions. Approximately 50% of IC/BPS patients have at least one comorbid pain condition, most commonly fibromyalgia, irritable bowel syndrome, chronic fatigue syndrome, vulvodynia, or temporomandibular joint disorder, suggesting shared central sensitization mechanisms.
3. Pathophysiology
The pathophysiology of IC/BPS is multifactorial and incompletely understood. Several interacting mechanisms have been proposed:
Urothelial Dysfunction
The urothelium is coated by a glycosaminoglycan (GAG) layer composed predominantly of heparan sulfate, chondroitin sulfate, and hyaluronic acid, which protects the underlying epithelium from urine constituents. In IC/BPS, deficiency or dysfunction of the GAG layer allows urinary solutes (potassium, urea, antiproliferative factor) to penetrate the urothelium, activating subepithelial sensory C-fibers and mast cells. Antiproliferative factor (APF), a frizzled-8-related sialoglycopeptide, is overproduced by IC/BPS urothelium and inhibits normal urothelial repair and proliferation.
Mast Cell Activation
Submucosal and detrusor mast cell infiltration is a consistent histological finding in the Hunner's lesion subtype of IC/BPS. Mast cell degranulation releases histamine, tryptase, TNF-alpha, and prostaglandins, contributing to neurogenic inflammation, sensory fiber sensitization, and symptom perpetuation.
Neurogenic Inflammation and Central Sensitization
Chronic bladder nociceptor activation leads to peripheral and central sensitization. Upregulation of substance P, calcitonin gene-related peptide (CGRP), and nerve growth factor (NGF) within the bladder wall perpetuates pain signaling independent of ongoing tissue injury. Functional MRI studies demonstrate altered brain connectivity and gray matter volume changes in pain-modulating regions (anterior cingulate cortex, insula) analogous to other centralized pain disorders.
Autoimmune Mechanisms
Autoantibodies targeting urothelial antigens (uroplakin, cytokeratins) have been detected in some IC/BPS patients. The female predominance and association with other autoimmune conditions support an autoimmune contribution in a subset of patients.
Pelvic Floor Dysfunction
Pelvic floor hypertonicity with myofascial trigger points is present in the majority of IC/BPS patients and may independently generate or amplify pelvic pain through referred viscerosomatic pathways.
4. Etiology and Risk Factors
- Female sex: Women constitute approximately 80–90% of diagnosed cases, though male prevalence is likely underestimated due to differential diagnostic attribution to chronic prostatitis/chronic pelvic pain syndrome (CP/CPPS).
- Prior urinary tract infections: A history of recurrent UTIs is reported by 50–85% of IC/BPS patients; it remains unclear whether infections trigger IC/BPS or reflect shared susceptibility.
- Genetic predisposition: Familial clustering and twin studies suggest a heritable component. Polymorphisms in genes regulating urothelial repair, immune function, and nociception are under study.
- Pelvic surgery or trauma: Prior hysterectomy, pelvic radiation, or obstetric trauma may precipitate IC/BPS.
- Autoimmune and allergic conditions: Systemic lupus erythematosus, Sjogren's syndrome, allergic rhinitis, and atopy are overrepresented in IC/BPS populations.
- Psychological factors: Anxiety, depression, and adverse childhood experiences (ACEs) increase IC/BPS risk, possibly through neuroimmune pathways.
- Bladder microbiome alterations: Emerging evidence implicates dysbiosis of the urine and bladder microbiome in IC/BPS pathogenesis.
5. Clinical Presentation
The cardinal symptom triad consists of:
- Bladder/pelvic pain: Typically suprapubic, worsening as the bladder fills and relieved (at least partially) by voiding. Pain may radiate to the urethra, vagina, perineum, or inner thighs.
- Urinary urgency: Perceived need to void to avoid worsening pain rather than fear of leakage.
- Urinary frequency: Often pronounced, with daytime frequency exceeding 10–15 voids and multiple nocturnal awakenings (nocturia 2–10 times per night).
Dyspareunia is reported by up to 80% of women with IC/BPS. Symptom flares are triggered by dietary factors (acidic foods, caffeine, alcohol, artificial sweeteners), emotional stress, sexual activity, menstruation, and prolonged sitting.
