Bladder Cancer
Table of Contents
- Overview
- Epidemiology
- Pathophysiology
- Etiology and Risk Factors
- Clinical Presentation and TNM Staging
- Diagnosis — Cystoscopy, Urodynamics, and Biomarkers
- Treatment
- Complications
- Prognosis
- Prevention
- Recent Research and Advances
- References
- Research Papers
- Connections
- Featured Videos
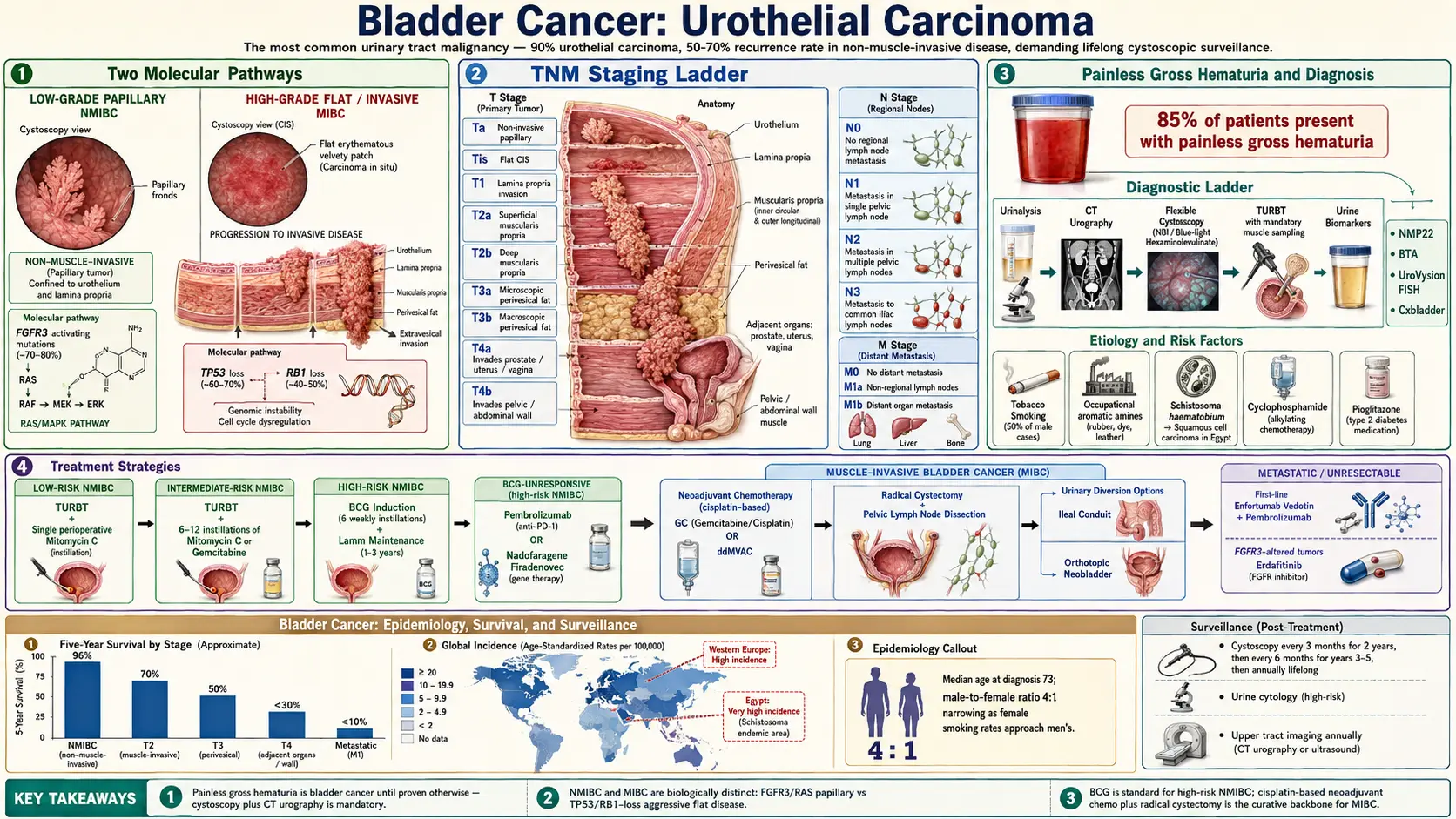
1. Overview
Bladder cancer is the most common malignancy of the urinary tract and the fourth most common cancer in men in Western nations. It encompasses a spectrum of tumors arising from the urothelium (transitional epithelium) lining the bladder, with distinct non-muscle-invasive (NMIBC) and muscle-invasive (MIBC) disease categories that carry fundamentally different prognoses and therapeutic approaches. Urothelial carcinoma (transitional cell carcinoma) accounts for approximately 90% of bladder cancers in industrialized countries, with squamous cell carcinoma (5%) and adenocarcinoma (less than 2%) comprising most remaining cases.
