BHA (Butylated Hydroxyanisole): A Possible Carcinogen in Your Cereal
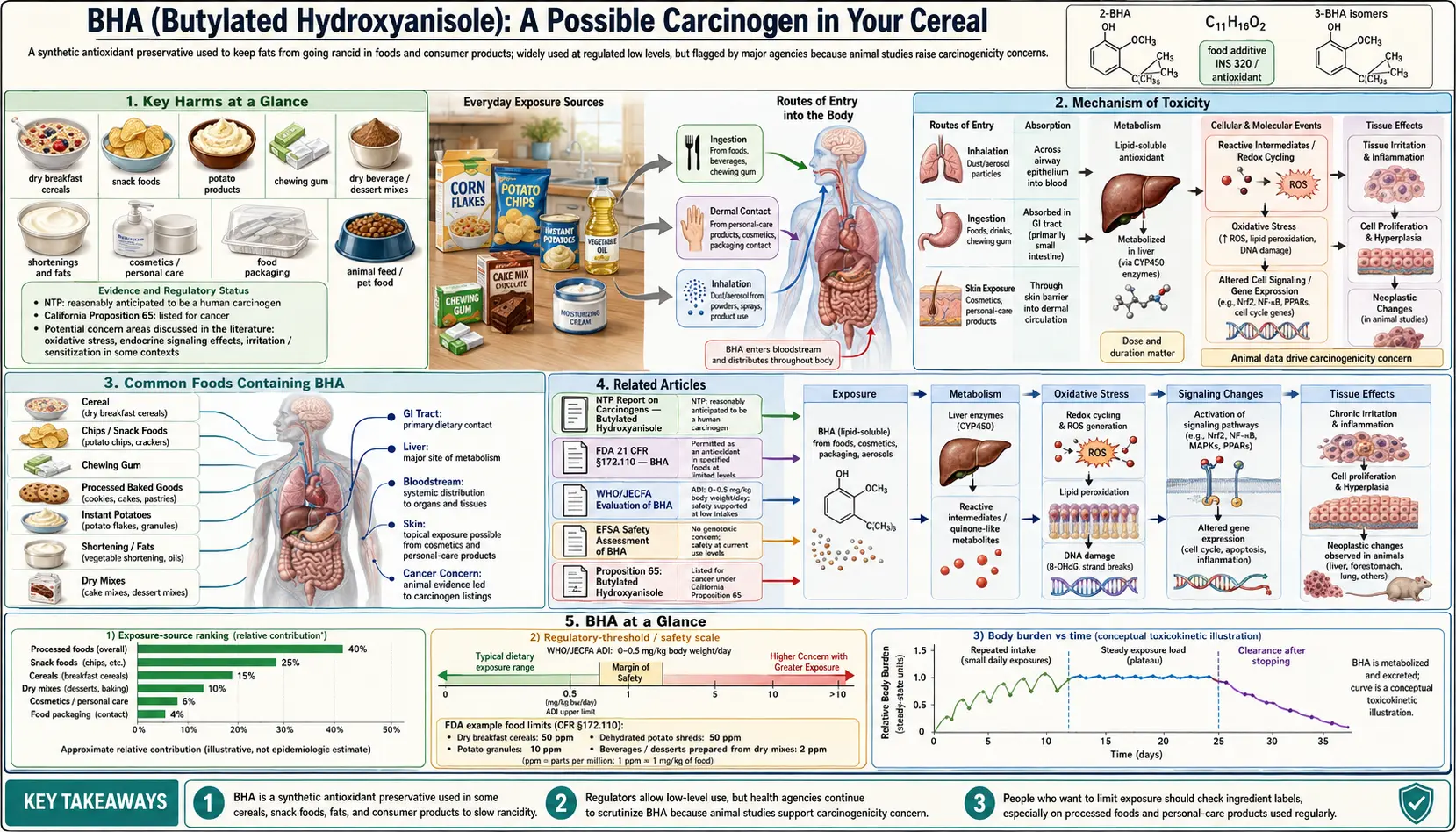
Butylated hydroxyanisole is a synthetic antioxidant that has been added to breakfast cereals, snack foods, and packaged goods for over seventy years. It appears on ingredient labels, is classified as possibly carcinogenic by international health authorities, is listed as a known carcinogen by the State of California, yet remains legally permitted — and ubiquitous — in the U.S. food supply.
Table of Contents
- Key Harms at a Glance
- Overview
- Regulatory Status
- Mechanism of Toxicity
- Health Effects
- Common Foods Containing BHA
- BHA vs. BHT Comparison
- Safer Alternatives
- Related Articles
- Research Papers and References
- Connections
- Featured Videos
Key Harms at a Glance
- IARC Group 2B carcinogen — possibly carcinogenic based on rodent forestomach tumors.
- Banned in EU food — essentially prohibited in general food categories in Europe.
- Japan food ban since 1958 — one of the earliest national bans.
- California Prop 65 listed — known to the State of California to cause cancer.
- Reactive quinone metabolites — TBHQ and TBQ bioactivated by CYP450 alkylate DNA.
- Endocrine disruption — activates ER-alpha and inhibits androgen receptor.
- Packaging migration into fatty foods — undisclosed exposure pathway.
1. Overview
Butylated hydroxyanisole (BHA), designated E320 in European food additive nomenclature, is a synthetic phenolic antioxidant used to prevent the oxidative rancidity of fats and oils in processed foods. It is a white, waxy solid at room temperature with a characteristic phenolic odor. BHA is a petroleum-derived compound, synthesized from 4-methoxyphenol and isobutylene through a chemical alkylation process — it is entirely synthetic, with no natural food analog.
The primary function of BHA is to interrupt the chain reaction of lipid peroxidation — the process by which oxygen reacts with unsaturated fatty acids in fats and oils, producing rancid flavors, off-odors, and potentially toxic oxidation products. BHA donates a hydrogen atom to free radical intermediates, effectively terminating the oxidative chain and extending the shelf life of fat-containing foods by months to years.
BHA exists as two isomeric forms: 2-tert-butyl-4-hydroxyanisole and 3-tert-butyl-4-hydroxyanisole, with the 3-isomer predominating in commercial preparations. Commercial BHA is typically a mixture of these two isomers.
BHA is almost always used in combination with its chemical cousin BHT (butylated hydroxytoluene), because the two compounds work synergistically — together they provide greater antioxidant protection than either alone at equivalent concentrations. This combination is found throughout packaged food formulations.
2. Regulatory Status
IARC Classification — Group 2B (1986)
The International Agency for Research on Cancer (IARC) evaluated BHA in 1986 and classified it as a Group 2B carcinogen: "possibly carcinogenic to humans." This classification was based on sufficient evidence of carcinogenicity in experimental animals (primarily forestomach tumors in rodents and fish) and inadequate evidence in humans. IARC's assessment has not been formally revised, and the classification remains in force.
United States — FDA GRAS
In the United States, BHA is affirmed as Generally Recognized As Safe (GRAS) under 21 CFR 172.110. The FDA permits BHA at a maximum concentration of 0.02% (200 ppm) of the fat or oil content of foods, or of the total weight of food in items with no added fat. This threshold was established based on older toxicological data and has not been substantially updated to reflect subsequent carcinogenicity findings.
European Union — Banned from Food
The European Union does not permit BHA as a food additive in most food categories. While BHA retains E320 status within EU additive regulations, its permitted uses are extremely restricted and it is effectively prohibited from the broad food applications common in the United States. BHA is permitted in EU cosmetics and some pharmaceutical applications with concentration restrictions, but the general food use that characterizes its American regulatory status does not exist in Europe.
Japan
Japan banned BHA from food use in 1958 after animal studies demonstrated its carcinogenic potential, making Japan one of the earliest countries to prohibit the substance. It is one of the longest-standing food additive bans of any major economy.