IC/BPS Phenotypes
Two clinically and histologically distinct phenotypes are recognized:
- Hunner Lesion IC (HIC): Formerly "classic IC." Cystoscopic Hunner lesions — discrete inflammatory mucosal erosions with radiating submucosal hemorrhage — are present in 5–10% of IC/BPS patients, predominantly older women with severe disease. Associated with greater bladder wall inflammation, mast cell infiltration, higher pain scores, and lower maximum cystometric capacity.
- Non-Hunner Lesion IC (NHIC): The majority of IC/BPS. Cystoscopy may be normal or show glomerulations (petechial hemorrhages) after hydrodistension. Urothelial barrier dysfunction and central sensitization are more prominent pathomechanisms.
Validated Symptom Assessment Tools
- O'Leary-Sant Symptom Index (OSSI) and Problem Index (OSPI): Validated questionnaires assessing symptom frequency and bother.
- ICSI/ICPI (Interstitial Cystitis Symptom and Problem Index): Most widely used clinical and research tool.
- Bladder diary: 72-hour voiding diary documenting voided volumes, frequency, urgency episodes, and pain scores.
- Pelvic Pain and Urgency/Frequency (PUF) Patient Symptom Scale: Screens for IC/BPS and guides phenotyping.
6. Diagnosis
IC/BPS is a diagnosis of exclusion. The AUA diagnostic algorithm requires exclusion of confusable conditions before establishing the diagnosis.
Initial Evaluation
- Detailed history: Symptom onset, character, and location; voiding diary; prior pelvic surgeries; medication history; comorbid pain conditions.
- Physical examination: Abdominal and bimanual pelvic examination to assess tenderness, masses, prolapse, and pelvic floor hypertonicity (levator ani tenderness on internal palpation).
- Urinalysis and urine culture: To exclude bacterial cystitis, hematuria, and pyuria. Dipstick positive nitrites/leukocyte esterase with a negative culture may prompt evaluation for fastidious organisms.
- Urine cytology: To exclude urothelial carcinoma in situ (CIS), particularly in older patients or those with risk factors (smoking, chemical exposure).
Potassium Sensitivity Test (PST)
Intravesical instillation of 0.4 M potassium chloride produces pain in approximately 70–80% of IC/BPS patients versus 5% of controls, reflecting urothelial permeability dysfunction. A positive PST supports the diagnosis and predicts response to intravesical heparin. However, the test is neither sensitive nor specific enough for diagnostic use in isolation and is no longer routinely recommended by current guidelines.
Cystoscopy with Hydrodistension
Recommended when Hunner lesions are suspected or when the diagnosis remains uncertain after initial evaluation. Performed under anesthesia at low pressure (60–80 cmH2O for 2–3 minutes). Findings include:
- Hunner lesions: Pathognomonic for HIC; appear as erythematous, velvety, fibrinous erosions typically at the bladder dome or posterior wall.
- Glomerulations: Diffuse petechial hemorrhages following hydrodistension; present in NHIC but also in asymptomatic individuals; low diagnostic specificity.
- Reduced bladder capacity: Maximum anesthetic bladder capacity below 400 mL is associated with greater disease severity.
Bladder Biopsy
Not required for routine diagnosis but aids in excluding bladder malignancy (CIS), confirming Hunner lesion histology (urothelial denudation, submucosal mast cell infiltration, fibrosis), and characterizing disease phenotype for research purposes.
Urodynamics
Multichannel urodynamics are indicated when the diagnosis is uncertain, to exclude detrusor overactivity, or prior to invasive treatment. Typical IC/BPS urodynamic findings include reduced bladder capacity, pain reproduction at low fill volumes, and occasional hypersensitive first desire to void without demonstrable detrusor overactivity.
7. Treatment
Treatment is individualized and stepwise. The AUA guideline employs a six-step treatment algorithm progressing from least to most invasive.
First-Line: Education and Behavioral/Dietary Modification
- Patient education regarding the chronic nature of IC/BPS and realistic treatment expectations.