The disease is characterized by a high propensity for recurrence in non-muscle-invasive disease (50–70% within 5 years) and a significant risk of progression to muscle-invasive disease (10–30%), demanding lifelong surveillance and imposing one of the highest per-patient treatment costs among all cancers.
2. Epidemiology
Bladder cancer is the ninth most common cancer globally, with approximately 573,000 new cases and 213,000 deaths annually (GLOBOCAN 2020). Incidence is approximately four times higher in men than women, reflecting historical differences in smoking rates and occupational exposures. However, the male-to-female ratio is narrowing as female smoking prevalence approaches male rates in many countries.
In the United States, approximately 83,000 new cases and 17,000 deaths occur annually. Median age at diagnosis is approximately 73 years; the disease is rare before age 40. Geographic variation is substantial: highest incidence rates are seen in Egypt (driven by schistosomiasis-associated squamous cell carcinoma), Lebanon, and Western Europe; rates are lower in Asia and sub-Saharan Africa.
Socioeconomic disparities exist: Black patients present with more advanced disease and have worse stage-adjusted survival than White patients, reflecting differences in healthcare access, diagnostic delay, and potentially biological factors.
3. Pathophysiology
Urothelial carcinogenesis follows two principal molecular pathways corresponding to the two clinical phenotypes:
Low-Grade Papillary (NMIBC) Pathway
Characterized by activating mutations in the FGFR3 gene (fibroblast growth factor receptor 3) and HRAS, leading to constitutive activation of the RAS/MAPK signaling cascade. These tumors display a papillary architecture, low proliferation rate, and favorable prognosis. FGFR3 mutations are present in approximately 70–80% of low-grade papillary tumors. PIK3CA mutations contribute to activation of the PI3K/AKT/mTOR pathway in a subset.
High-Grade Flat/Invasive (CIS/MIBC) Pathway
Characterized by loss of function of TP53 (tumor protein p53) and RB1 (retinoblastoma), leading to cell cycle checkpoint failure, genomic instability, and aggressive biological behavior. Carcinoma in situ (CIS) — a flat, high-grade, non-invasive lesion — represents the precursor to muscle-invasive disease. Loss of CDKN2A (p16), PTEN deletion, and chromatin remodeling gene mutations (KDM6A, ARID1A, KMT2D) are also frequent in MIBC.