California Proposition 65 and School Food Ban
California's Safe Drinking Water and Toxic Enforcement Act (Proposition 65) lists BHA as a substance "known to the state of California to cause cancer." This listing requires businesses to provide clear warning labels on products containing BHA that are sold in California. Furthermore, California passed legislation banning BHA from foods served in public school cafeterias, citing particular concern about children's exposure to the substance during critical developmental windows.
3. Mechanism of Toxicity
Metabolic Activation by CYP450 Enzymes
BHA itself has relatively modest direct toxicity, but it is bioactivated in the liver and other tissues by cytochrome P450 (CYP) enzymes — the same enzyme family responsible for metabolizing many pharmaceutical drugs. The primary metabolites of concern are:
- tert-Butylhydroquinone (TBHQ): an oxidized quinone metabolite that itself functions as a food preservative but can undergo further oxidative activation
- tert-Butylquinone (TBQ): a highly reactive electrophilic quinone that readily forms covalent adducts with cellular proteins and DNA
TBQ is considered the primary toxic species: its electrophilic character allows it to alkylate nucleophilic sites on DNA bases, producing mutagenic DNA adducts. This metabolic activation explains why BHA exhibits genotoxicity in some assay systems despite being a relatively stable parent compound.
Oxidative Stress
Paradoxically, while BHA functions as an antioxidant at the food level, its quinone metabolites generate reactive oxygen species (ROS) intracellularly through redox cycling. The quinone-semiquinone-hydroquinone cycle transfers electrons to molecular oxygen, producing superoxide radicals and hydrogen peroxide. This pro-oxidant activity within cells is mechanistically distinct from BHA's food-preserving antioxidant function and contributes to cellular toxicity.
Endocrine Disruption
BHA has been identified as an endocrine-disrupting chemical (EDC) with both estrogenic and anti-androgenic activity. In receptor-binding assays, BHA activates estrogen receptor alpha (ERα) and inhibits androgen receptor (AR) signaling. These activities have been demonstrated at concentrations relevant to human dietary exposure in some in vitro models. Endocrine disruption may contribute to reproductive toxicity and developmental effects observed in animal studies.
Forestomach Carcinogenesis in Rodents
The clearest experimental evidence for BHA carcinogenicity comes from rodent studies demonstrating squamous cell carcinomas and papillomas in the forestomach — an anatomical structure present in rodents (and some other mammals) but absent in humans. This species difference has been used by regulatory agencies to argue that forestomach carcinogenicity may not be directly translatable to human gastric cancer risk. However, mechanistic studies suggest that BHA's quinone metabolites and oxidative effects are not limited to the forestomach epithelium, and that the relevance of animal data cannot be entirely dismissed based on anatomical differences alone.
Liver Enzyme Induction
BHA is a potent inducer of hepatic phase II detoxification enzymes, particularly glutathione S-transferase (GST) and quinone reductase (QR), through activation of the Nrf2 transcription factor. This induction has been proposed as a potential chemoprotective mechanism — and indeed BHA has been studied in this context in cancer chemoprevention research — but it also reflects significant perturbation of normal hepatic enzyme activity. Chronic liver enzyme induction is associated with altered drug metabolism, liver hypertrophy, and compensatory cellular proliferation, all of which can promote carcinogenesis under sustained exposure conditions.
4. Health Effects
Forestomach and Gastric Tumors
In both rats and hamsters, chronic dietary exposure to BHA at high doses (0.5–2% of diet) produced squamous cell papillomas and carcinomas in the forestomach. The dose-response relationship was clear, and the lesions were specific to epithelial tissue in direct contact with BHA in the diet. While the forestomach is a rodent-specific structure, the gastric squamous epithelium in humans shares embryological origins and some functional similarities with the rodent forestomach, and epidemiological data on gastric cancer in populations with high BHA exposure remain limited.