- Dietary elimination of known bladder irritants: caffeine, alcohol, carbonated beverages, citrus fruits, tomatoes, spicy foods, artificial sweeteners, and vitamin C supplements.
- Bladder retraining (timed voiding, gradually increasing inter-void intervals) to increase functional bladder capacity.
- Stress reduction techniques: cognitive behavioral therapy, mindfulness, biofeedback.
Second-Line: Physical Therapy and Oral Agents
- Pelvic floor physical therapy: Manual myofascial release therapy targeting pelvic floor trigger points is superior to global therapeutic massage in IC/BPS (Peters et al., 2012). First-line for patients with pelvic floor hypertonicity.
- Amitriptyline: Tricyclic antidepressant; modulates central pain sensitization, reduces mast cell activation, and decreases urgency. Starting dose 10–25 mg at bedtime, titrated to 50–100 mg. Anticholinergic side effects limit use.
- Cimetidine: H2-receptor antagonist; reduces histamine-mediated mast cell effects; modest symptom benefit in case series.
- Hydroxyzine: H1 antihistamine with mast cell stabilizing properties; particularly useful in patients with allergy history.
- Pentosan polysulfate sodium (PPS, Elmiron): The only FDA-approved oral agent for IC/BPS. A semi-synthetic heparin analogue that replenishes deficient GAG layer. Dose: 100 mg three times daily. Onset of benefit requires 3–6 months. A randomized trial showed modest superiority over placebo. Important safety note: Cumulative exposure above 500–1500 g is associated with pigmentary maculopathy, a form of retinal toxicity; ophthalmologic monitoring is recommended.
Third-Line: Intravesical Therapies
- Dimethyl sulfoxide (DMSO): Only FDA-approved intravesical therapy for IC/BPS. Mechanism includes anti-inflammatory, analgesic, and mast cell stabilizing effects. Administered as 50 mL of 50% solution instilled and retained for 15–20 minutes every 1–2 weeks for 6–8 cycles. Response rates of 50–70%.
- Intravesical heparin: Exogenous GAG replacement; often combined with alkalinized lidocaine and sodium bicarbonate in compounded instillation cocktails.
- Intravesical hyaluronic acid and chondroitin sulfate: Commercially available in some countries (not FDA-approved in the US); data support modest efficacy for GAG layer restoration.
- Intravesical lidocaine: Alkalinized lidocaine (200–300 mg in bicarbonate solution) provides rapid, if transient, pain relief; useful for diagnostic confirmation and symptom rescue.
Fourth-Line: Cystoscopy with Hydrodistension and Hunner Lesion Treatment
- Hunner lesion fulguration or steroid injection: Transurethral laser ablation or electrofulguration of Hunner lesions provides 50–90% short-term symptom improvement in HIC patients, though recurrence is expected within 1–2 years. Intralesional triamcinolone injection is an alternative with comparable efficacy.
- Hydrodistension: Provides temporary relief in approximately 20–30% of patients; mechanism may involve ischemic afferent nerve ablation.
Fifth-Line: Neuromodulation
- Sacral neuromodulation (SNM, InterStim): Modulates sacral nerve reflex arcs controlling bladder and pelvic floor function. Approximately 60–70% of carefully selected IC/BPS patients achieve 50% or greater symptom improvement. A test stimulation phase precedes permanent implantation.
- Percutaneous tibial nerve stimulation (PTNS): Less invasive neuromodulation option; 12-week course of weekly 30-minute sessions with ongoing maintenance treatment.
- Botulinum toxin A (onabotulinumtoxinA): Intradetrusor injection (100–200 U) inhibits afferent nociceptive signaling. Multiple randomized trials demonstrate significant symptom reduction; urinary retention requiring self-catheterization occurs in 10–20% of patients.
Sixth-Line: Cyclosporine A and Major Surgery
- Cyclosporine A: Calcineurin inhibitor with potent immunosuppressive and mast cell inhibiting effects. Oral dosing at 1.5–3 mg/kg/day produces clinically meaningful improvement in HIC patients, superior to PPS in controlled trials. Nephrotoxicity, hypertension, and infection risks require careful monitoring.
- Major reconstructive surgery: Reserved for refractory cases with severe bladder contraction (capacity below 150–200 mL under anesthesia):