Molecular Subtypes of MIBC
Consensus molecular classification (TCGA, Lund taxonomy, Consensus classification 2019) identifies five major MIBC subtypes:
- Luminal papillary: FGFR3-altered, low immune infiltrate, best prognosis
- Luminal unstable: TP53 mutations, genomic instability
- Luminal non-specified: Moderate prognosis; E-cadherin expression preserved
- Stroma-rich: High stromal content; variable prognosis
- Basal/squamous: Loss of E-cadherin, KRT5/14 expression, squamous differentiation; aggressive but potentially chemosensitive; characterized by immune infiltrate in some subsets
- Neuroendocrine-like: Rare; extremely aggressive; similar to small cell carcinoma
4. Etiology and Risk Factors
Tobacco Smoking
The single most important modifiable risk factor, accounting for approximately 50% of bladder cancer cases in men and 30% in women. Carcinogens in tobacco smoke (aromatic amines, polycyclic aromatic hydrocarbons, nitrosamines) are excreted in urine, producing prolonged epithelial exposure. Risk is dose-dependent; current smokers have a two- to fourfold increased risk versus never-smokers. Risk declines substantially but not to baseline after cessation.
Occupational Exposures
Aromatic amines (2-naphthylamine, benzidine, 4-aminobiphenyl) used in the dye, rubber, textile, leather, and printing industries carry two- to tenfold increased risk. Hairdressers, painters, truck drivers, and machinists have elevated risk. Latency from exposure to cancer development averages 20–45 years.
Schistosomiasis
Schistosoma haematobium infestation causes chronic bladder inflammation and squamous metaplasia, accounting for the predominantly squamous cell carcinoma histology in endemic regions (Egypt, sub-Saharan Africa).
Chronic Urinary Tract Infections and Stones
Recurrent bacterial UTIs and bladder calculi are associated with squamous cell carcinoma, particularly in patients with long-term urinary catheters or neurogenic bladder.
Medications and Chemicals
- Cyclophosphamide: Alkylating agent; acrolein metabolite causes urothelial damage; mesna prophylaxis mitigates risk.
- Pioglitazone: Thiazolidinedione diabetes medication; associated with 40% increased bladder cancer risk in long-term users (EMA label warning).
- Aristolochic acid: Herbal nephropathy and urothelial carcinoma (upper tract predominance).
- Chlornaphazine: Withdrawn alkylating agent; historical association.
Genetic and Inherited Factors
- NAT2 slow acetylator phenotype: Reduced ability to detoxify aromatic amines; modified risk from occupational exposure.
- GSTM1 null genotype: Reduced glutathione S-transferase-mediated carcinogen detoxification.
- Lynch syndrome (HNPCC): MSH2 and MLH1 mutations confer increased upper tract urothelial carcinoma risk; bladder cancer risk also elevated.
- First-degree family history: Two- to threefold increased risk.
Other Risk Factors
- Pelvic radiation therapy (prior cervical, prostate, or rectal cancer treatment)
- Chronic bladder irritation (neurogenic bladder, long-term catheterization)
- Low fluid intake (reduced urinary dilution of carcinogens)
5. Clinical Presentation and TNM Staging
Clinical Presentation
Painless gross hematuria is the presenting symptom in approximately 85% of patients. Hematuria may be intermittent; its absence on a single urinalysis does not exclude bladder cancer. Microscopic hematuria (three or more red blood cells per high-power field on two of three urinalyses) warrants urological evaluation in high-risk patients.