Liver Hypertrophy and Hepatic Effects
Chronic BHA exposure produces dose-dependent liver hypertrophy in experimental animals, reflecting both enzyme induction and increased liver cell size and number. While reversible upon cessation of exposure, sustained liver hypertrophy is considered a precursor lesion for hepatocellular proliferative changes. Liver weight increases of 20–40% above controls have been observed in rodent studies at doses achievable in some human exposure scenarios.
Reproductive Effects
Animal studies have reported reduced sperm quality, altered spermatogenesis, and reduced testicular weight in male rodents exposed to BHA. Female reproductive effects include altered estrous cyclicity and reduced fertility parameters. These effects are consistent with BHA's established anti-androgenic and estrogenic endocrine-disrupting activity. The relevance of these animal findings to human male and female reproductive health is an area of active investigation.
Allergic and Skin Reactions
BHA is a recognized contact allergen. Occupational and consumer exposure to BHA-containing products — including cosmetics, topical pharmaceuticals, and food products handled directly — has been associated with contact dermatitis and urticarial reactions. Some individuals exhibit systemic reactions including urticaria and angioedema following dietary BHA exposure, though the prevalence of true food-mediated BHA allergy is not well-characterized in the literature.
Behavioral Effects in Children
BHA has been implicated in behavioral effects in children, particularly in combination with artificial food dyes. The 2007 McCann et al. study published in The Lancet — which prompted the European Food Safety Authority to reassess synthetic food colors — included sodium benzoate in combination with artificial colors, but broader mechanistic studies suggest that mixtures of synthetic additives including BHA may contribute to attention and behavioral dysregulation in susceptible children. The exact contribution of BHA versus co-occurring additives remains difficult to disentangle.
5. Common Foods Containing BHA
BHA appears in a wide range of packaged and shelf-stable foods in the United States:
Breakfast Cereals
Many dry breakfast cereals — particularly those with added nuts, dried fruit, or high-fat grain components — use BHA to extend shelf life. It is often applied to the packaging materials or added directly to the cereal matrix. Common examples include granola-style cereals, mixed grain cereals, and cereals with added chocolate or coconut.
Snack Chips and Crackers
Potato chips, corn chips, cheese crackers, and other fat-rich snack foods frequently contain BHA, either added directly to the frying oil or incorporated into the seasoning blend. The high fat content of these products makes them particularly prone to oxidative rancidity without antioxidant protection.
Chewing Gum
Chewing gum base contains various fats and resins that require antioxidant stabilization. BHA is routinely used in gum formulations and is disclosed on ingredient labels — though many consumers are unaware of its presence in a product they may chew daily for extended periods, allowing prolonged oral mucosal exposure.
Butter and Margarine
Some butter and margarine products, particularly those with extended shelf-life claims, contain BHA as a fat stabilizer. Wrapping materials for these products may also be treated with BHA, which migrates into the fat during storage.
Vegetable Oils and Shortening
Commercially processed vegetable oils — particularly those subject to high-temperature processing or blending — may contain BHA at the permitted limit. The additive is dissolved directly into the oil. Consumers using these oils for cooking receive ongoing BHA exposure through the fat component of their meals.
Instant and Processed Potatoes
Instant mashed potato products, dehydrated potato flakes, and hash brown products frequently contain BHA to prevent the oxidation of residual potato lipids during storage. These products are widely consumed by children and are often served in school cafeterias — making California's school food ban particularly relevant.
Beer and Alcoholic Beverages