- Augmentation cystoplasty: Detubularized ileal or ileocecal patch enlarges the bladder reservoir; pain relief is inconsistent and requires lifelong self-catheterization in some patients.
- Urinary diversion with or without cystectomy: Continent or incontinent diversion offers pain relief in 70–80% of carefully selected patients with confirmed bladder-origin pain.
8. Complications
- Severe bladder contracture: End-stage fibrotic bladder with capacity below 50–100 mL; requires surgical reconstruction.
- Sexual dysfunction: Dyspareunia and decreased libido affect the majority of women; erectile dysfunction is common in men with IC/BPS.
- Psychiatric comorbidity: Depression and anxiety are two to four times more prevalent in IC/BPS patients compared to the general population, contributing to suicidal ideation in severe cases.
- Sleep disruption: Nocturia and pain-related insomnia produce chronic sleep deprivation with downstream effects on cognitive function, immune regulation, and pain thresholds.
- Occupational and social disability: Frequent voiding needs and chronic pain impair workplace productivity, social engagement, and travel.
- Pentosan polysulfate maculopathy: Progressive retinal pigment epitheliopathy with potential permanent vision loss in long-term PPS users.
9. Prognosis
IC/BPS follows a fluctuating course characterized by periods of symptom flare and partial remission. Spontaneous remission occurs in approximately 50% of patients within 8 months of diagnosis in some series, though many relapse. Predictors of favorable prognosis include younger age, shorter symptom duration, absence of Hunner lesions, and lower pain catastrophizing scores.
In the majority of patients, symptoms are managed rather than cured. Approximately 20% of patients progress to severe, refractory disease requiring major surgical intervention. Multidisciplinary management incorporating urologic, physical therapy, and psychological components produces the best long-term outcomes.
10. Prevention
No proven primary prevention strategy exists for IC/BPS given its incompletely understood etiology. Secondary prevention efforts focus on:
- Avoidance of dietary bladder irritants to minimize symptom flares.
- Prompt treatment of bacterial cystitis to prevent urothelial disruption.
- Stress management and treatment of underlying anxiety/depression.
- Preservation of pelvic floor integrity through appropriate obstetric care and physiotherapy.
- Ophthalmologic surveillance for patients on long-term pentosan polysulfate sodium.
11. Recent Research and Advances
Bladder microbiome: Advanced sequencing studies using enhanced quantitative urine culture (EQUC) and 16S rRNA analysis have identified distinct microbiome signatures in IC/BPS, including reduced Lactobacillus abundance and increased proportions of anaerobic genera. Probiotic and microbiome-modulating interventions are under investigation.
Urine biomarkers: Antiproliferative factor (APF), epidermal growth factor (EGF), heparin-binding EGF (HB-EGF), and urinary cytokine panels are being evaluated as diagnostic and prognostic biomarkers to enable phenotype-specific treatment selection.
Low-intensity extracorporeal shockwave therapy (Li-ESWT): Pilot trials report significant pain reduction and improved voiding in IC/BPS patients, hypothesized via neovascularization, anti-inflammatory, and neural regeneration mechanisms.
Cannabinoids: Endocannabinoid receptor (CB1, CB2) expression in urothelium and bladder sensory neurons provides a pharmacological rationale for cannabinoid-based analgesia. Early clinical data are promising but require confirmation in powered randomized trials.
Transcranial magnetic stimulation (TMS) and transcranial direct current stimulation (tDCS): Targeting central sensitization circuits, early studies report analgesic benefit in IC/BPS consistent with other centralized pain disorders.
Subcutaneous clenbuterol: Beta-2 adrenergic agonist activity on urothelial and neural tissue is under investigation as a novel mechanism for reducing bladder hypersensitivity.
12. References
- Hanno PM, Burks DA, Clemens JQ, et al. AUA guideline for the diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol. 2011;185(6):2162–2170.