Additional presenting symptoms include:
- Irritative LUTS (frequency, urgency, dysuria) — particularly associated with carcinoma in situ
- Flank pain from ureteric obstruction (locally advanced disease)
- Pelvic mass or lymphadenopathy (advanced disease)
- Constitutional symptoms (weight loss, fatigue, bone pain) in metastatic disease
TNM Staging (AJCC 8th Edition, 2017)
Primary Tumor (T):
- Ta: Non-invasive papillary carcinoma (confined to urothelium)
- Tis (CIS): Carcinoma in situ (flat, high-grade)
- T1: Invasion into lamina propria (subepithelial connective tissue)
- T2a: Invasion into superficial muscularis propria (inner half)
- T2b: Invasion into deep muscularis propria (outer half)
- T3a: Microscopic perivesical tissue invasion
- T3b: Macroscopic perivesical tissue invasion (extravesical mass)
- T4a: Invasion of prostate stroma, seminal vesicles, uterus, or vagina
- T4b: Invasion of pelvic wall or abdominal wall
Regional Lymph Nodes (N):
- N0: No lymph node metastasis
- N1: Single node in true pelvis (hypogastric, obturator, external iliac, or presacral)
- N2: Multiple nodes in true pelvis
- N3: Common iliac lymph nodes
Distant Metastasis (M):
- M0: No distant metastasis
- M1a: Non-regional lymph nodes
- M1b: Distant metastasis (bone, lung, liver most common)
Stage groupings: Stage 0a (Ta N0 M0), Stage 0is (Tis N0 M0), Stage I (T1 N0 M0), Stage II (T2 N0 M0), Stage IIIA (T3–T4a N0 M0 or T1–T4a N1 M0), Stage IIIB (T1–T4a N2–N3 M0), Stage IVA (T4b any N M0 or any T N any M1a), Stage IVB (any T any N M1b).
Risk Stratification of NMIBC
European Association of Urology (EAU) risk stratification for NMIBC guides surveillance and adjuvant treatment intensity:
- Low risk: Primary solitary Ta low-grade, less than 3 cm, no CIS
- Intermediate risk: Multiple or recurrent low-grade Ta; high-grade Ta less than 3 cm, no CIS
- High risk: T1 high-grade; any CIS; multiple/large/recurrent high-grade Ta; variant histology; lymphovascular invasion; BCG failure
- Very high risk: T1 high-grade + CIS; T1 high-grade in diverticulum; multiple large T1 high-grade; variant histology (micropapillary, plasmacytoid, neuroendocrine)
6. Diagnosis — Cystoscopy, Urodynamics, and Biomarkers
Urine Cytology
Exfoliative urine cytology detects shed malignant cells. Reported using the 2016 Paris System for Reporting Urinary Cytology. High specificity (approximately 95%) for high-grade urothelial carcinoma and CIS; poor sensitivity (approximately 15–40%) for low-grade tumors. Voided urine specimens from three consecutive morning voids maximize yield; catheterized specimens improve sensitivity.
Cystoscopy
The gold standard for bladder cancer diagnosis. Flexible cystoscopy under local anesthesia is the initial diagnostic procedure for hematuria evaluation or surveillance. Findings are described using standardized TURBT reporting forms: number, size, location, morphology (papillary, sessile, flat), and presence of CIS (erythematous velvety patches). Narrow band imaging (NBI) and photodynamic diagnosis (PDD/blue light cystoscopy, Cysview/hexaminolevulinate) enhance detection of flat lesions and CIS, reducing false-negative rates.
Transurethral Resection of Bladder Tumor (TURBT)
Diagnostic and therapeutic procedure performed under regional or general anesthesia. Systematic sampling of tumor base including detrusor muscle is mandatory for accurate T staging. Re-TURBT (repeat resection within 4–6 weeks) is recommended for T1 high-grade tumors (to confirm staging, given 30–45% upstaging rates on re-resection) and incompletely resected tumors. PDD-guided TURBT reduces residual tumor and recurrence rates.
Upper Tract Imaging
CT urography (CTU) is the preferred imaging modality, providing simultaneous evaluation of the upper tracts, lymph nodes, and distant organs. Indicated for hematuria evaluation and staging. MRI urography is an alternative in patients with contrast allergies or renal insufficiency. Retrograde pyelography and ureteroscopy evaluate the upper urinary tract when CTU is indeterminate.
Urine-Based Molecular Biomarkers
Several FDA-approved urine biomarker tests supplement cytology:
- NMP22 (Nuclear Matrix Protein 22): Elevated in bladder cancer; higher sensitivity than cytology for low-grade tumors; lower specificity due to inflammation-related false positives.
- BTA stat and BTA TRAK (Bladder Tumor Antigen): Detect complement factor H-related protein; FDA-approved for surveillance.