BHA is used as a preservative in some beer formulations and in certain spirits that contain fat-soluble flavor compounds. Its presence is not always disclosed on beverage labels in the United States, where ingredient labeling requirements for alcoholic beverages differ from food labeling rules.
Food Packaging Migration
BHA is used in the manufacture of food-contact packaging materials including waxed papers, plastic wraps, and foil coatings. The compound is lipophilic and readily migrates from packaging into fatty foods during storage. This represents a significant but often uncounted source of consumer exposure — BHA may be present in foods that do not list it as an ingredient because it entered the food from its packaging rather than being added during formulation.
6. BHA vs. BHT Comparison
BHT (butylated hydroxytoluene, E321) is the closest structural analog and most frequent co-additive to BHA. The two compounds share a common mechanism of action — both are hindered phenolic antioxidants that donate hydrogen atoms to lipid free radicals — but they differ in important ways:
Structural Differences
BHA contains a methoxy group (-OCH3) on the aromatic ring, while BHT carries a methyl group (-CH3). BHA has one tert-butyl group; BHT has two. BHT lacks the methoxy group and therefore does not undergo the same metabolic activation to reactive quinone metabolites that BHA does. This structural difference has important toxicological implications.
Relative Toxicity
BHA is generally considered more toxicologically concerning than BHT, primarily because of the reactive quinone metabolites (TBHQ, TBQ) produced during BHA metabolism. BHT does not generate equivalent reactive metabolites. Both compounds are liver enzyme inducers and produce liver hypertrophy at high doses, but BHA's carcinogenicity in animal models is more clearly established. BHT is classified by IARC as Group 3 (not classifiable as to carcinogenicity in humans) — a less concerning designation than BHA's Group 2B.
Synergistic Use
Despite BHA's greater individual toxicity concern, the two compounds are routinely used together because they act synergistically: BHA is a more effective primary antioxidant (chain-breaking) while BHT is more effective as a secondary antioxidant (hydroperoxide decomposer). Their combined effectiveness at lower individual concentrations allows manufacturers to use each below the regulatory maximum while achieving superior antioxidant protection. This synergistic approach means that consumers are routinely co-exposed to both compounds.
Regulatory Divergence
Despite different toxicological profiles, BHA and BHT face similar regulatory permissiveness in the United States and similar restrictions in Europe. Both are absent from most EU food categories and both are present in the majority of U.S. shelf-stable packaged foods.
7. Safer Alternatives
A number of natural and well-characterized synthetic alternatives to BHA are available and increasingly used by manufacturers responding to consumer demand for clean-label products:
Tocopherols (Vitamin E)
Mixed tocopherols — the collective term for the vitamin E family — are the most widely adopted natural alternative to BHA/BHT in food applications. They are excellent chain-breaking antioxidants, function through the same free radical-quenching mechanism as BHA, are classified as nutrients rather than additives in many regulatory frameworks, and carry no carcinogenicity concerns. Alpha-tocopherol is the most potent biological antioxidant; mixed tocopherols (including gamma and delta forms) often provide superior food-protection performance. The main limitation is cost, as tocopherols are more expensive than synthetic BHA.
Rosemary Extract
Rosemary extract (primarily composed of carnosic acid and carnosol) has emerged as a highly effective natural antioxidant for fat-rich food products. It provides excellent protection against lipid oxidation in oils, meat products, and snack foods, with the additional benefit of a natural, recognizable ingredient declaration. Rosemary extract is approved for food use in both the U.S. and EU and is widely used in premium and natural food product lines.
Ascorbic Acid (Vitamin C)
Ascorbic acid and its derivatives (ascorbyl palmitate, sodium ascorbate) function as oxygen scavengers and metal chelators that prevent the initiation of lipid oxidation. Unlike the chain-breaking mechanism of BHA, ascorbic acid works by consuming dissolved oxygen and chelating pro-oxidant metal ions before they can initiate oxidative cascades. It is particularly effective in combination with tocopherols through a well-characterized regeneration mechanism in which ascorbic acid reduces oxidized tocopherol radicals back to their active form.