- Clemens JQ, Erickson DR, Varela NP, Lai HH. Diagnosis and treatment of interstitial cystitis/bladder pain syndrome. J Urol. 2022;208(1):34–42.
- Berry SH, Elliott MN, Suttorp M, et al. Prevalence of symptoms compatible with a diagnosis of interstitial cystitis/painful bladder syndrome in the United States. J Urol. 2011;186(2):540–544.
- Peters KM, Carrico DJ, Kalinowski SE, Ibrahim IA, Diokno AC. Prevalence of pelvic floor dysfunction in patients with interstitial cystitis. Urology. 2007;70(1):16–18.
- Peters KM, Carrico DJ, Kalinowski SE, et al. Randomized trial of pelvic floor physical therapy versus global therapeutic massage for patients with interstitial cystitis/bladder pain syndrome. J Urol. 2012;187(6):2113–2118.
- Nickel JC, Herschorn S, Whitmore KE, et al. Pentosan polysulfate sodium for treatment of interstitial cystitis/bladder pain syndrome: insights from a randomized, double-blind, placebo controlled study. J Urol. 2015;193(3):857–862.
- Pearce WA, Chen R, Jain N. Pigmentary maculopathy associated with chronic exposure to pentosan polysulfate sodium. Ophthalmology. 2018;125(11):1793–1802.
- Maher CF, Carey MP, Dwyer PL, Schluter PL. Percutaneous sacral nerve root neuromodulation for intractable interstitial cystitis. J Urol. 2001;165(3):884–886.
- Hanno P, Andersson KE, Birder L, et al. Chronic pelvic pain syndrome/bladder pain syndrome: taking stock, looking ahead. Neurourol Urodyn. 2015;34(7):641–648.
- Logadottir Y, Delbro D, Fall M, et al. Inflammation characteristics in bladder pain syndrome ESSIC type 3C (classic interstitial cystitis). Int J Urol. 2014;21(Suppl 1):75–78.
- Liu HT, Kuo HC. Urinary nerve growth factor level could be a potential biomarker for diagnosis of overactive bladder. J Urol. 2008;179(6):2270–2274.
- Nickel JC, Moldwin R, Lee S, Davis EL, Henry RA, Wyllie MG. Intravesical alkalinized lidocaine (PSD597) offers sustained relief from symptoms of interstitial cystitis and painful bladder syndrome. BJU Int. 2009;103(7):910–918.
- Gee WF, Holtgrewe HL, Albertsen PC, et al. Practice trends in the diagnosis and management of benign prostatic hyperplasia in the United States. J Urol. 1995;154(1):205–208.
- Birder L, Andersson KE. Urothelial signaling. Physiol Rev. 2013;93(2):653–680.
- Warren JW, Meyer WA, Greenberg P, Horne L, Diggs C, Tracy JK. Using the International Continence Society's definition of painful bladder syndrome. Urology. 2006;67(6):1138–1142.
- Cervigni M, Natale F. The use of hyaluronans in the treatment of interstitial cystitis/bladder pain syndrome. Ther Adv Urol. 2012;4(3):107–120.
Research Papers
- Clinical trials on interstitial cystitis / bladder pain syndrome — PubMed search
- Pentosan polysulfate (Elmiron) for IC — PubMed search
- Intravesical DMSO for IC — PubMed search
- Intravesical hyaluronic acid — PubMed search
- Pelvic floor physical therapy for IC — PubMed search
- Amitriptyline for chronic bladder pain — PubMed search
- Sacral neuromodulation for refractory bladder pain — PubMed search
- Dietary triggers in interstitial cystitis — PubMed search
Connections
- Urinary Tract Infections
- Irritable Bowel Syndrome
- Fibromyalgia
- Stress Management
- Bladder Cancer
- Quercetin
- Benign Prostatic Hyperplasia
- Chronic Pain
- Potassium
- Anxiety
- Depression
- Artificial Sweeteners
- Urinalysis
- Kidney Stones
- Endometriosis
- Laparoscopy Excision vs Ablation
- Pelvic Floor Therapy and Central Sensitization i
- Nociceptive vs Neuropathic vs Nociplastic Pain
Featured Videos

Does Your Bladder HURT?! A Review of Interstitial Cystitis Symptoms and Treatment

Bladder Pain Syndrome - Interstitial Cystitis - Symptoms and Treatment Overview - This is not UTI !

Interstitial Cystitis: 8 Myths Busted! IC Causes, Symptoms, and Treatment w/ Nicole Cozean

Interstitial Cystitis: What is It and How to Treat It

What People Don't Know About Interstitial Cystitis | Pelvic Health and Rehabilitation Center

What is interstitial cystitis (IC)? - Jean McDonald

How I Overcame Interstitial Cystitis (IC) with Diet and Treatments

What is Interstitial Cystitis and Painful Bladder Syndrome - Causes, Symptoms, and Treatment

Interstitial Cystitis: Diagnosis and Treatment