- FISH (Fluorescence In Situ Hybridization, UroVysion): Detects chromosomal abnormalities (gains of chromosomes 3, 7, 17 and loss of 9p21 CDKN2A). High specificity for high-grade disease; useful for evaluating equivocal cytology.
- ImmunoCyt (uCyt+): Immunofluorescence assay detecting mucin and carcinoembryonic antigen-related markers on exfoliated cells; combined with cytology improves sensitivity.
- Cxbladder: Multigene mRNA expression test with high sensitivity for ruling out high-grade disease.
Staging Imaging for MIBC
CT chest/abdomen/pelvis with intravenous contrast is standard for MIBC staging. MRI pelvis (mpMRI) provides superior soft tissue characterization for assessing perivesical extension, prostate stroma invasion, and lymph node involvement. Bone scan is reserved for bone pain symptoms or elevated alkaline phosphatase. PET-CT with fluorodeoxyglucose (FDG) is increasingly used in MIBC staging, particularly for lymph node evaluation.
7. Treatment
Non-Muscle-Invasive Bladder Cancer (NMIBC)
TURBT: Initial resection is both diagnostic and potentially curative for low-risk disease.
Intravesical Chemotherapy:
- Single perioperative instillation (SPIMC): A single dose of intravesical mitomycin C (MMC, 40 mg) or gemcitabine administered within 24 hours of TURBT reduces recurrence rates by approximately 40% in low-risk NMIBC by ablating floating tumor cells. Contraindicated if bladder perforation is suspected.
- Adjuvant course: For intermediate-risk disease, 6–12 instillations of MMC, epirubicin, or gemcitabine over 6–12 months reduce recurrence rates.
BCG Immunotherapy:
Bacillus Calmette-Guerin (BCG) intravesical immunotherapy is the standard of care for high-risk NMIBC. Mechanism involves innate and adaptive immune activation at the bladder urothelium (TH1 response, NK cell activation, cytokine release). Induction course: weekly instillations for 6 weeks. Maintenance schedule (Lamm protocol): 3-weekly instillations at 3, 6, 12, 18, 24, 30, and 36 months. BCG maintenance for 1–3 years reduces progression to MIBC by 30–40%. Contraindicated in immunosuppressed patients and after traumatic catheterization. Side effects: irritative LUTS (common, self-limited), BCG cystitis, systemic BCGosis (rare, requires anti-tuberculous therapy).
BCG-Unresponsive NMIBC: Defined as high-grade disease persisting or recurring within 6 months of adequate BCG therapy. Options include:
- Radical cystectomy (definitive; recommended for BCG-unresponsive T1HG disease)
- Pembrolizumab (FDA-approved 2020): PD-1 checkpoint inhibitor; complete response rate approximately 41% in BCG-unresponsive CIS at 3 months
- Nadofaragene firadenovec (Adstiladrin, FDA-approved 2022): Intravesical adenoviral gene therapy delivering IFN-alpha; complete response rate approximately 53% in CIS
- Nogapendekin alfa inbakicept (IL-15 agonist) + BCG: Combination under regulatory review
Muscle-Invasive Bladder Cancer (MIBC)
Neoadjuvant Cisplatin-Based Chemotherapy (NAC): Standard of care preceding radical cystectomy for eligible patients. Cisplatin/gemcitabine (GC) or MVAC (methotrexate/vinblastine/doxorubicin/cisplatin) regimens achieve pathologic complete response (pT0) in 25–35% of patients. A meta-analysis demonstrated a 5% absolute 5-year survival benefit from NAC. CISH/ddMVAC (dose-dense MVAC) achieves higher pCR rates with less toxicity than standard MVAC.