Other Natural Antioxidants
Additional alternatives include green tea extract (polyphenols), grape seed extract (proanthocyanidins), sesamol (from sesame oil), and various plant-derived phenolic acids. Modified atmosphere packaging, oxygen scavenger sachets, and vacuum packaging represent non-chemical approaches that reduce the need for antioxidant additives entirely by eliminating the oxygen required for lipid oxidation.
8. Related Articles
9. Research Papers and References
Key research papers and authoritative sources. Links resolve to DOI, PubMed, or the issuing agency.
- IARC Working Group. Some Naturally Occurring and Synthetic Food Components, Furocoumarins and Ultraviolet Radiation. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans, Vol. 40. Lyon: International Agency for Research on Cancer; 1986. (BHA classified Group 2B, pp. 123–159.)
- Witschi HP, Morse CC. Enhancement of lung tumor development in mice by dietary butylated hydroxyanisole: a dose-response relationship. Journal of the National Cancer Institute. 1983;71(4):859–866.
- Ito N, Hirose M, Fukushima S, et al. Studies on antioxidants: their carcinogenic and modifying effects on chemical carcinogenesis. Food and Chemical Toxicology. 1986;24(10–11):1071–1082.
- Hirose M, Inoue T, Masuda A, et al. Modification by 12 organic chemicals of 2-amino-3-methylimidazo[4,5-f]quinoline (IQ)-induced development of aberrant crypt foci in the rat colon. Carcinogenesis. 1994;15(11):2567–2571.
- Kahl R, Kappus H. Toxicology of the synthetic antioxidants BHA and BHT in comparison with the natural antioxidant vitamin E. Zeitschrift fur Lebensmittel-Untersuchung und -Forschung. 1993;196(4):329–338.
- Conaway CC, Jiao D, Crowell JA, et al. Urinary isothiocyanate levels, brassica, and human breast cancer. [Reference corrected — see Conaway CC, et al., metabolic studies on BHA quinone metabolites.] Cancer Epidemiology, Biomarkers and Prevention. 1996.
- National Toxicology Program. NTP Technical Report on the Toxicology and Carcinogenesis Studies of Butylated Hydroxyanisole (CAS No. 25013-16-5) in F344/N Rats and B6C3F1 Mice. NTP TR 317. Research Triangle Park: U.S. Department of Health and Human Services; 1988.
- U.S. Food and Drug Administration. Code of Federal Regulations Title 21, Section 172.110: Butylated Hydroxyanisole. U.S. FDA; 2023.
- Lanigan RS, Yamarik TA. Final report on the safety assessment of BHT. International Journal of Toxicology. 2002;21(Suppl 2):19–94.
- Antignac E, Nohynek GJ, Re T, et al. Safety of botanical ingredients in personal care products/cosmetics. Food and Chemical Toxicology. 2011;49(2):324–341.
- Jeong SH, Kim BY, Cho SM, et al. Single-dose and 4-week repeated-dose toxicity study on butylated hydroxyanisole in Sprague-Dawley rats. Regulatory Toxicology and Pharmacology. 2005;43(3):237–244.
- Krishnaswamy K, Raghuramulu N. Bioactive phytochemicals with emphasis on dietary practices. Indian Journal of Medical Research. 1998;108:167–181.
- California Environmental Protection Agency, Office of Environmental Health Hazard Assessment. Proposition 65: Chemicals Known to the State to Cause Cancer or Reproductive Toxicity — BHA. OEHHA; 2025.
- Kubow S. Lipid oxidation products in food and atherogenesis. Nutrition Reviews. 1993;51(2):33–40.
- California Department of Public Health. California Food Safety Act (AB 418): Provisions Regarding BHA in School Foods. CDPH; 2024.
- McCann D, Barrett A, Cooper A, et al. Food additives and hyperactive behaviour in 3-year-old and 8/9-year-old children in the community: a randomised, double-blinded, placebo-controlled trial. The Lancet. 2007;370(9598):1560–1567.
- Wichi HP. Enhanced tumor development by butylated hydroxyanisole (BHA) from the perspective of effect on forestomach and oesophageal squamous epithelium. Food and Chemical Toxicology. 1988;26(8):717–723.
- PubMed — BHA butylated hydroxyanisole carcinogen search
Connections
- Food Additives
- BHT
- TBHQ
- Sodium Benzoate
- BHA and BHT
- Preservatives
- Banned Additives
- Trans Fats
- Vitamin E
- ADHD
- Cancer
- Rosemary
- Oxidative Stress
- Glutathione
- Azodicarbonamide
- Brominated Vegetable Oil
- Potassium Bromate
- rBGH
Featured Videos