Radical Cystectomy (RC): Gold standard treatment for MIBC. In men: removal of bladder, prostate, seminal vesicles, and proximal urethra. In women: anterior exenteration (bladder, uterus, ovaries, anterior vaginal wall). Bilateral pelvic lymph node dissection with extended template (above the aortic bifurcation) improves staging and may offer therapeutic benefit. Open, laparoscopic, and robotic-assisted approaches demonstrate equivalent oncological outcomes; robotic approach reduces blood loss and length of stay.
Urinary Diversion after Cystectomy:
- Ileal conduit (Bricker): Simplest; segment of ileum serves as conduit draining to skin stoma requiring external appliance.
- Orthotopic neobladder: Detubularized ileal or ileocolonic reservoir anastomosed to the urethra; allows per-urethral voiding; requires intact sphincter mechanism; daytime continence approximately 85–90%, nighttime 70–80%.
- Continent cutaneous diversion (Indiana pouch): Catheterizable abdominal stoma; appropriate when orthotopic diversion is not feasible.
Trimodality Therapy (TMT): Bladder-preserving approach combining maximal TURBT, concurrent radiosensitizing chemotherapy (cisplatin, MMC/5-FU, or gemcitabine), and external beam radiation therapy (64–65 Gy). Appropriate for carefully selected cT2 patients unwilling or unfit for cystectomy, achieving 5-year disease-specific survival of approximately 60–65% in selected series. Requires close cystoscopic surveillance for recurrence.
Metastatic Bladder Cancer
- First-line cisplatin-based chemotherapy: Gemcitabine/cisplatin (GC) is the global standard (equivalent efficacy to MVAC with better tolerability). Cisplatin eligibility criteria: ECOG PS 0–1, GFR above 60 mL/min, hearing PN below grade 2, no NYHA Class III/IV heart failure.
- Carboplatin-based regimens: For cisplatin-ineligible patients; GC with carboplatin substitution or gemcitabine/carboplatin; inferior objective response rates.
- Immune checkpoint inhibitors (ICIs):
- Pembrolizumab: FDA-approved first-line in cisplatin-ineligible patients with PD-L1 CPS ≥10, and second-line after platinum-based chemotherapy (KEYNOTE-045).
- Atezolizumab: First-line in cisplatin-ineligible, PD-L1 high (IC2/3); second-line approval.
- Avelumab: FDA-approved first-line maintenance after platinum-based chemotherapy regardless of PD-L1 (JAVELIN Bladder 100).
- Antibody-drug conjugates (ADCs):
- Enfortumab vedotin (EV): Nectin-4-targeted; FDA-approved second-line and first-line (in combination with pembrolizumab, EV+P, FDA-approved 2024 as first-line for all metastatic UC regardless of cisplatin eligibility based on EV-302 trial showing superiority over platinum chemotherapy).
- Sacituzumab govitecan: TROP2-targeted; active in post-platinum/ICI setting.
- FGFR3 inhibitors: Erdafitinib (FDA-approved 2019): First targeted therapy approved for bladder cancer; for patients with FGFR2/3 alterations progressing after platinum-based chemotherapy; objective response rate approximately 40%.
8. Complications
- Disease recurrence and progression: Ta/T1 tumors recur in 50–70% within 5 years; 10–30% progress to MIBC.
- Ureteral obstruction: Local tumor growth or lymphadenopathy; may cause hydronephrosis and renal failure.
- Complications of TURBT: Bladder perforation (intra- or extraperitoneal), hemorrhage, TUR syndrome (rare in bladder versus prostate).
- BCG complications: Granulomatous cystitis, prostatitis, epididymo-orchitis, BCG sepsis, pulmonary or hepatic granulomas requiring systemic isoniazid treatment.
- Cystectomy complications: Major early complications include hemorrhage, bowel injury, and thromboembolic events; late complications include ureteroenteric anastomotic stricture, reservoir complications (stone formation, mucus plugging), and continent neobladder hypercontinence or urinary retention.
- Radiation therapy complications: Radiation cystitis (hematuria, frequency, urgency), proctitis, vaginal stenosis, fistula formation.
- Chemotherapy toxicity: Cisplatin: nephrotoxicity, ototoxicity, peripheral neuropathy, myelosuppression; carboplatin: myelosuppression predominates.
- Upper tract recurrence: Metachronous upper tract urothelial carcinoma in 2–4% of bladder cancer survivors (especially with CIS or upper tract involvement).
9. Prognosis
Prognosis varies dramatically by stage at presentation. Five-year survival rates by AJCC stage:
- Stage 0 (Ta/Tis): Approximately 95–98%
- Stage I (T1): Approximately 80–90%
- Stage II (T2): Approximately 55–70%
- Stage III (T3/T4a, N0/N1): Approximately 30–50%
- Stage IV (T4b or N2/N3 or M1): Approximately 5–15% (M1 less than 5%)
Pathologic complete response (pT0N0) at cystectomy following NAC is associated with a 5-year disease-specific survival exceeding 85%. The paradigm-shifting EV-302 trial (2023) demonstrated that enfortumab vedotin plus pembrolizumab achieves a median overall survival of 31.5 months in metastatic UC compared to 16.1 months with GC, fundamentally redefining first-line metastatic treatment.
10. Prevention
- Smoking cessation: The single most impactful preventive intervention; attributable fraction of bladder cancer from tobacco exceeds 50% in men.
- Occupational exposure reduction: Engineering controls, personal protective equipment, and elimination of carcinogenic aromatic amines from industrial processes.
- Adequate hydration: Higher fluid intake dilutes urine carcinogen concentrations; each 240 mL increment in fluid intake associated with approximately 7% reduced bladder cancer risk.
- Schistosomiasis control: Praziquantel treatment, sanitation, and vector control programs in endemic regions.
- Cyclophosphamide mesna co-administration: Prevents acrolein-induced urothelial toxicity in patients requiring alkylating agents.
- Dietary factors: Cruciferous vegetables (sulforaphane), green tea polyphenols, selenium, and vitamin E have demonstrable in vitro anti-urothelial carcinogenesis properties, though clinical trial evidence for supplementation is limited.
11. Recent Research and Advances
EV+Pembrolizumab (EV-302/KEYNOTE-A39 trial): The combination of enfortumab vedotin plus pembrolizumab demonstrated superiority over gemcitabine/platinum chemotherapy in first-line metastatic urothelial carcinoma across all subgroups in a phase III randomized trial, achieving median OS of 31.5 versus 16.1 months (HR 0.47). This represents the most significant advance in first-line metastatic bladder cancer in over 30 years.
Perioperative immunotherapy: Multiple trials (NIAGARA, JAVELIN Bladder 100) are evaluating durvalumab, avelumab, and pembrolizumab in perioperative MIBC settings. NIAGARA demonstrated improved event-free survival with durvalumab added to NAC and maintenance.
Liquid biopsy and circulating tumor DNA (ctDNA): ctDNA detection after TURBT or radical cystectomy predicts metastatic relapse with high sensitivity, potentially enabling minimal residual disease (MRD)-guided therapy. ATLAS and IMvigor011 trials are investigating ctDNA-selected adjuvant immunotherapy.
Novel intravesical devices: Continuous low-dose intravesical delivery via thermosensitive hydrogels (MitoGel, vicinium) is being evaluated in NMIBC, including BCG-unresponsive disease.
FGFR inhibition: Second-generation pan-FGFR inhibitors (pemigatinib, infigratinib) and FGFR3-targeted ADCs are in clinical development for the approximately 20% of MIBC and 70% of low-grade NMIBC with FGFR3 alterations.
12. References
- Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17–48.
- Babjuk M, Burger M, Capoun O, et al. European Association of Urology guidelines on non-muscle-invasive bladder cancer (Ta, T1, and Carcinoma in Situ). Eur Urol. 2022;81(1):75–94.