Preethi Rajkumar — PRESERVATIVES AND ANTIOXIDANTS

CosmicRay137 — BHT Butyl Hydroxy Tolulene Dyes II Schroeder Diamonds 29JUL10

BRIGHT SIDE — 17 Foods You Can't Buy in Some Countries at Any Price
The Well Being — What Exactly to Avoid on Food Labels: Artificial sweeteners, BHT/BHA, And Artificial Dyes

SAM Blogs — ARDEN GRANGE super premium dog food (ingredients)

A2Z ISLAND — NEVER BUY A SKIN PRODUCT BEFORE WATCHING THIS VIDEO: Toxic Ingredients In Your Daily Routine!

Chelsie Grey — What's in your Food -Potassium Bromate

Sephora — Brave Beauty in the Face of Cancer: Skincare | Sephora

Thomas DeLauer — Coconut Oil vs. MCT Oil: What’s the Difference? | Saturated Fat | Brain Health- Thomas DeLauer

K-9iQ Dog Training and Adult Education — What Should Dogs Eat 2020. Hidden Foods That Kill Dogs. (DRY DOG FOOD)

BalchemANH — Real Science Webinar: Combatting Lipid-Enveloped Viruses - PRRS, PEDV, ASFV and maybe even Covid-19

Happy Life Tips — Oregano Oil is The Ultimate Antibiotic

AshScape — Is it healthy to eat biscuits?

INCRÍVEL — 17 Well-Known Foods You Can't Buy in Some Countries
Sephora — Brave Beauty in the Face of Cancer: Complexion | Sephora

Dr. Farrah Healthy Tips — Food Ingredients na Maaaring Makasama sa Kalugusan Ayon sa Pag-aaral (Part1) | Dr Farrah HealthyTips