- Alfred Witjes J, Bruins HM, Cathomas R, et al. European Association of Urology guidelines on muscle-invasive and metastatic bladder cancer. Eur Urol. 2021;79(1):82–104.
- Chang SS, Bochner BH, Chou R, et al. Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/ASTRO/SUO guideline. J Urol. 2017;198(3):552–559.
- Powles T, Csoszi T, Ozguroglu M, et al. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361). Lancet Oncol. 2021;22(7):931–945.
- Bellmunt J, de Wit R, Vaughn DJ, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376(11):1015–1026.
- Powles T, Valderrama BP, Gupta S, et al. Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer. N Engl J Med. 2024;390(10):875–888.
- Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003;349(9):859–866.
- Lamm DL, Blumenstein BA, Crissman JD, et al. Maintenance bacillus Calmette-Guerin immunotherapy for recurrent TA, T1 and carcinoma in situ transitional cell carcinoma of the bladder. J Urol. 2000;163(4):1124–1129.
- Witjes JA, Moonen PM, van der Heijden AG. Comparison of hexaminolevulinate-based flexible and rigid fluorescence cystoscopy with white light cystoscopy in bladder cancer detection. Eur Urol. 2005;47(3):319–322.
- The Cancer Genome Atlas Research Network. Comprehensive molecular characterization of urothelial bladder carcinoma. Nature. 2014;507(7492):315–322.
- Sylvester RJ, van der Meijden AP, Oosterlinck W, et al. Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables. Eur Urol. 2006;49(3):466–477.
- Balar AV, Galsky MD, Rosenberg JE, et al. Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma. Lancet. 2017;389(10064):67–76.
- Rosenberg JE, Hoffman-Censits J, Powles T, et al. Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy. Lancet. 2016;387(10031):1909–1920.
- Black PC, Dinney CPN, Kassouf W, et al. The society of urologic oncology (SUO) annual meeting: 2022 highlights. Urol Oncol. 2023;41(2):66–78.
- Moschini M, D'Andrea D, Korn S, et al. Characteristics and clinical significance of histological variants of bladder cancer. Nat Rev Urol. 2017;14(11):651–668.
Research Papers
- Clinical trials on bladder cancer (urothelial carcinoma) — PubMed search
- Intravesical BCG immunotherapy for NMIBC — PubMed search
- Enfortumab vedotin + pembrolizumab in urothelial cancer — PubMed search
- Radical cystectomy for muscle-invasive bladder cancer — PubMed search
- Neoadjuvant cisplatin chemotherapy in MIBC — PubMed search
- FGFR3 inhibitors (erdafitinib) in bladder cancer — PubMed search
- Smoking and bladder cancer risk — PubMed search
- Hematuria workup and cystoscopy for bladder cancer — PubMed search
Connections
- Urinary Tract Infections
- Selenium
- Green Tea
- Kidney Stones
- Interstitial Cystitis
- Vitamin D3
- Cancer Overview
- NAC
- NAC and Glutathione
- Urinalysis
- Benign Prostatic Hyperplasia
- Prostate Conditions
- Diabetes
- Vitamin E
- Artificial Sweeteners
- Arsenic
- Cancer Prevention
Featured Videos

Bladder Cancer Signs and Symptoms (what to look out for)

Bladder Cancer - Overview (types, pathophysiology, diagnosis, treatment)

Bladder cancer detection and treatment | Ohio State Medical Center
Bladder Cancer: Basics of Diagnosis, Workup, Pathology, and Treatment
Bladder Cancer, Causes, Signs and Symptoms, Diagnosis and Treatment.

Mary Beth's Bladder Cancer Journey

I Thought My UTIs Were Normal - It Was CANCER! - Monica | Bladder Cancer | The Patient Story

Bladder Cancer 101 | Symptoms, Diagnosis & Treatment

Bladder Cancer: What to Know If You’ve Just Been Diagnosed